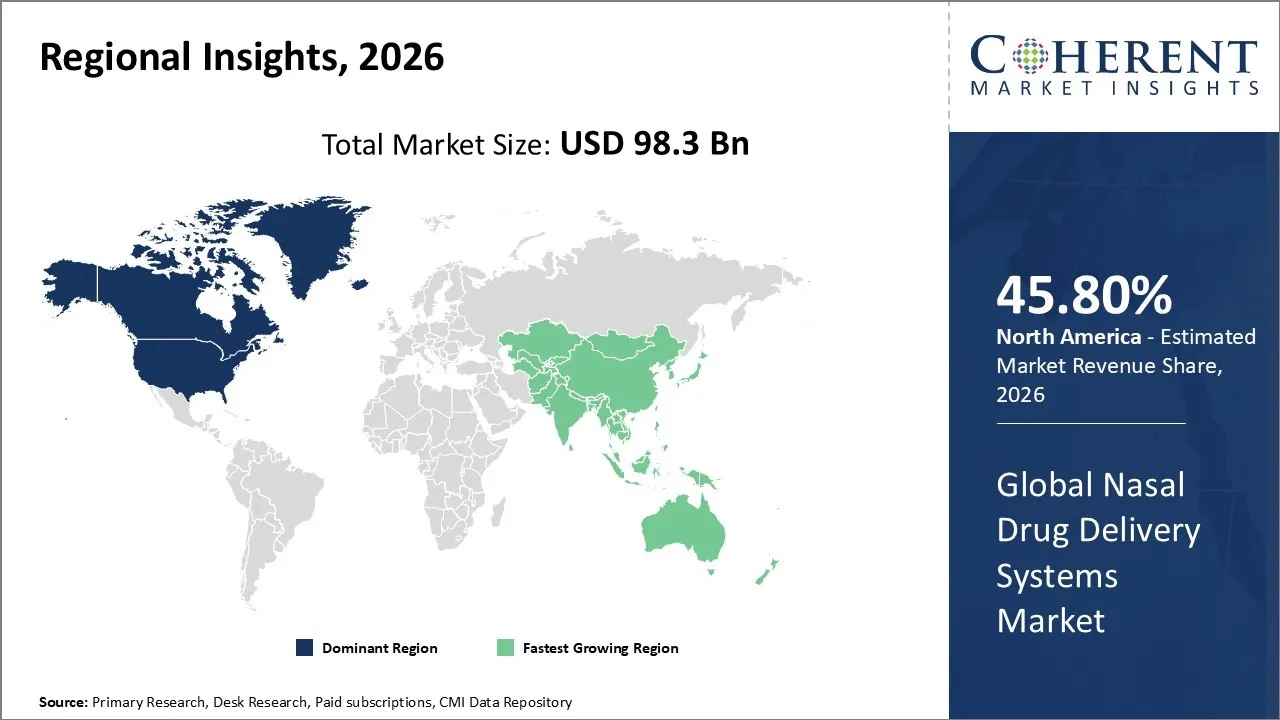
The Nasal Drug Delivery Systems Market is estimated to be valued at USD 98.3 Bn in 2026 and is expected to reach USD 134.67 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 8.2% from 2026 to 2033.
The Nasal Drug Delivery Systems Market is expanding rapidly as patients and healthcare providers increasingly prefer nasal routes for drug administration due to their non-invasive nature, fast action, and ease of use. The rising prevalence of respiratory disorders, allergies, and chronic conditions drives growth across segments such as nasal sprays, vaccines, and multi-dose systems. Continuous advances in formulations, smart delivery devices, and broader therapeutic applications are accelerating adoption, while hospitals, retail outlets, and pharmacies actively distribute these products to patients.
|
Current Events |
Description and its impact |
|
Geopolitical Tensions Affecting Supply Chains |
|
|
Regional Regulatory and Safety Standards Evolution |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Non-Pressurized Containers expected to hold largest market share of 52.2% in 2026. The non-pressurized container segment in the Nasal Drug Delivery Systems Market is gaining traction as patients and healthcare providers increasingly value its simple, easy-to-use, and patient-friendly design for home and self-administration. Its propellant-free mechanism attracts environmentally conscious users and minimizes safety risks. Manufacturers actively choose it for cost-efficient production and its ability to accommodate various formulations, including liquids, gels, and powders. At the same time, rising demand for over-the-counter treatments and flexible dosing options drives broader adoption through hospitals, pharmacies, and retail outlets.
Nasal Spray hold the largest market share of 45.7% in 2026. The nasal spray segment in the Nasal Drug Delivery Systems Market is expanding as patients and healthcare providers increasingly prefer its convenient, non-invasive administration and fast therapeutic effects. Rising cases of allergic rhinitis, sinusitis, and other respiratory conditions boost demand, while patients actively choose self-administration over injections. Continuous improvements in device design, formulation stability, and targeted delivery enhance effectiveness and comfort, promoting broader use. At the same time, growing over-the-counter availability and expanding applications in central nervous system and hormonal therapies further accelerate market adoption. For instance, Haleon and Aptar Pharma launched Otrivin Nasal Mist, a next-generation nasal spray designed to improve comfort for people with nasal congestion caused by colds, allergic rhinitis, and sinusitis. The spray uses microdroplet technology to deliver a gentle, evenly distributed mist without dripping into the back of the nose and throat.
Multi Dose acquired the prominent market share of 41.1% in 2026. The multi-dose segment in the Nasal Drug Delivery Systems Market is expanding as patients and healthcare providers actively choose convenient, reusable devices for chronic and frequent treatments. By providing multiple doses in a single container, these systems minimize the need for frequent refills and streamline treatment routines. Manufacturers take advantage of efficient production and lower costs, while patients benefit from consistent dosing and ease of use. Growing applications in allergy management, pain relief, and vaccines continue to drive adoption across hospitals, pharmacies, and retail outlets. For instance, in June 2025, Zeteo Biomedical secured new patents in the United States and European Union for its ZTech drug delivery technologies. The patents cover cartridge-based and hand-operated medicament administration devices, strengthening the ZTech platform. These approvals advance the development of ZTech-L and ZTech-P, which include single-use disposable and multi-dose, cartridge-reloadable handheld devices.
Vaccination captures the largest market share of 46.6% in 2026. The vaccination segment in the Nasal Drug Delivery Systems Market is expanding as patients and healthcare providers actively choose needle-free, non-invasive immunization methods. Intranasal vaccines trigger both mucosal and systemic immunity, providing effective protection against respiratory infections. Their simple administration, suitability for mass immunization, and reduced reliance on trained personnel encourage widespread use in hospitals, clinics, and community programs. Continuous innovations in formulations, delivery devices, and stability increase their applications, while raising awareness of respiratory diseases drives broader adoption.
Hospital Pharmacies expected to hold largest market share of 39.9% in 2026. The hospital pharmacy segment in the Nasal Drug Delivery Systems Market is growing as hospitals actively distribute prescription and specialized nasal therapies. Healthcare providers depend on hospital pharmacies to deliver controlled dosing, ensure safe handling, and provide immediate access to critical treatments in emergency and inpatient care. By working closely with medical staff, hospital pharmacies ensure accurate administration and patient monitoring. They also facilitate clinical trials, support innovative formulations, and manage advanced devices, positioning themselves as a vital channel for introducing new nasal drug delivery products.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 45.80% in 2026. The North America Nasal Drug Delivery Systems Market is expanding as patients increasingly prefer non-invasive therapies and healthcare providers leverage advanced medical infrastructure. Rising cases of respiratory and chronic conditions boost demand for nasal sprays, vaccines, and multi-dose systems. Pharmaceutical companies actively develop innovative formulations, smart devices, and intranasal biologics, while hospitals, clinics, and retail pharmacies distribute these products efficiently. Growing awareness of needle-free options, combined with regulatory support and technological innovations, drives wider adoption in both clinical and self-care environments. For instance, in February 2024, Lupin Limited announced the launch of Cyanocobalamin Nasal Spray, 500 mcg per spray, following approval from the United States Food and Drug Administration (U.S. FDA).
The Asia Pacific Nasal Drug Delivery Systems Market is growing as patients and healthcare providers increasingly adopt non-invasive therapies. Rising cases of respiratory disorders, allergies, and chronic illnesses boost demand for nasal sprays, vaccines, and multi-dose systems. Pharmaceutical companies develop cost-effective, user-friendly devices, while hospitals, clinics, and pharmacies actively distribute them. Technological innovations, supportive government initiatives, and expanding self-care practices further accelerate adoption across the region. For instance, in October 2025, Viatris Inc. acquired Aculys Pharma, Inc., gaining rights to develop and commercialize pitolisant and Spydia® in the central nervous system therapy area. The deal expands Viatris’ innovative portfolio in Japan and includes exclusive rights in Japan and select Asia-Pacific markets for Spydia Nasal Spray, approved in Japan in June 2025 to treat status epilepticus.
The United States Nasal Drug Delivery Systems Market is expanding as patients and healthcare providers actively choose fast-acting, non-invasive treatments. Rising cases of allergies, respiratory disorders, and chronic conditions increase demand for nasal sprays, vaccines, and multi-dose systems. Pharmaceutical companies develop advanced formulations, smart devices, and intranasal biologics, while hospitals, clinics, and retail pharmacies actively distribute these products. Growing awareness of needle-free options, along with regulatory support and technological innovations, drives wider adoption in both clinical care and self-administered therapies. For instance, in December 2025, Aptar Group, Inc. announced that its Bidose (BDS) Liquid Nasal Spray System serves as the delivery mechanism for the newly approved CARDAMYST™ (etripamil) Nasal Spray.
The India Nasal Drug Delivery Systems Market is growing as patients and healthcare providers actively adopt non-invasive, easy-to-use therapies. Increasing cases of respiratory disorders, allergies, and chronic conditions boost demand for nasal sprays, vaccines, and multi-dose systems. Pharmaceutical companies develop affordable formulations and user-friendly devices to serve diverse populations, while hospitals, clinics, and retail pharmacies actively distribute these products. Government initiatives, rising awareness of needle-free options, and technological innovations continue to accelerate the adoption of nasal drug delivery solutions across the country.
Patients and healthcare providers increasingly prefer needle-free, non-invasive administration due to ease, rapid onset, and improved compliance. Nasal delivery offers direct access to systemic circulation or the central nervous system without injections. This trend is expanding use across allergy treatments, pain management, vaccines, and neurological therapies, while encouraging pharmaceutical companies to develop innovative nasal formulations and devices that prioritize comfort and efficiency in self-administration and clinical settings.
The market is shifting towards technologically advanced nasal devices equipped with features like dose metering, digital tracking, and ergonomic designs. Smart systems enhance accuracy, patient adherence, and convenience, particularly for chronic or multi-dose therapies. Integration with mobile applications and healthcare monitoring platforms allows providers to track compliance, improving treatment outcomes. These innovations also support wider adoption in hospitals, clinics, and home healthcare, meeting growing demand for precision and usability in nasal drug delivery.
There is a significant opportunity to develop ergonomic, portable, and easy-to-use nasal devices that cater to pediatric, adult, and elderly populations. End-users increasingly value comfort, convenience, and accurate dosing. Manufacturers can introduce smart devices with digital tracking and dose control, improving adherence and treatment outcomes. Retail and hospital pharmacies can further drive market penetration by offering user-friendly solutions for at-home therapy, chronic care, and emergency use, addressing diverse patient needs.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 98.3 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.2% | 2033 Value Projection: | USD 134.67 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Merck & Co., Inc., GlaxoSmithKline plc, AstraZeneca plc, Johnson & Johnson, Pfizer Inc., Baxter International, Inc., Valeant Pharmaceuticals International, Inc., Becton, Dickinson and Company, and Novartis AG. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients