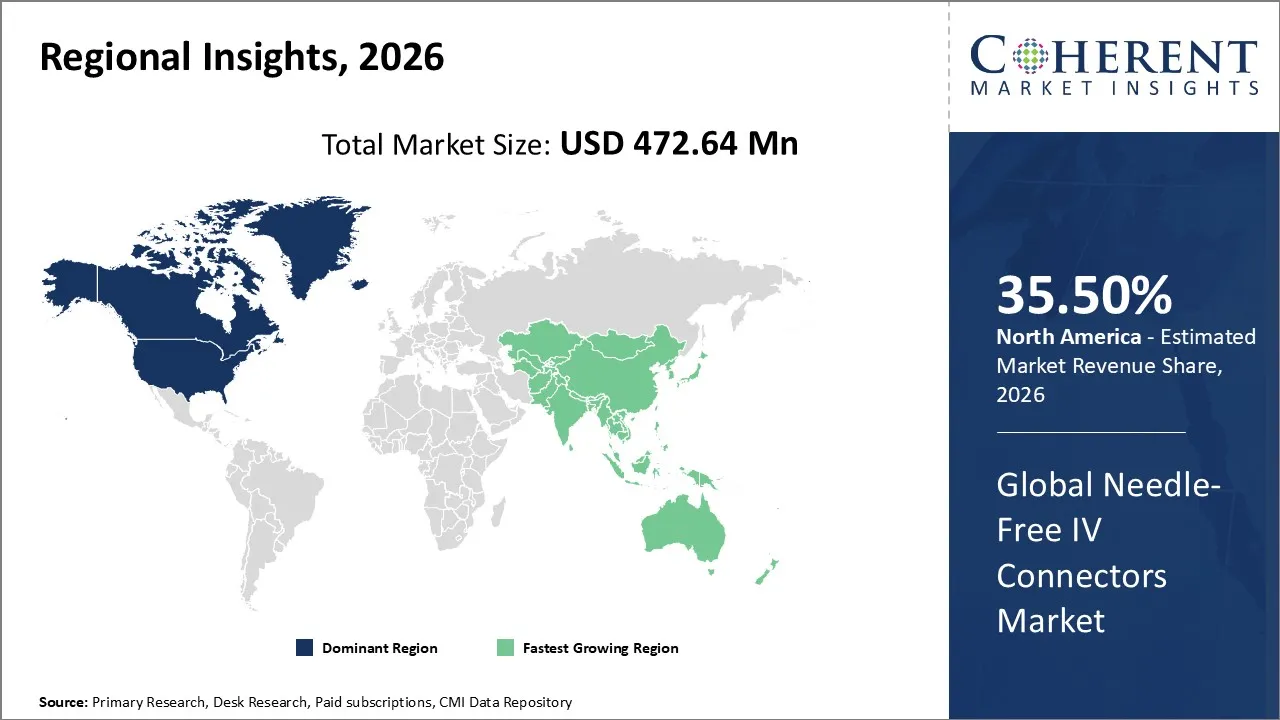
The Needle-Free IV Connectors Market is estimated to be valued at USD 472.64 Mn in 2026 and is expected to reach USD 798.65 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 6.2% from 2026 to 2033.
The Needle-Free IV Connectors Market grows as healthcare providers prioritize patient and worker safety, strengthen infection prevention measures, and enhance intravenous therapy practices. These connectors actively reduce needlestick injuries and catheter-related bloodstream infections while enabling efficient medication and fluid administration. Increasing hospital admissions, the rising burden of chronic diseases requiring IV therapy, and expanding adoption across hospitals, ambulatory centers, and home healthcare settings continue to drive market growth globally.
|
Current Events |
Description and its impact |
|
Technological Advancements in Safety-Engineered Medical Devices |
|
|
Regulatory and Policy Changes in Healthcare Equipment |
|
|
Geopolitical and Regional Healthcare Infrastructure Development |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Straight Channel hold the largest market share of 42.2% in 2026. The straight channel segment grows as healthcare providers choose its simple structure and direct fluid pathway for everyday IV therapy. This design enables smooth fluid movement, limits dead space, and makes flushing easier during clinical use. Providers also value straight channel connectors for their affordability and compatibility with standard IV systems, which allows efficient use in high-volume hospital environments and sustains demand across acute and general care settings.
Neutral expected to hold largest market share of 46.4% in 2026. The neutral displacement segment grows as healthcare providers rely on its balanced fluid control to prevent blood reflux during connection and disconnection. Hospitals and clinics adopt neutral connectors to enhance IV safety, simplify maintenance, and ensure consistent flushing practices. Their versatility for long-term and routine infusions makes them a preferred option across hospitals, outpatient centers, and home care environments, driving widespread and continuous adoption. For instance, ICU Medical, Inc. secured two six-year group purchasing contracts with Premier Healthcare Alliance, covering its full range of needlefree IV connectors, sets, and accessories. For the first time, the company's entire line—including Neutron®, MicroClave®, MicroClave Clear, and NanoClave® connectors—is included in a single Premier contract.
Seven-day acquired the prominent market share of 36.6% in 2026. The seven-day segment expands as healthcare providers use connectors that stay safely in place for extended periods, cutting down on frequent replacements. Providers rely on these connectors to streamline workflow, reduce patient discomfort, and maintain reliable IV access over several days. Their robust design and infection-control features make them ideal for both acute and long-term care, driving adoption across hospitals, outpatient centers, and home healthcare settings.
Hospitals hold the largest market share of 43.3% in 2026. Hospitals propel the Needle-Free IV Connectors Market by performing large volumes of intravenous procedures while prioritizing patient safety. Healthcare teams implement these connectors to prevent needlestick injuries, reduce catheter-related infections, and streamline IV therapy across departments. Trained staff, standardized protocols, and established infrastructure enable effective use of advanced needle-free systems. Hospitals also rely on these connectors to enhance workflow efficiency, maintain regulatory compliance, and deliver consistent care in both acute and long-term patient settings.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 35.5% in 2026. In North America, healthcare providers drive the Needle‑Free IV Connectors Market by adopting needle‑free systems across hospitals, clinics, and home infusion services to strengthen infection control and advance clinical practices. Strong healthcare infrastructure and strict safety standards promote the use of closed IV access technologies. Key manufacturers continuously innovate, and providers focus on minimizing catheter-related infections and needlestick injuries, influencing product choices and sustaining demand across diverse care settings.
In the Asia Pacific region, healthcare providers and hospitals drive the Needle‑Free IV Connectors Market by expanding infrastructure and increasing capacity. Hospitals in countries like China, India, and Japan invest in modern medical facilities and strengthen infection control practices, encouraging broader adoption of needle‑free IV systems. Growing awareness of patient safety, government healthcare reforms, and rising middle-class populations further promote the use of advanced connectors. Local manufacturers and global partnerships actively support the region’s market growth and product availability.
In the United States, hospitals and healthcare providers drive the Needle‑Free IV Connectors Market by prioritizing infection prevention and patient safety, standardizing closed IV access systems in line with CDC and FDA guidelines. Providers actively select advanced connector designs to minimize contamination risks and enhance efficiency in inpatient, outpatient, and home infusion care. Major manufacturers, strong regulatory support, and significant healthcare investment further promote adoption and encourage ongoing innovation in connector technologies.
In India, hospitals and healthcare providers drive the Needle‑Free IV Connectors Market by adopting needle‑free systems as they modernize facilities and strengthen infection control practices. Rising chronic disease cases and increased focus on patient safety encourage this shift. Expanding healthcare infrastructure and government support for advanced medical technologies further boost demand in public and private facilities. Domestic manufacturers, broader distribution networks, and partnerships with global companies actively improve product availability and accessibility across urban areas as well as tier‑2 and tier‑3 markets.
Healthcare providers increasingly adopt needle-free connectors to minimize needlestick injuries and catheter-related bloodstream infections. Hospitals and clinics focus on integrating closed IV access systems into standard protocols, emphasizing aseptic practices. The trend reflects heightened regulatory attention, growing patient safety awareness, and efforts to reduce healthcare-associated infections, driving continuous demand for advanced, safe, and reliable needle-free connector solutions across acute, outpatient, and home care settings.
Manufacturers are introducing connectors with enhanced anti-reflux, antimicrobial, and low dead-space designs. Innovations aim to improve fluid flow, reduce infection risk, and simplify flushing and maintenance. User-friendly designs and ergonomic features help healthcare providers handle connectors efficiently. This trend reflects ongoing R&D investment, clinical feedback integration, and the demand for advanced connectors that improve therapy efficiency while ensuring safety in diverse clinical environments.
There is an opportunity to develop connectors with improved anti-reflux, antimicrobial, and low dead-space features. Incorporating ergonomic and user-friendly designs enhances workflow efficiency for healthcare staff. Continuous R&D and collaboration with clinicians can yield products that address specific clinical challenges, such as reducing blood reflux or contamination risk, positioning companies as leaders in safety-focused, high-performance needle-free IV connector solutions across acute, outpatient, and long-term care settings.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 472.64 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.2% | 2033 Value Projection: | USD 798.65 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Baxter International Inc., B. Braun Melsungen AG, Becton, Dickinson and Company, CareFusion Corporation, ICU Medical, Inc., Nexus Medical LLC, RyMed Technologies, LLC, and Vygon S.A. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In December 2025, HAI Solutions, a leader in UVC-based microbial reduction medical devices, announced that the U.S. Food and Drug Administration (FDA) granted De Novo classification to its QIKCAP System. This decision establishes the QIKCAP System as the first and only FDA-cleared ultraviolet light–based microbial reduction device for luer-activated valves, creating a new Class II medical device category.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients