Nephroblastoma treatment market is estimated to be valued at USD 1.97 Bn in 2026 and is expected to reach USD 2.81 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 5.2% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
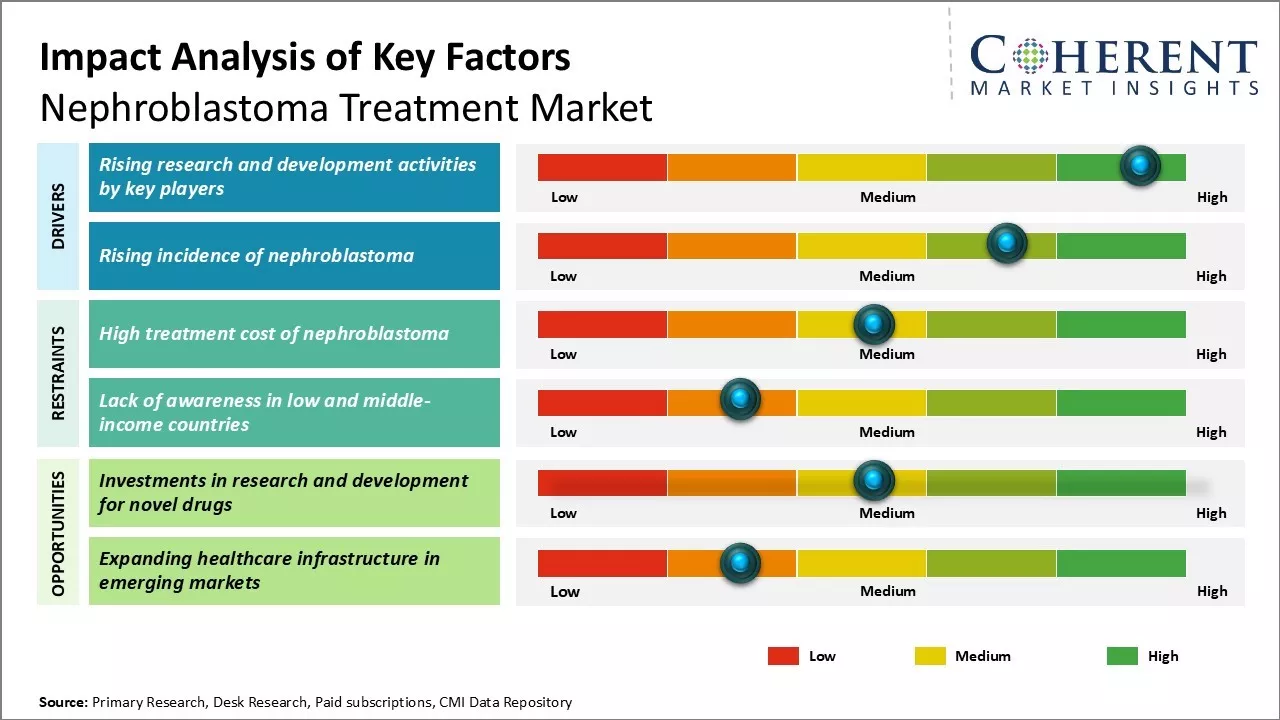
The nephroblastoma treatment market is poised for positive growth due to the rising incidence of nephroblastoma worldwide, which increases the demand for effective therapies. Nephroblastoma treatment is primarily treated through a combination of surgery, chemotherapy, and radiation therapy. The development of various pipeline drugs and therapies currently in clinical trials presents significant opportunities for market expansion upon approval. However, high cost of treatment and limited awareness about nephroblastoma in developing regions may hinder the market growth.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
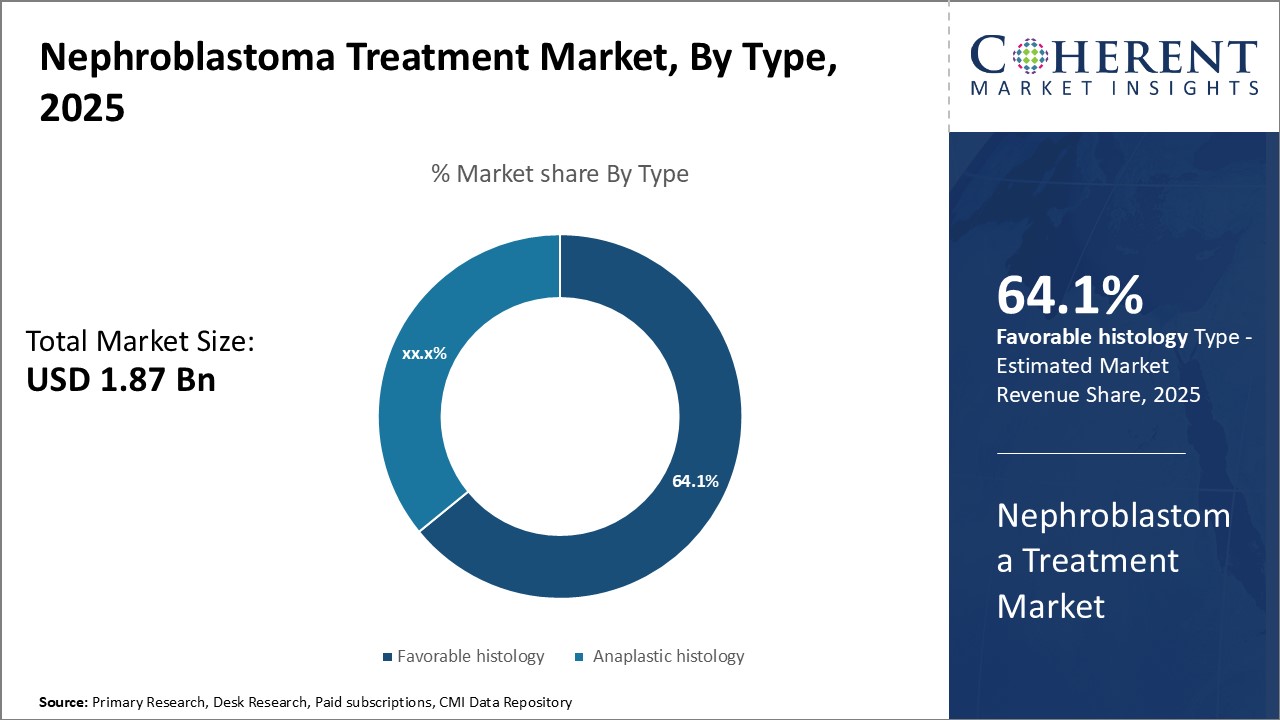
Insights By Type- Favorable histology dominates due to superior treatment outcomes
In terms of type, favorable histology segment is estimated to contribute the highest share of 65.1% in 2026 in nephroblastoma treatment market, due to its superior treatment outcomes compared to anaplastic histology. Nephroblastoma with favorable histology responds well to chemotherapy, boasting a 90% five-year survival rate, while anaplastic histology is linked to aggressive disease and poor chemotherapy response.
To learn more about this report, Request Free Sample
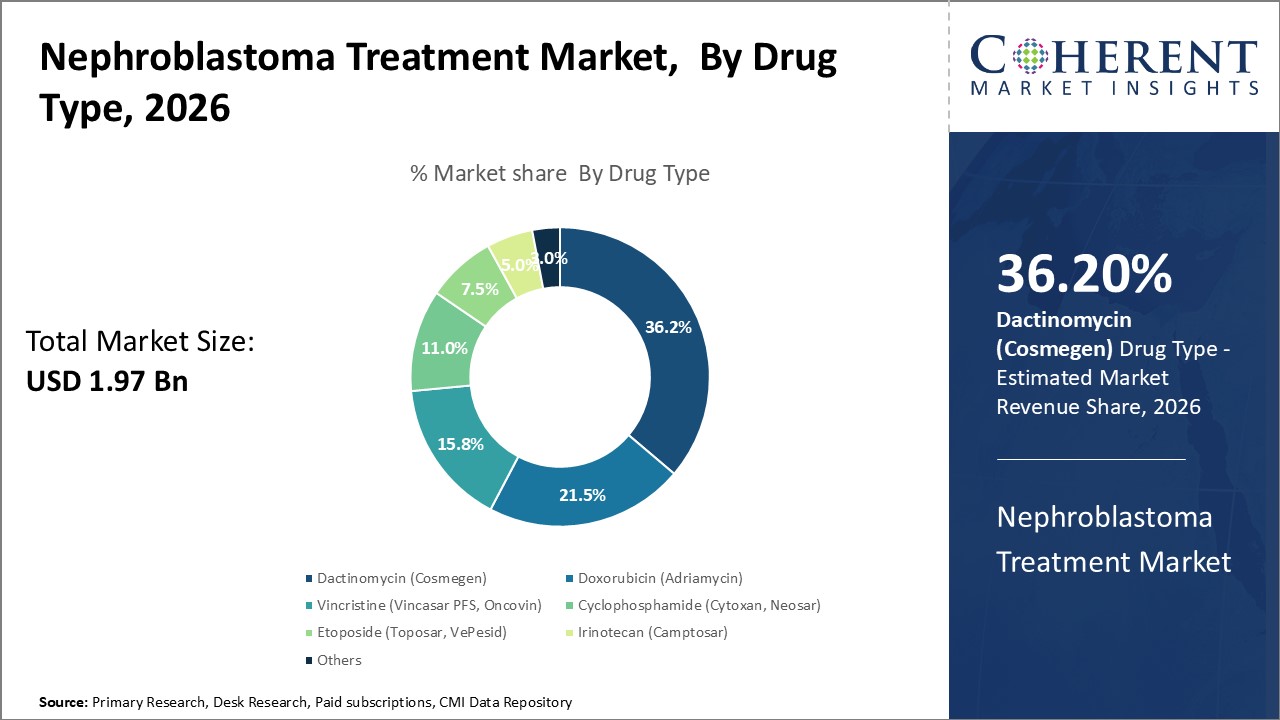
Insights, By Drug Type- Dactinomycin dominates as the standard first line therapy
In terms of drug type, dactinomycin (cosmegen) segment is estimated to contribute the highest share of 36.2% in 2026 in nephroblastoma treatment industry, as it is the standard first-line therapy for nephroblastoma. It intercalates DNA and inhibits RNA and protein synthesis, yielding excellent response rates. Its favorable toxicity profile makes it a preferred treatment option, often combined with Doxorubicin and Vincristine in multi-agent regimens.
Insights, By Distribution Channel- Hospital pharmacies lead due to complex treatment needs
In terms of distribution channel, hospital pharmacies segment is estimated to contribute the highest market share of 52.9% in 2026, due to complex treatment needs of patients. Nephroblastoma treatment often involve multi-drug chemotherapy administered via intravenous infusion pumps, necessitating inpatient care where specialized supervision and critical infrastructure are available. Hospital pharmacies are equipped to handle the stringent requirements for oncology drugs.
Need a Different Region or Segment? Request Free Sample
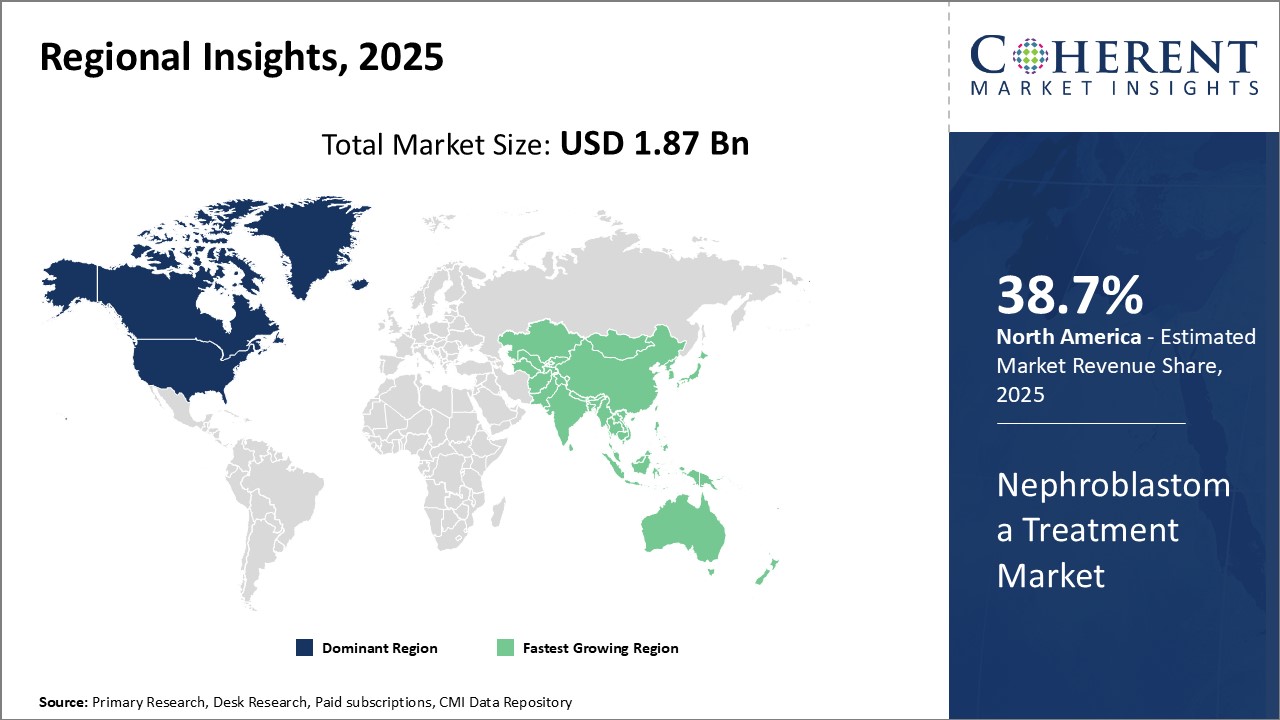
Dominating Region- North America
North America is expected to dominate the market with an estimated market share of 38.7% in 2026, due to the strong presence of leading pharmaceutical companies and advanced healthcare infrastructure. Major regional players are continuously investing in research and development to create novel targeted therapies.
Fastest-Growing Region- Asia Pacific
Asia Pacific region exhibits the fastest growth, with an estimated market share of 21.8% in 2026, owing to rising healthcare expenditure, large patient population, and increasing awareness about early disease diagnosis and treatment. Governments are also implementing new healthcare policies to enhance access and affordability of cancer care.
Nephroblastoma Treatment Market Outlook for Key Countries
U.S.’ increasing number of clinical trials
The U.S. nephroblastoma treatment market is experiencing significant growth, driven by an increasing number of clinical trials for investigational drugs and the rising adoption of combination therapies. Major players like Merck & Co. and Pfizer Inc. are pivotal, holding substantial market shares and demonstrating a strong commitment to enhancing treatment options and improving outcomes for pediatric patients suffering from this condition.
China's robust economic development
China's rapid economic development has led to a significant increase in healthcare spending, resulting in marked improvements in medical facilities and services. This financial boost enables investments in advanced treatment options for nephroblastoma and other conditions, thereby enhancing the overall quality of care. Government initiatives are also expanding insurance coverage, making these treatments more accessible to a wider population.
India’s leadership in generic drug manufacturing and exports
India continues to be a leader in generic drug manufacturing and exports. However, high treatment costs for nephroblastoma treatment present significant challenges for patient. In response, government is implementing various measures to enhance cancer screening and treatment facilities nationwide, aiming to improve access and affordability for patients requiring specialized care. These measures aim to improve access and affordability for patients requiring specialized care, ensuring that effective treatments are within reach for a broader population.
Japan’s investment in research and development
Japan nephroblastoma treatment industry is dominated by leading global drug makers focusing on advanced disease stages through innovative therapeutic approaches. These companies are investing in research and development to create novel treatments that target specific tumor characteristics, aiming to improve patient outcomes. This strategic focus aligns with Japan's commitment to enhancing pediatric cancer care and ensuring access to cutting-edge therapies.

Get actionable strategies to beat competition: Request Free Sample
Top Strategies Followed by Nephroblastoma Treatment Market Players
Emerging Startups in the Nephroblastoma Treatment Market
Key Takeaways from Analyst
Nephroblastoma Treatment Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1.97 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.2% | 2033 Value Projection: | USD 2.81 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Merck & Co., Inc., Pfizer Inc., Sun Pharmaceutical Industries Ltd., Actiza Pharmaceutical Private Limited, Teva Pharmaceutical Industries Ltd., Alvogen, Accord Healthcare Ireland Ltd., Amneal Pharmaceuticals LLC., Cadila Pharmaceuticals Ltd., and Xediton Pharmaceuticals |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Driver- Rising research and development activities by key players
Nephroblastoma treatment market growth is driven by increasing research focus of pharmaceutical players. Major companies in the oncology segment are aggressively investing in developing novel drug candidates and treatment methods for nephroblastoma. For instance, the Children's Oncology Group, a clinical trials group supported by the National Cancer Institute, is currently studying the effect of combination chemotherapy and surgery in treating young patients with Wilms tumor.
Market Challenge- High treatment cost of nephroblastoma
Global nephroblastoma treatment sector faces challenges like high cost of treatment. Nephroblastoma, a kidney cancer primarily affecting children, requires expensive procedures such as advanced surgeries, chemotherapy, and targeted therapies. This financial burden significantly impacts families, especially in developing countries where access to care is restricted. Even in developed markets, rising healthcare costs threaten overall market growth, necessitating collaborative efforts among governments, healthcare providers, insurance companies, and pharmaceutical firms to improve access to essential treatments.
Market Opportunity- Investments in research and development for novel drugs
Nephroblastoma treatment industry can witness growth opportunities due to increased investments in research and development for novel drugs. Enhanced R&D can expedite drug development, addressing unmet needs such as disease relapse and resistance to existing treatments. This focus allows pharmaceutical companies to create precise, personalized medicines with improved safety profiles, opening new revenue streams and market potential. Collaborative funding from governments and international organizations further encourages innovation in pediatric oncology, creating growth avenue for market players.
Primary Research Interviews:
Databases:
Magazines:
Journals:
Newspapers:
Associations:
Public Domain Sources:
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients