Plasma Therapy Market is estimated to be valued at USD 499.0 Mn in 2026 and is expected to reach USD 1,279.7 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 14.4% from 2026 to 2033.
Analysts’ Views on Global Plasma Therapy Market :
Increasing number of partnerships, collaborations, and partnerships agreements are expected to boost the growth of the global plasma therapy market over the forecast period. For instance,on July 3, 2023, Personalis Inc., an advanced genomics provider for precision oncology, announced that it will continue its collaboration with AstraZeneca, a biopharmaceutical company, to explore ultra-sensitive molecular residual disease (MRD) measurement, including clinically relevant and personalized variant tracking, for clinical research and drug development. The trial will investigate the role of circulating tumor DNA (ctDNA) and Human Papillomavirus (HPV) detection in plasma in locally advanced cervical cancer.
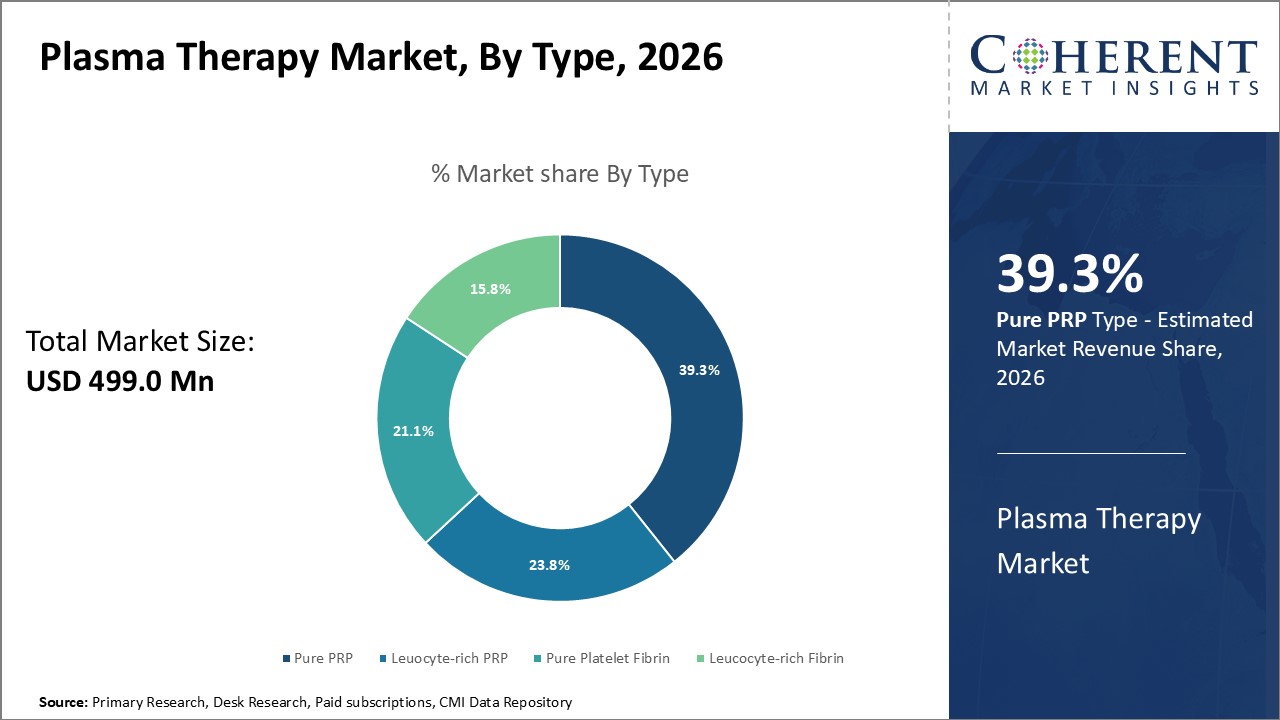
Figure 1. Global Plasma Therapy Market Share (%), by Type, 2026
To learn more about this report, Request Free Sample
Global Plasma Therapy Market– Driver
Increasing collaborations among pharmaceutical and biopharmaceutical companies
Collaborations and partnerships between pharmaceutical companies are a major factor leading to the high demand for plasma therapy. For instance, in Novemebr 2022, CSL, an Australia-based multinational specialty biotechnology company, announced a license agreement with Translational Sciences, Inc., a targeted drug therapy provider, to license TS23, an anti-α2-antiplasmin monoclonal antibody. TS23 is being developed to dissolve thrombi that cause serious conditions such as pulmonary embolism (PE) and acute ischemic stroke (AIS). The treatment candidate is soon to be evaluated in the U.S. in the NAIL-IT Phase II study, which has been designed to evaluate the safety and thrombolytic effect of ascending doses of TS23 in patients with sub-massive (intermediate risk) PE.
Establishment of newer technologies regarding plasma therapy
The development of new tewchnologies regarding plasma tharapy can drive the growth of the global plasma therapy market. For instance, in March 2021, Grifols, S.A, a developer of plasma-derived therapies and diagnostic solutions, announced its first-ever installation in North America of the DG Reader Net semi-automated analyser, used to facilitate pre-transfusion blood type compatibility testing.
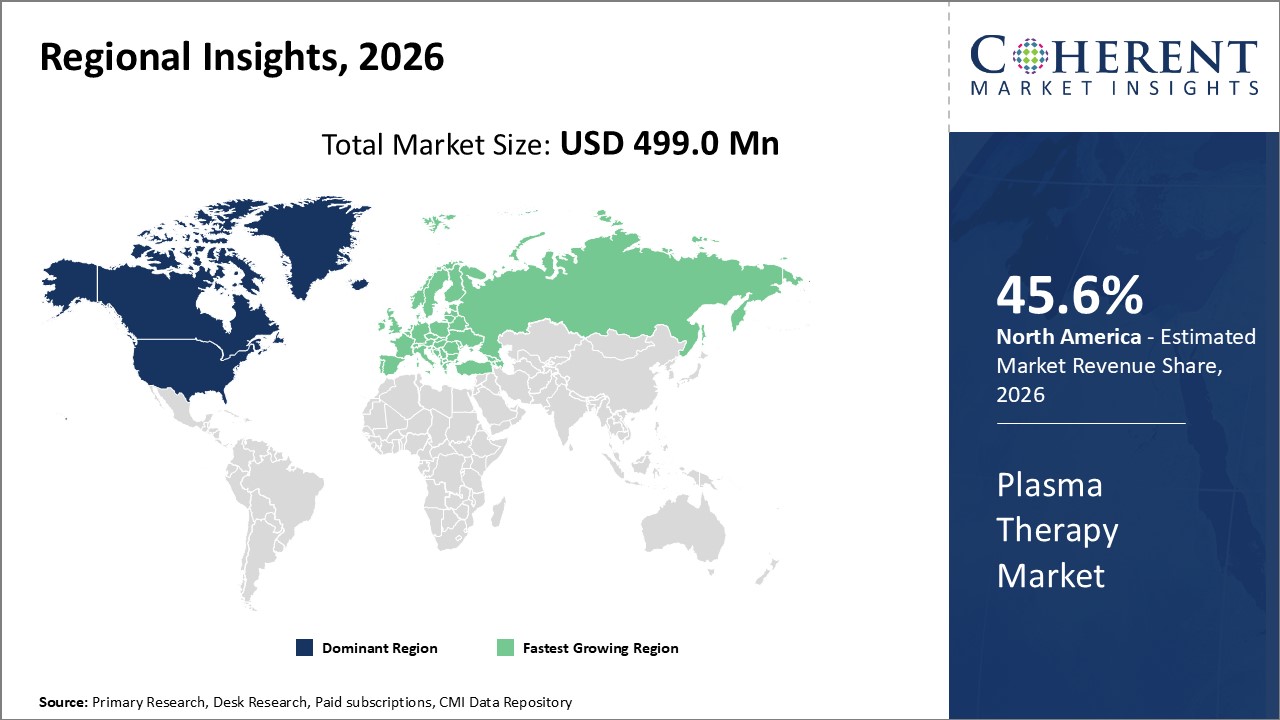
Figure 2. Global Plasma Therapy Market Value (US$ Million), by Region, 2026
To learn more about this report, Request Free Sample
Global Plasma Therapy Market- Regional Analysis
Among regions, North America is estimated to hold a dominant position in the global plasma therapy market over the forecast period. This is due to the increasing number of produt approvals in the region. For instance, on April 25, 2026, Biogen, a U.S.-based multinational biotechnology company, announced that the U.S. Food and Drug Administration (FDA) has approved QALSODY (tofersen) 100 mg/15mL injection for the treatment of amyotrophic lateral sclerosis (ALS) in adults who have a mutation in the superoxide dismutase 1 (SOD1) gene. This indication is approved under accelerated approval based on reduction in plasma neurofilament light chain (NfL) observed in patients treated with QALSODY.
Plasma is used to treat medical emergencies such as trauma, shock, and burns. The proteins in a human’s plasma can also be extracted to create medications for rare chronic conditions such as autoimmune disorders and hemophilia. In most cases, plasma proteins cannot be manufactured synthetically, so proteins from a person’s donated plasma are the only source for medical therapies for Mns of people each year. In order to simplify the plasma donation process, many companies created softwares for the plasma donation process. The software matches patients in clinical trials for convalescent plasma therapy with recovered patients who are willing to donate antibodies.
Plasma Therapy Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 499.0 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 14.4% | 2033 Value Projection: | USD 1,279.7 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Octa pharma AG, Top Bio Group Co., Ltd, Bio Products Laboratory Ltd, Kedrion S.p.A, Grifols, S.A, CSL, Biotest AG, Takeda Pharmaceutical Company Limited, DePuy Synthes (A subsidiary of Johnson & Johnson Services, Inc.), Regen Lab SA, LFB S.A, ImmunoTek Plasma. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Plasma Therapy Market Segmentation:
The global plasma therapy market report is segmented into product type, interface, end user, and region.
By Type, the market is segmented into Pure PRP, leucocyte-rich PRP, Pure Platelet-rich Fibrin (PRF), and Leukocyte-rich Fibrin (L-PRF). Out of which, the Pure PRP segment is expected to hold a dominant position in the global plasma therapy market during the forecast period and this is attributed to its greater effectiveness.
By Application, the market is segmented into infections diseases, dental, nerve injury, cardiac muscle injury, and other applications. Out of which, the infectious diseases segment is expected to hold a dominant position in the global plasma therapy market during the forecast period and this is attributed to the increasing global burden of infectious diseases.
By End user, the market is segmented hospitals & clinics, research institutes, and others. Out of which, the hospitals & clinics segment is expected to dominate the market over the forecast period and this is attributed to the availability of blood banks in hospitals and clinics.
Among all the segmentations, the product type segment has the highest potential due to the increasing product approvals across the world over the forecast period. For instance, in February 2020, EmCyte Corporation, a company specialized in platelet rich plasma and progenitor stem cell biologics, announced its FDA 510(k) clearance for its PurePRP Supraphysiologic Concentrating System for platelet rich plasma. The system is cleared to be used for the safe and rapid preparation of autologous platelet rich plasma (PRP) from a small sample of blood at the patient's point of care. This clearance completes EmCyte Corporation’s PURE Supraphysiologic product portfolio allowing practitioners to use the most advanced in autologous biologic concentrating systems.
Global Plasma Therapy Market Cross Sectional Analysis:
Increase in research & development activities of invasive ventilators in North America region is expected to drive growth of Pure PRP segment in this region. For instance, in August 2022, researchers working at Regeneris Medical Boston, a healthcare and clinical research organization specializing in regenerative medicine and cellular therapies, introduced a novel way to stimulate hair growth in men and women using growth factors from High Density Platelet Rich Plasma (HDPRP) introduced to the scalp via a novel 1,927 nm fractional thulium laser system bypassing the traditional route of scalp injections.
Global Plasma Therapy Market: Key Developments
On August 24, 2020, the Food and Drug Administration (U.S.
On 3 June 2020, researchers from the Houston Methodist HospitalAccording to the researchers, 19 out of 25 patients were showing signs of recovery post plasma therapy and 11 patients were later discharged from the hospital following recovery.
The treatment plan involves using blood plasma of patients who have recovered from the infection by further isolating the antibodies from the plasma and injecting it in patients. This therapy triggers an immune response in the body of the patients which can help them survive the infection.
The project aims to provide free plasma therapy at 21 government centers in Maharashtra to increase the chances of survival of the infected patients. The government is also spreading awareness among recovered patients regarding plasma donation so that the plasma can be processed and utilized for therapy.
Global Plasma Therapy Market: Key Trends
Kamada Pharmaceuticals would be responsible for product development, manufacturing and clinical development with the help of Kedrion S.p.A. Post regulatory approval, Kedrion S.p.A will be responsible for commercialization of the product in the U.S, Europe, Australia, and South Korea, while, distribution of the product to the rest of the world will be undertaken by Kamada Pharmaceuticals.
Collaborations and partnership agreements between biopharmaceutical companies can propel the growth of the global plasma therapy market. For instance, in April 2021, GEA Group, a Germany-based supplier of systems and components to the food, beverage, and pharmaceutical industries, signed a partnership agreement with Biopharma- a Ukraine-based pharmaceutical company focused on the development and production of drugs based on human plasma, to deliver separators for blood plasma fractionation in Ukraine.
Global Plasma Therapy Market: Restraint
Potential side-effects of Platelet-Rich Plasma Therapy
The side-effects of Platelet-Rich Plasma Therapy can restrain the growth of the global plasma therapy market. Some people who undergone PRP therapy complain about an acute ache or soreness in the spot of the injection. Sometimes this pain is even felt deep inside the area, whether in the muscle or bone.
In the advent of such side-effects, contacting a doctor or healthcare provider as soon as possible can serve as an effective counterbalance against this restraint.
Product Recalls
The World Health Organization is recommending the use of plasma therapy in conditions like Hemophilia B. But there are several barriers like faults in production and manufacturing that are restraining the growth of the market. For instance, in January 2021, CSL, a biopharmaceutical company, issued a voluntary recall covering one batch of Mononine (Its long-standing treatment for preventing and controlling bleedings in people with hemophilia B) due to a manufacturing problem that occurred during the filling process for that batch of Mononine. Mononine is a type of plasma replacement therapy that involves supplying plasma-derived factor IX, the blood coagulation protein missing or defective in hemophilia B, to patients.
This can be overcome by strict compliance with regulatory procedures as well as adoption of Good Manufacturing Practices (GMP).
Global Plasma Therapy Market- Key Players
Major players operating in the global plasma therapy market include Octapharma AG, Top Bio Group Co., Ltd, Bio Products Laboratory Ltd, Kedrion S.p.A, Grifols, S.A, CSL, Biotest AG, Takeda Pharmaceutical Company Limited, DePuy Synthes (A subsidiary of Johnson & Johnson Services, Inc.), Regen Lab SA, LFB S.A, and ImmunoTek Plasma.
*Definition: Convalescent is the term given to a person who is recovering, or convalescing from an illness, injury, infection, surgery, procedure, therapy, treatment, or any health situation. The recovery period is called convalescence. The plasma of such a person is rich in antibodies that were produced by the body, in order to fight the pathogen concerned (virus or bacteria). These antibodies can be harvested from a convalescent’s plasma and injected into the blood of another person suffering from the same infection. The latter’s body may not be able to produce enough antibodies to fight the infection, due to various reasons. So, by harvesting antibodies from another person who has successfully fought the infection and injecting it here, this person’s immunity is boosted. This approach and procedure are what’s called Convalescent Plasma Therapy (CPT) or plasma therapy.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients