Global pneumococcal vaccines market is estimated to be valued at USD 9.88 Bn in 2026 and is expected to reach USD 14.18 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 5.3% from 2026 to 2033.
To learn more about this report, Request Free Sample
The global Pneumococcal Vaccines Market is experiencing steady growth, fuelled by increasing awareness about pneumococcal infections and rising immunization efforts across age groups. Advancements in conjugate vaccine formulations and supportive government healthcare policies are key growth drivers. Adult vaccination is gaining prominence due to aging populations and preventive healthcare focus. Hospital pharmacies remain the preferred distribution channel.
|
Current Event |
Description and its impact |
|
Global Vaccine Awareness Campaigns |
|
|
Technological Advancements in Vaccine Development |
|
|
Evolving Regulatory Policies and Global Health Funding |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Artificial Intelligence (AI) is playing a transformative role in the pneumococcal vaccines market by streamlining vaccine development, optimizing distribution, and enhancing disease surveillance. AI-powered predictive modelling accelerates R&D by identifying potential vaccine candidates and forecasting serotype evolution, enabling pharmaceutical companies to develop broader and more effective conjugate vaccines.
Machine learning algorithms are used to analyse vast datasets from clinical trials, improving vaccine safety and efficacy assessments. In distribution, AI enhances cold-chain logistics by predicting demand patterns and optimizing supply routes, reducing vaccine wastage and ensuring timely delivery.
Public health organizations also employ AI to monitor pneumococcal disease outbreaks, identify high-risk populations, and design targeted immunization campaigns. These innovations improve decision-making, lower costs, and expand access, particularly in resource-limited settings. As AI continues to advance, it is expected to play a vital role in driving efficiency, precision, and equity across the global pneumococcal vaccines landscape.
The pricing structure of pneumococcal vaccines varies significantly based on factors such as vaccine type, geographic region, procurement model, and manufacturer. Conjugate vaccines (PCVs), which offer broader protection, are generally priced higher than polysaccharide vaccines (PPVs) due to advanced formulation, longer efficacy, and higher production costs.
In high-income countries, PCVs can cost between $100 to $150 per dose in the private market, whereas in low- and middle-income countries, organizations like Gavi subsidize prices, bringing them down to as low as $3.50 per dose through negotiated agreements. Public procurement programs and tiered pricing models help governments maintain affordability and accessibility.
Hospital pharmacies typically offer more competitively priced vaccines under government immunization schemes, while retail and private providers may charge premiums. As competition increases and biosimilar PCVs enter the market, pricing pressure is expected to grow, potentially improving global access and affordability, especially in developing regions.
End-users across sectors express positive feedback regarding the role and performance of pneumococcal vaccines in public health outcomes. Biotechnology and pharmaceutical companies emphasize the demand for broader-spectrum conjugate vaccines and highlight the importance of AI and genomic tools in streamlining R&D and regulatory approval processes.
IVF clinics and biobanks underscore the need for safe and effective immunization, particularly for immunocompromised or neonatal populations, stressing cold-chain reliability and vaccine traceability. Research and academic institutions value the increasing availability of real-world data and collaborative research opportunities, which have accelerated innovations in serotype coverage and immunogenicity profiling.
Pharmaceutical companies also note growing interest in adult immunization, driving market expansion. However, cost remains a challenge in low-resource settings, with calls for more equitable pricing models. Overall, end-users view the pneumococcal vaccines market as essential, dynamic, and increasingly data-driven, with significant potential to address unmet needs through targeted innovation and public-private collaboration.
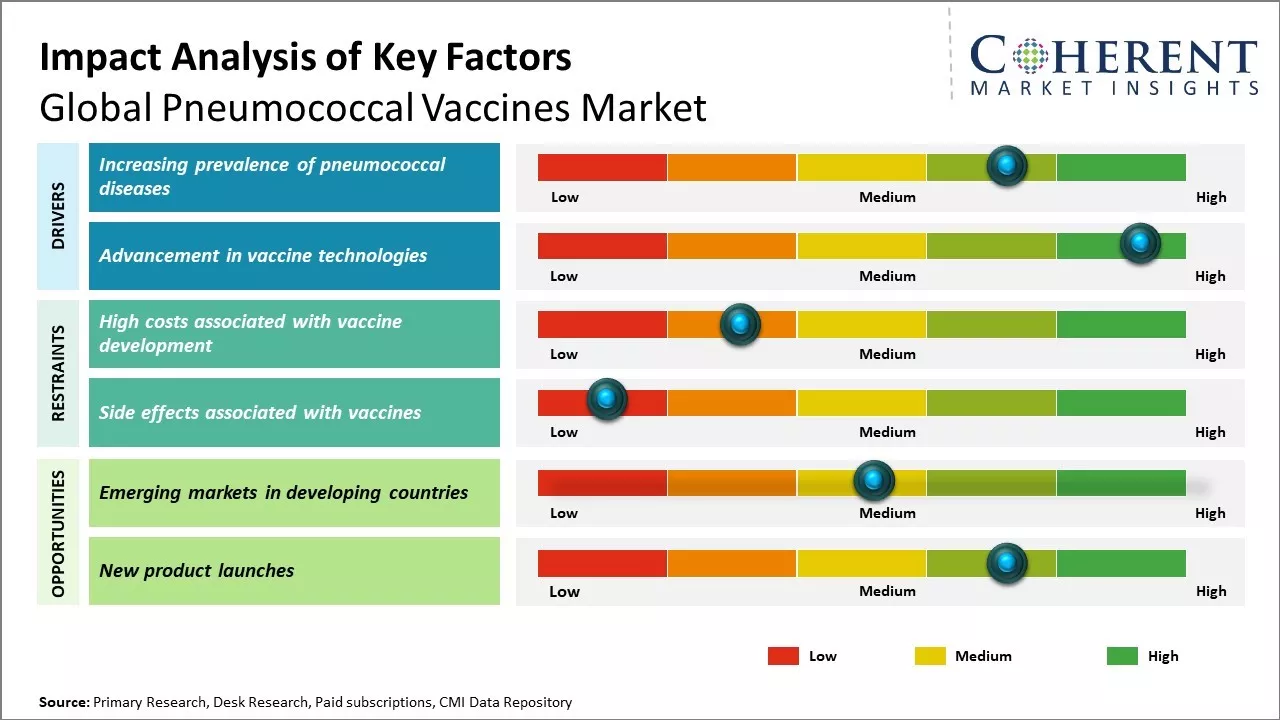
Rising burden of pneumococcal diseases can drive the market growth. Streptococcus pneumoniae, also known as pneumococcus, is a major cause of morbidity and mortality worldwide. It is a leading cause of community-acquired pneumonia as well as other infections such as bacteraemia, sepsis and meningitis, especially in children and the elderly population.
Pneumonia is a significant cause of death among children under 5 years of age. According to the data published by WHO, pneumonia caused over 800,000 deaths in children under age of 5 years in 2019. Although vaccines are available to prevent pneumococcal diseases, it is still endemic in many developing and underdeveloped regions with inadequate access to vaccination and healthcare services. Even in developed nations, growing elderly population who are more vulnerable is becoming a key concern area.
With aging population worldwide, the susceptibility to pneumococcal infections is projected to increase in the near future. This, coupled with emergence of antibiotic-resistant strains, has heightened the risks from these preventable diseases.
Rising awareness about pneumococcal diseases and their prevention through vaccination programs boosts the need for effective pneumococcal vaccines across all regions. Growing threat posed by these infections warrants strengthened immunization efforts and availability of advanced generation vaccines to counter the global burden of pneumococcal diseases.
In October 2024, Reuters reported that the U.S. Centers for Disease Control and Prevention (CDC) recommended expanding pneumococcal vaccination to adults aged 50–64, aiming to offer broader protection using vaccines like Pfizer’s Prevnar 20 and Merck’s Capvaxive.
Vaccine technology has witnessed tremendous progress, with new approaches enabling development of more targeted and effective vaccines. Major advancements like conjugate vaccines that stimulate improved immune response and provide longer lasting immunity have revolutionized immunization against pneumococcal diseases.
Advancements like cell-culture and protein-based vaccine production platforms offer scalability and efficiency benefits over traditional cultured methods. These innovative technologies facilitate development of next generation pneumococcal vaccine formulations with broader serotype coverage and better immunogenicity.
For example, second generation pneumococcal conjugate vaccines now provide protection against over 80% of disease-causing serotypes globally as compared to less than 30% coverage of earlier vaccines. Continued R&D can expand serotype coverage and improve other attributes such as thermostability and one-dose convenience. Novel platform technologies such as protein subunit vaccines also promise better safety by avoiding live pathogens. Driven by the need for universal vaccines, new mechanisms involving polypeptide constructs and glyco-engineering are being actively pursued.
With escalating antibiotic resistance challenge, vaccine innovation is increasingly being relied upon for long-term pneumococcal disease control. This augurs well for next generation pneumococcal vaccine products with capabilities beyond serotype replacement, offering even more comprehensive protection through novel mechanisms of action.
In September 2024, Reuters highlighted that Vaxcyte’s shares surged over 40% after Phase I data showed its experimental VAX‑31 vaccine generated immune responses across 31 pneumococcal serotypes—surpassing those of Prevnar 20—and paving the way for Phase III trials.
There presents a major opportunity for growth in the emerging markets of developing countries for pneumococcal vaccines. Pneumonia is a leading killer of children under age of 5 years in many low and middle-income nations. However, vaccination rates remain low currently due to the high cost of existing vaccines and lack of widespread availability.
As economies grow and living standards improve in developing regions like Asia Pacific, Latin America and Africa, more people will be able to afford vaccinating their kids. Countries are also focusing on expanding immunization coverage as part of their national healthcare goals. This growing demand, coupled with initiatives to make vaccines more affordable through subsidies and funding from global health organizations, can offer opportunities for manufacturers to tap new customer bases in emerging markets.
Vaccine prices tailored for price-sensitive developing world markets and innovative partnerships for local production and distribution can help address major expansion in these regions.
The Pneumococcal Conjugate Vaccines (PCVs) segment is projected to dominate the global pneumococcal vaccines market, commanding a significant 61.7% share in 2026. This leading position is driven by increasing patient awareness of PCVs' superior immunological protection and longer-lasting immunity compared to Pneumococcal Polysaccharide Vaccines (PPVs).
The rising adoption of PCVs reflects a strategic shift in global immunization practices, focusing on broader age group protection, especially among infants and immunocompromised populations. In contrast, the PPV segment holds a smaller share, primarily due to its limited effectiveness in younger populations and more restricted application in adult-specific use cases.
To learn more about this report, Request Free Sample
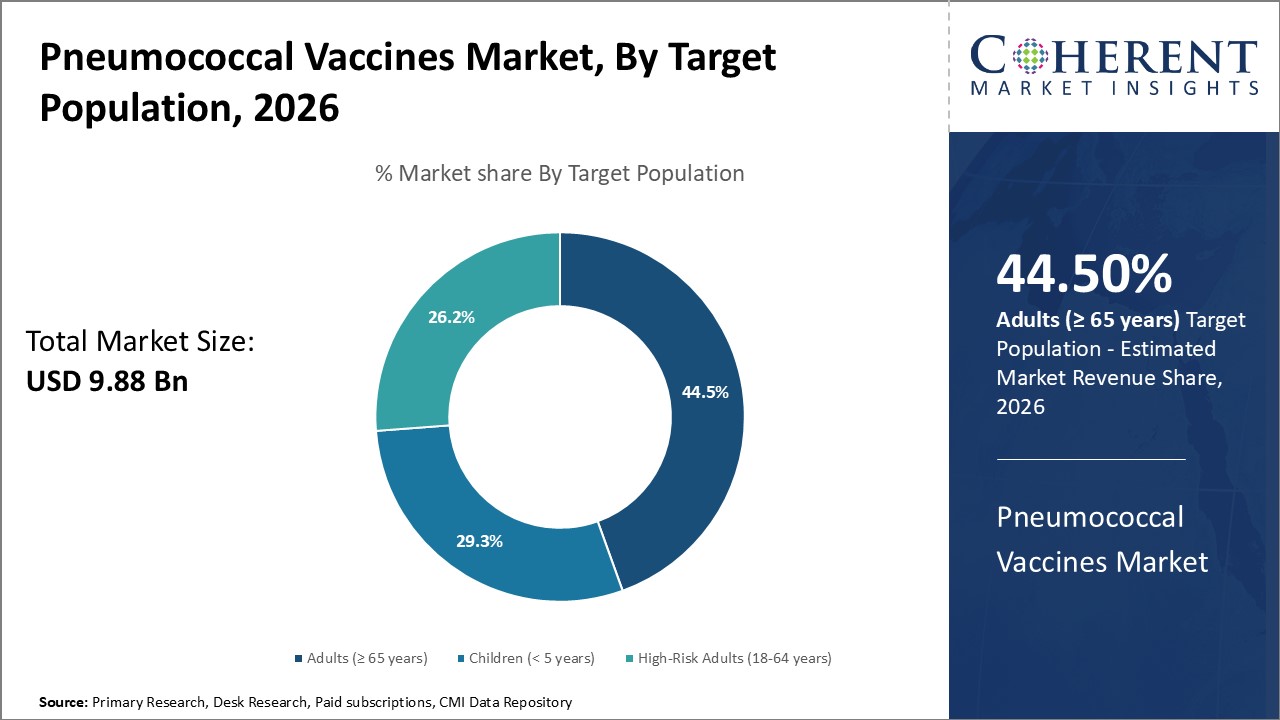
The Adults (≥ 65 years) segment is expected to capture the highest market share at 44.5% in 2026, reflecting a global healthcare trend emphasizing preventive immunization for the elderly. This demographic is particularly vulnerable to pneumococcal infections, making them a priority in both public and private healthcare agendas.
Increased life expectancy and healthcare reforms have prompted the expansion of adult immunization programs, especially in developed countries. This shift toward age-targeted vaccination supports market growth and encourages the development of adult-specific vaccine formulations and outreach strategies.
Hospital pharmacies are anticipated to lead the pneumococcal vaccines market by route of administration, contributing a dominant 61.5% share in 2026. This preference is attributed to the trust healthcare providers and patients place in hospital environments for vaccine administration, ensuring proper handling, storage, and monitoring.
Hospital-based delivery also enables better clinical support in case of adverse events, boosting overall safety perception. In comparison, retail and online pharmacy channels account for a smaller share, mainly due to limitations in cold-chain logistics and lower patient confidence in off-site immunization services.
To learn more about this report, Request Free Sample
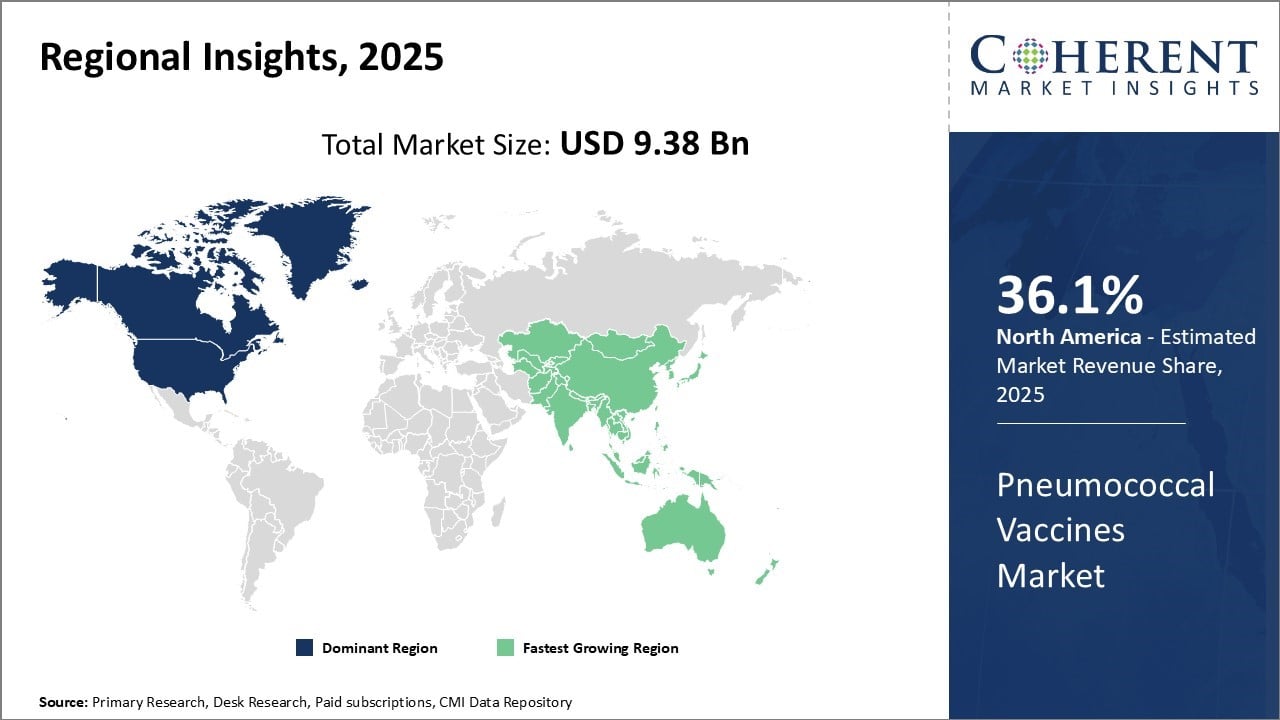
North America is expected to maintain a dominant position in the global pneumococcal vaccines market, holding a substantial 36.1% share in 2026. This regional leadership is driven by strong healthcare infrastructure, widespread government-backed immunization programs, and high public awareness regarding vaccine-preventable diseases.
The presence of advanced vaccine procurement and distribution systems ensures high coverage rates, especially among vulnerable groups such as the elderly and children. In addition, ongoing investments in R&D by key players and favorable regulatory frameworks further solidify North America's position as a mature and highly competitive market.
Europe follows closely with a significant market share, underpinned by universal healthcare systems, high vaccine coverage, and supportive public health policies. Countries such as Germany, the UK, and France have well-established adult and pediatric immunization programs that actively promote pneumococcal vaccination.
The region's commitment to preventive healthcare and routine immunization ensures consistent demand. Moreover, regulatory harmonization and centralized procurement mechanisms enhance vaccine accessibility, making Europe a stable and policy-driven contributor to the global pneumococcal vaccines market.
The United States plays a leading role in the North American pneumococcal vaccines market, which is projected to hold a 36.1% global share by 2026. Its dominance stems from a highly developed healthcare infrastructure, extensive public immunization programs led by the CDC, and broad insurance coverage for adult and pediatric vaccines. High public awareness, rapid regulatory approvals, and strong R&D investments by companies like Pfizer and Merck support widespread vaccine accessibility and innovation.
Canada also contributes significantly to the region’s market share, supported by its universal healthcare system and proactive vaccination policies. With structured programs targeting children and elderly populations, Canada ensures high coverage rates. Government-backed procurement systems and strategic partnerships with global vaccine manufacturers strengthen Canada’s position as a consistent and trusted pneumococcal vaccine consumer.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 9.88 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.3% | 2033 Value Projection: | USD 14.18 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pfizer Inc., Merck & Co., Inc., GlaxoSmithKline plc (GSK), Sanofi, CSL Limited, Serum Institute of India Pvt. Ltd., Vaxcyte, Inc., Astellas Pharma Inc., Panacea Biotec Ltd., Walvax Biotechnology Co., Ltd., Beijing Minhai Biotechnology Co., Ltd., Merck KGaA, Takeda Pharmaceutical Company Limited, AbbVie Inc., Novartis AG, Johnson & Johnson, Biovac, Bharat Biotech, Inovio Pharmaceuticals, Inc., Hilleman Laboratories Pvt. Ltd |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Abhijeet Kale is a results-driven management consultant with five years of specialized experience in the biotech and clinical diagnostics sectors. With a strong background in scientific research and business strategy, Abhijeet helps organizations identify potential revenue pockets, and in turn helping clients with market entry strategies. He assists clients in developing robust strategies for navigating FDA and EMA requirements.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients