The Polymerase Chain Reaction Market is estimated to be valued at USD 19.14 Bn in 2026 and is expected to reach USD 26.04 Bn by 2033, growing at a compound annual growth rate (CAGR) of 4.5% from 2026 to 2033.
The polymerase chain reaction market is advancing significantly with the rising demand for high-precision molecular diagnostics and advanced genetic analysis in the clinical, research, and forensic sectors. The growing consumer awareness regarding early disease detection as well as a strategic shift away from traditional culture-based testing are expected to propel market growth over the forecast period.
Polymerase chain reaction technology is a critical molecular biology method that allows precise amplification of specific DNA sequences for comprehensive examination. It serves as a potent method for identifying infectious pathogens, genetic mutations, and forensic markers. This makes polymerase chain reaction a vital component in modern genomic medicine and public health surveillance formulations.
The industry is entering a transformative period owing to the use of advanced automation technologies, a surge in point-of-care testing, and the integration of digital PCR in diverse fields like oncology and prenatal screening. These advancements enhance the sensitivity, speed, and absolute quantification capabilities of polymerase chain reaction platforms. As a result, the manufacturers have the ability to substitute manual laboratory workflows with automated high-performance systems. These systems improve diagnostic accuracy and assist in adhering to stringent international regulatory standards.
|
Current Event |
Description and the Impacts |
|
Industry Consolidation and Competitive Landscape |
|
|
Technological Innovations and Advancements |
|
|
Public Health and Epidemiological Factors |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of product type, the reagents & consumables segment contributes the highest share of 54.10% in 2026 of the market. The growth is attributable to continual repeat demand and the large number of assays performed in research and clinical environments. Unlike hardware, which involves a one-time capital investment, reagents like master mixes, primers, and enzymes are consumed in every cycle, thereby ensuring a continuous revenue stream. The rise of multiplex PCR which allow for the simultaneous detection of multiple targets, has also increased the requirement for sophisticated, high-quality chemical components. In addition, the shift toward digital PCR (dPCR) has spared a need for specialized consumables like nanoplates and droplet-generating oils, which are essential for achieving absolute quantification.
For instance, in November 2025, PCR Biosystems has introduced its new Lyo-Ready Inhibitor-Tolerant reagents. This new product combines the company's advanced inhibitor-tolerant chemistry with its growing collection of lyophilization-ready reagents designed for molecular diagnostics.
In terms of end user, the clinical diagnostics contributes the highest share of 44.30% in 2026 of the market. The growth is attributable to its role in infectious disease management, oncology, and prenatal testing. The PCR technology has become a gold standard for rapid identification of pathogens, a priority that has intensified with the global focus on pandemic preparedness and early intervention. The growing use of PCR in personalized oncology care for biomarker driven treatment selection is constituting to the segment's growth. The shift toward decentralized healthcare from centralized laboratories to point-of-care environments has increased the use of compact PCR systems in clinical facilities to enable quick results.
For instance, in June 2025, QIAGEN and GENCURIX, Inc. have formed a partnership to develop oncology assays for the QIAcuityDx platform. It is a high-performance digital PCR system intended for clinical diagnostics.
To learn more about this report, Request Free Sample
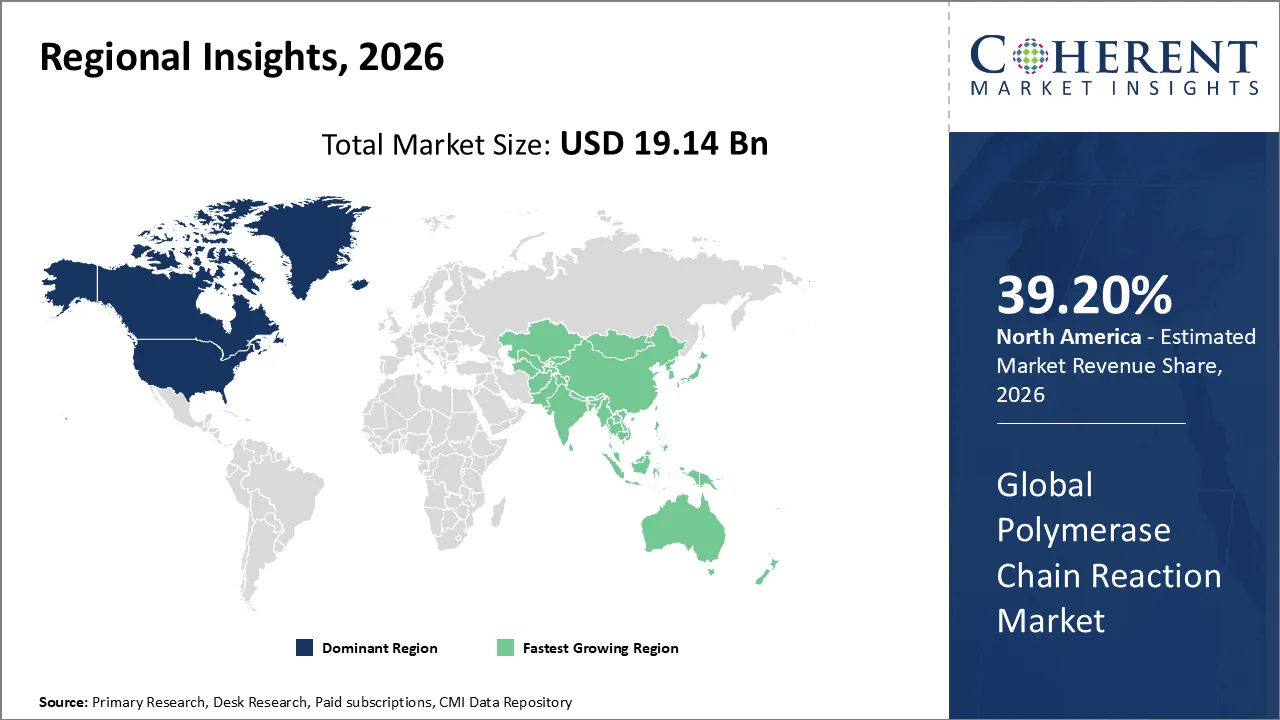
North America has maintained its position as the dominant region in the global Polymerase Chain Reaction (PCR) market, accounting for 39.20% in 2026. The growth owing to a sophisticated healthcare infrastructure, substantial federal research and development funding, and a swift transition toward precision medicine. The US is the primary driver of this growth. This is due to the rising prevalence of chronic diseases and the incorporation of PCR technology into routine clinical diagnostics for oncology and infectious diseases. The market has transitioned to a broader diagnostic focus, where quantitative PCR (qPCR) and digital PCR (dPCR) are utilized for early-stage cancer detection and monitoring minimal residual disease.
Technological advancements are currently centered on automation and speed, with a significant push toward Point-of-Care (POC) PCR testing that provides results in under 30 minutes. Major industry participants, including Thermo Fisher Scientific, Bio-Rad Laboratories, F. Hoffmann-La Roche, and Agilent Technologies, are currently focusing on the development of multi-target assays and AI-integrated systems to enhance diagnostic accuracy. This regional market is projected to sustain its steady growth until the end of the decade, while preserving a competitive advantage through ongoing innovations in high-throughput genomic analysis.
For instance, in July 2025, Bio-Rad Laboratories, Inc. has introduced four new Droplet Digital PCR (ddPCR) platforms. Among these newly launched instruments are the QX Continuum™ real-time polymerase chain reaction (ddPCR) system and the QX700™ series of ddPCR platforms.
The Asia Pacific region is currently the fastest growing market for polymerase chain reaction. The substantial government investments in genomics like the Genome India Project and China's Healthy China 2030 program, are responsible for the growth. These comprehensive population health programs utilize PCR technology for genetic screening and to promote the advancement of precision medicine. In addition, the region faces a burden of infectious diseases like tuberculosis and hepatitis, which ensures a steady and high-volume demand for PCR-based diagnostics.
As healthcare infrastructure modernizes across Southeast Asia and India, the shift from traditional testing methods to advanced molecular diagnostics is speeding up, positioning the region as a primary hub for global biotechnology growth. Chinese and Indian companies are competing with global leaders like Thermo Fisher Scientific and F. Hoffmann-La Roche by providing more affordable, high-throughput systems designed for local diagnostic centers. The Asia Pacific region is anticipated to experience substantial growth fueled by the expansion of clinical lab networks and supportive government policies aimed at localizing medical devices.
For instance, in December 2025, Roche Diagnostics China, the unit of Roche focused on diagnostic tests, instruments, and digital solutions, has announced the launch of its first quantitative polymerase chain reaction system developed in China.
The US is the largest market worldwide owing to its advanced healthcare infrastructure and heavy investment in personalized medicine. The market is defined by the widespread adoption of digital technologies that offer extreme sensitivity for identifying rare genetic mutations and monitoring cancer progression. There is a strong emphasis on integrating artificial intelligence into diagnostic platforms to automate the analysis of complex genomic sequences, thereby improving clinical decision-making process. Furthermore, many companies are currently restructuring their supply chains to produce essential chemical agents closer to home while developing specialized testing kits for the emerging field of cell and gene therapy.
China is the world’s fastest-growing market with the continuous government emphasis on domestic innovation and localization of medical technology production. The national health care strategy has now started to focus on early detection of cancers and control of infectious diseases in its population. There is a significant upscaling of portable and automated test systems within community health centers to enhance diagnostic access outside the major cities. Chinese manufacturers are increasingly successful at providing high-throughput platforms that combine sample processing with rapid genetic analysis to support the country’s massive clinical demands and its goal of etiological independence.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 19.14 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.5% | 2033 Value Projection: | USD 26.04 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
F. Hoffmann-La Roche Ltd, Bio-Rad Laboratories, Inc., Thermo Fisher Scientific Inc., Agilent Technologies, Inc., bioMérieux, S.A., Enzo Life Science, Inc., Abbott Laboratories, Takara Bio Inc., Becton, Dickinson and Company, and Danaher Corporation |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The rising incidence rate of infectious diseases acts as the driver for the growth of the polymerase chain reaction market. This is because pathogens such as influenza, tuberculosis, and HIV require sensitive and specific diagnostic techniques that can help in the early detection of the diseases. The PCR technology provides the necessary precision by amplifying genetic material to identify these infectious agents even at low viral loads. The global health challenges of the emerging respiratory viruses and drug-resistant bacterial agents have become more common. This has created an ever-increasing need for results of reliable molecular tests. The healthcare facilities have rapidly started adopting advanced PCR technologies to enable efficient screening and surveillance. This drive for faster diagnostic tests is creating demand for reagents and instruments. This in turn has helped develop technologies that allow laboratories to keep up with the constant requirement for accurate pathogen identification. These innovations ensure that laboratories can keep pace with the ongoing need for accurate pathogen identification in a globalized world.
The Polymerase Chain Reaction Market serves as a cornerstone in molecular diagnostics, research, and biotechnology applications. Market data indicate steady growth, driven by increased adoption in clinical laboratories, pharmaceutical R&D, and genomics research. Reagents and consumables consistently contribute the largest portion of revenue, while real-time PCR remains the dominant technique. Instrumentation, including thermocyclers and digital PCR systems, continues to support market expansion as laboratories upgrade equipment for improved efficiency and throughput.
Regionally, North America remains the leading contributor, supported by robust healthcare infrastructure, high research investment, and widespread molecular diagnostics deployment. Asia-Pacific is identified as the fastest-growing region, propelled by expanding healthcare capacity, local manufacturing, and rising adoption of PCR technologies in research and clinical settings.
Technological advancements such as digital PCR and automation have improved assay precision, reproducibility, and throughput, enabling large-scale testing and complex biomarker analysis. End users include clinical laboratories, hospital systems, CROs, and pharmaceutical R&D centers, reflecting broad demand across diagnostics and research sectors.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients