Global respiratory inhalers market is estimated to be valued at USD 37.28 Bn in 2026 and is expected to reach USD 56.79 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 6.2% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
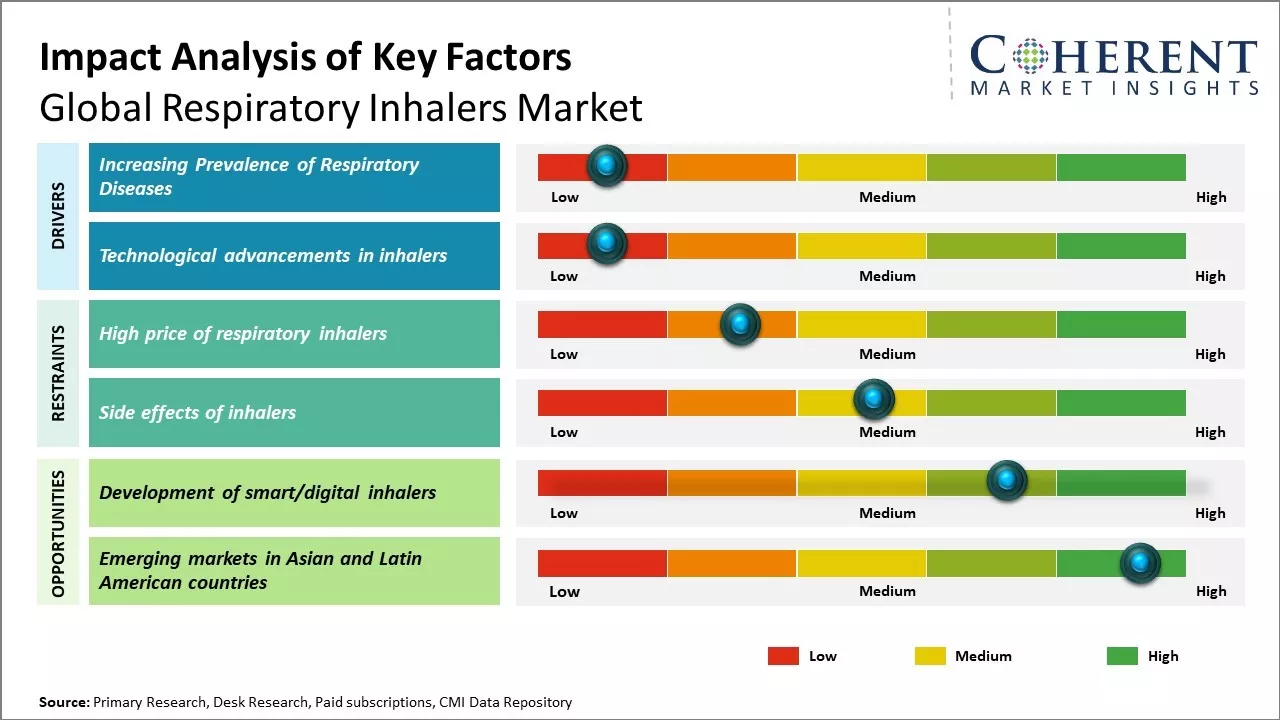
Rising prevalence of respiratory diseases such as asthma and chronic obstructive pulmonary disease (COPD) can boost demand for respiratory inhalers. Asthma currently affects over 300 million people worldwide and is one of the most common chronic diseases among children. Incorrect usage of inhalers and lack of disease awareness regarding proper utilization of inhalers in developing nations can hamper the market growth. However, ongoing research & development activities for development of more effective and affordable respiratory inhalers can offer growth opportunities for the market players.
Increasing Prevalence of Respiratory Diseases
Rising prevalence of respiratory diseases such as asthma and COPD across the world can boost demand for respiratory inhalers. Chronic respiratory conditions tend to worsen with age, and with an increasingly aging global population, the problem is further exacerbated. Data from health organizations shows a steady growth in the number of people diagnosed with asthma and COPD each year. This upward trend is seen prominently in developed as well as developing nations. For instance, as per some studies, around 235 million people are estimated to be suffering from asthma currently worldwide. This number can reach 400 million by 2025. Exposure to indoor and outdoor pollution, occupational hazards, and smoking are the primary causes of respiratory illnesses. As patient pool expands consistently, respiratory inhalers are used as the preferred mode of drug delivery in asthma and COPD management. Inhaled medications allow for direct targeting of the airways, ensuring better therapeutic response with minimal systemic side effects. This also permits use of higher dose with greater safety. The ongoing development and approval of newer and more effective inhaled drugs can boost inhaler consumption. with progressive morbidity and mortality associated with respiratory infections, inhaler therapy adoption is likely to maintain its northward trajectory in the coming years from both healthcare as well as patient perspectives. Unrelenting urbanization and pollution levels across large sections of the globe can cause asthma and COPD.
Get actionable strategies to beat competition: Request Free Sample
Technological advancements in inhalers
Technological advancements in inhalers can drive the global respiratory inhalers market growth. Inhalers have increasingly become smart and digital with new features which help patients better manage their chronic respiratory conditions like asthma and COPD. Many new digital inhalers are equipped with sensors that can track usage data and send it via Bluetooth to a app in the patient's smartphone. This provides both patients and doctors real-time insights into medication adherence and symptoms. The data collection functionality is useful for doctors to monitor patient's condition remotely and tweak treatment plans accordingly. It also helps patients to take medications on time as dosage schedule and remaining timeline is visible on mobile apps. Some high-end smart inhalers also have in-built reminders and feedback systems to ensure proper inhalation technique. All these features are proving especially helpful for the growing elderly population suffering from multiple chronic conditions. The tracked usage data is also valuable for pharmaceutical companies to enhance therapeutics and services over time. On the product front, new single-dose dry powder inhalers that are easy-to-use and require less coordination during inhalation compared to older variants have witnessed huge demand. Furthermore, the introduction of affordable generic versions of blockbuster respiratory drugs has made treatment more accessible to patients globally. On May 2024, According to World Health Organization (WHO), the number of people suffering from asthma is estimated to increase by over 100 million globally by 2025. This growing disease burden coupled with technological upgrades in medication delivery methods can drive the respiratory inhalers market growth.
Key Takeaways from Analyst
Rising prevalence of respiratory diseases such as asthma and chronic obstructive pulmonary disease (COPD) around the world can boost demand for inhalers. Growing geriatric population who are more susceptible to such illnesses can also drive the market growth. However, patent expirations of blockbuster drugs can pose challenge for leading manufacturers in the short term.
North America currently dominates the respiratory inhalers market due to high healthcare expenditure and growing healthcare awareness in the region. However, Asia Pacific market is likely to emerge as the most lucrative region due to its massive patient pool and rising healthcare standards.
The dry powder inhalers (DPI) segment holds the largest share, owing to benefits such as better stability and increased efficacy compared to other types. Emergence of digital inhalers integrated with sensors and connectivity offers a major opportunity for players to stay ahead of the competition. Major companies need to focus on new product launches, especially those targeting COPD patients, to strengthen their market position.
Market Challenges: High price of respiratory inhalers
The high price of respiratory inhalers can hamper the global respiratory inhalers market growth. Respiratory diseases are highly prevalent across the world and cost of treatment has been rising constantly. According to the World Health Organization (WHO), approximately 265 million people suffer from asthma globally and it impacts the quality of life of patients and their families. However, high cost of inhalers makes it very difficult for many people to afford prescribed treatments on a regular basis. This is a major concern particularly in developing nations where a significant percentage of the population lacks health insurance or means to pay out of their pocket for expensive medications. For instance, preventer inhalers that are required to be taken daily cost upwards of US$ 100-150 per month in many countries. Similarly, reliever inhalers needed during asthma attacks can cost US$ 30-50 each time without insurance. This disproportionately burdens families with low income. As a result, many patients are either forced to abandon preventive treatments or rely only on cheaper options like oral medications. This leads to poor control of symptoms, increased risk of exacerbations, rise in emergency room visits and hospitalizations. According to a WHO report published in 2022, around 1.6 million annual emergency room visits and hospitalizations due to asthma in the U.S. could be avoided with good control through affordable preventive inhalers. The indirect economic burden due to lost work and wages has been estimated to be over US$ 50 billion annually in the U.S. High out-of-pocket costs discourage regular prescription fills that are crucial for effective long-term management of respiratory diseases.
Market Opportunities: Development of smart/digital inhalers
Smart/digital inhalers have the potential to revolutionize the respiratory inhalers market and significantly improve patient outcomes. Traditional inhalers are sometimes misused which can reduce their effectiveness. Smart inhalers address this issue by incorporating connected digital technologies that track usage and provide feedback to patients and doctors. These can sense when an inhaler is shaken, opened and closed and record how much medication was released. This data is sent via Bluetooth to a mobile app, allowing patients to better manage their condition and ensure proper administration of medications. It also provides physicians insights to see adherence rates and make more informed care decisions. Many major pharmaceutical companies are now investing heavily in the development of smart inhalers. For example, GSK partnered with the Adherium, a digital health company, to integrate the latter's SmartTurbo technology into GSK's Ellipta products. Clinical trials have found this digital Ellipta improved asthma control and led to 50% fewer severe exacerbations compared to the regular inhaler, as per data from National Institutes of Health in 2021. As the technology matures and more versatile products enter the market, smart inhalers have the potential to transform respiratory care from crisis management to prevention. Their connected functionalities could encourage greater accountability and long-term behavioral changes in patients. This is expected to significantly reduce healthcare costs associated with exacerbation treatment like emergency room visits and hospitalization.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
By Type - Technological Advancements Drive Digitally Operated Segment
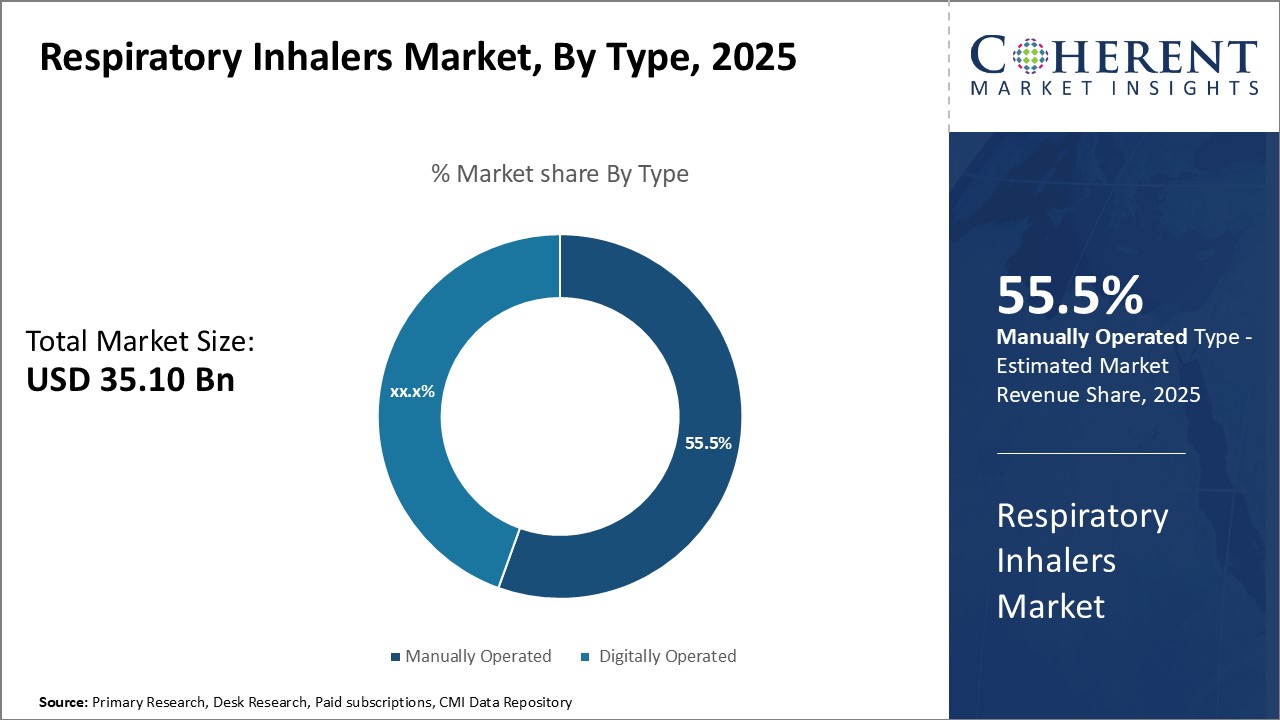
In terms of type, digitally operated segment is estimated to contribute the highest market share of 56.5% in 2026, owing to continuous technological advancements. Digitally operated inhalers are integrated with sensors and smart devices that provide real-time usage data and remind patients to take their prescribed doses. This enhances treatment adherence and monitoring. The digitization has also made these inhalers more user-friendly through automated dose counters and feedback systems. Manufacturers are increasingly launching Bluetooth-enabled and app-connected inhalers to improve medication management remotely. The convenience and effectiveness offered by digital capabilities are boosting preference over conventional manually operated inhalers. Tech giants are venturing into digital therapeutics and developing combination therapies that integrate inhalers with AI platforms. This can transform respiratory care through personalized treatment approaches.
To learn more about this report, Request Free Sample
By Application - Asthma Indications Stimulate Higher Adoption
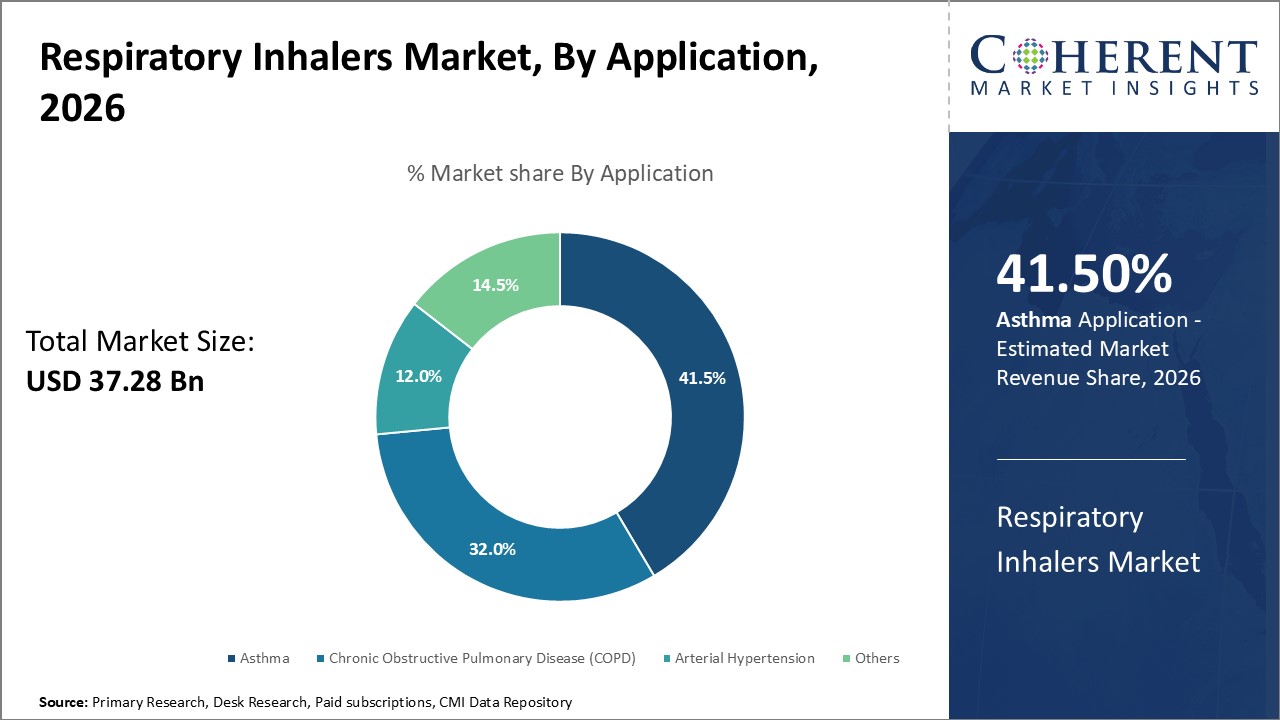
In terms of application, asthma segment is estimated to contribute the highest market share of 41.5% in 2026, owing to rising prevalence of the condition globally. Asthma is a chronic inflammatory disease characterized by obstruction of airflow in the lungs. It affects people of all ages with varying severity levels. Inhalers provide quick relief during asthma attacks and are prescribed for long-term control of symptoms. Increased environmental pollution, lifestyle changes and genetic predisposition can increase the asthma burden. This wide patient pool and life-long nature of treatment boosts demand for respiratory inhalers that are used for asthma. Moreover, inhalers are the preferred mode of administration for asthma as these deliver medication directly to the airways for faster action. Growing awareness about asthma management and new product launches attracts patients towards inhalers.
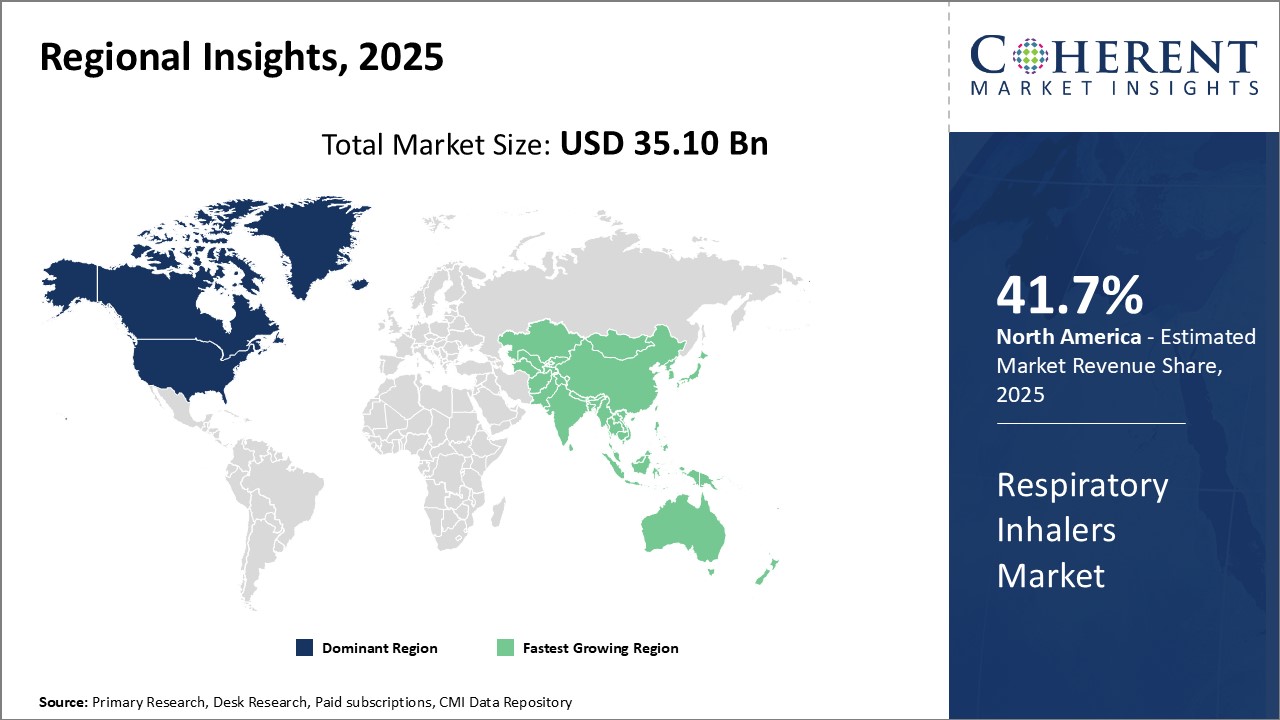
Need a Different Region or Segment? Request Free Sample
North America dominates the respiratory inhalers market with an estimated market share of 41.7% in 2026 The presence of leading global pharmaceutical companies such as GSK, Teva, Chiesi, and Sunovion provides the region with easy access to most innovative portfolio of inhalers. Majority of the phase 3 clinical trials for new inhalers are conducted across the U.S. and Canada which ensures early introduction of novel products in the region. Furthermore, North America has a highly developed healthcare infrastructure and favorable reimbursement policies supporting increased adoption of respiratory inhalers.
The region is also a hub for generic inhalers manufacturing. Several domestic manufacturers focus on developing low-cost generic versions, making respiratory therapy more affordable. This has increased the reach of inhalers to a broader patient base suffering from respiratory disorders like asthma and COPD. Growing geriatric population susceptible to chronic respiratory conditions in the U.S. and Canada can boost demand. High awareness levels among healthcare providers and patients regarding inhalers as primary mode of treatment administration further strengthens North America's position as the most profitable market.
Asia Pacific region is emerging as the fastest growing market for respiratory inhalers. Countries like China, India, and Japan have a massive patient pool suffering from both communicable and non-communicable respiratory diseases. This large prevalence rate along with the improving access to healthcare present significant growth opportunities. Manufacturers are focusing their expansion strategies in Asia Pacific by establishing local manufacturing plants and distribution networks.
Respiratory Inhalers Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 37.28 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.2% | 2033 Value Projection: | USD 56.79 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
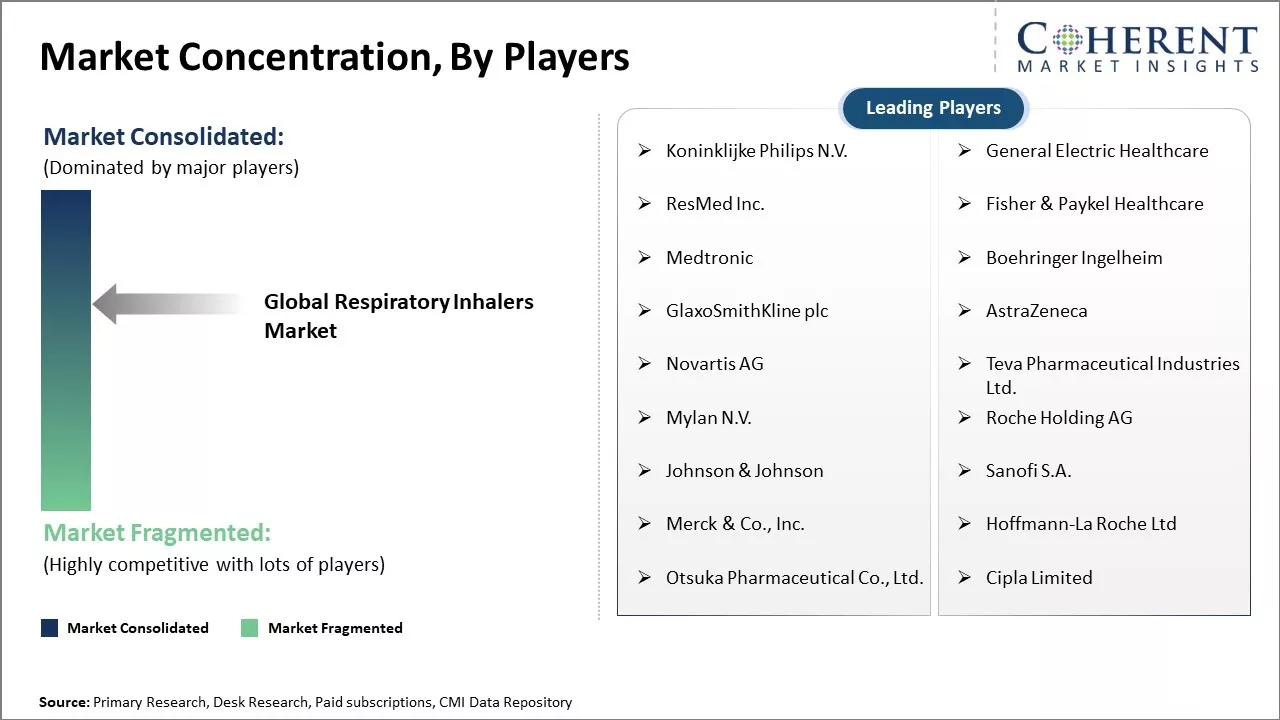
Koninklijke Philips N.V., General Electric Healthcare, ResMed Inc., Fisher & Paykel Healthcare, Medtronic, Boehringer Ingelheim, GlaxoSmithKline plc, AstraZeneca, Novartis AG, Teva Pharmaceutical Industries Ltd., Mylan N.V., Roche Holding AG, Johnson & Johnson, Sanofi S.A., Merck & Co., Inc., Hoffmann-La Roche Ltd, Otsuka Pharmaceutical Co., Ltd., Cipla Limited |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: Global Respiratory Inhalers Market includes various products used for the treatment of respiratory diseases like asthma and chronic obstructive pulmonary disease (COPD). Respiratory inhalers help deliver medication directly to the airways and lungs. Some of the major products in this market are metered dose inhalers, dry powder inhalers and nebulizers. The global market has been growing due to the high prevalence of respiratory diseases and rising awareness about the effective treatment options available in the form of respiratory inhalers. Key players in this market are constantly innovating and developing more effective and user-friendly respiratory inhalers.
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients