Global chronic obstructive pulmonary disease (COPD) treatment market is estimated to be valued at USD 21.59 Bn in 2026 and is expected to reach USD 32.68 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 6.1% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
Global chronic obstructive pulmonary disease (COPD) treatment market has been witnessing growth over the past few years due to huge adoption of inhaled medications and bronchodilators for management of COPD symptoms. Furthermore, pipeline drugs with novel mechanisms of action are being evaluated for treatment of COPD, and this can expand therapeutic options. Many pharmaceutical companies are also investing heavily in development of combination therapies to ensure better treatment outcomes. However, high treatment costs can hamper the market growth. Increasing global disease burden of COPD supported by growing awareness regarding its management can boost demand for COPD therapeutics during the forecast period.
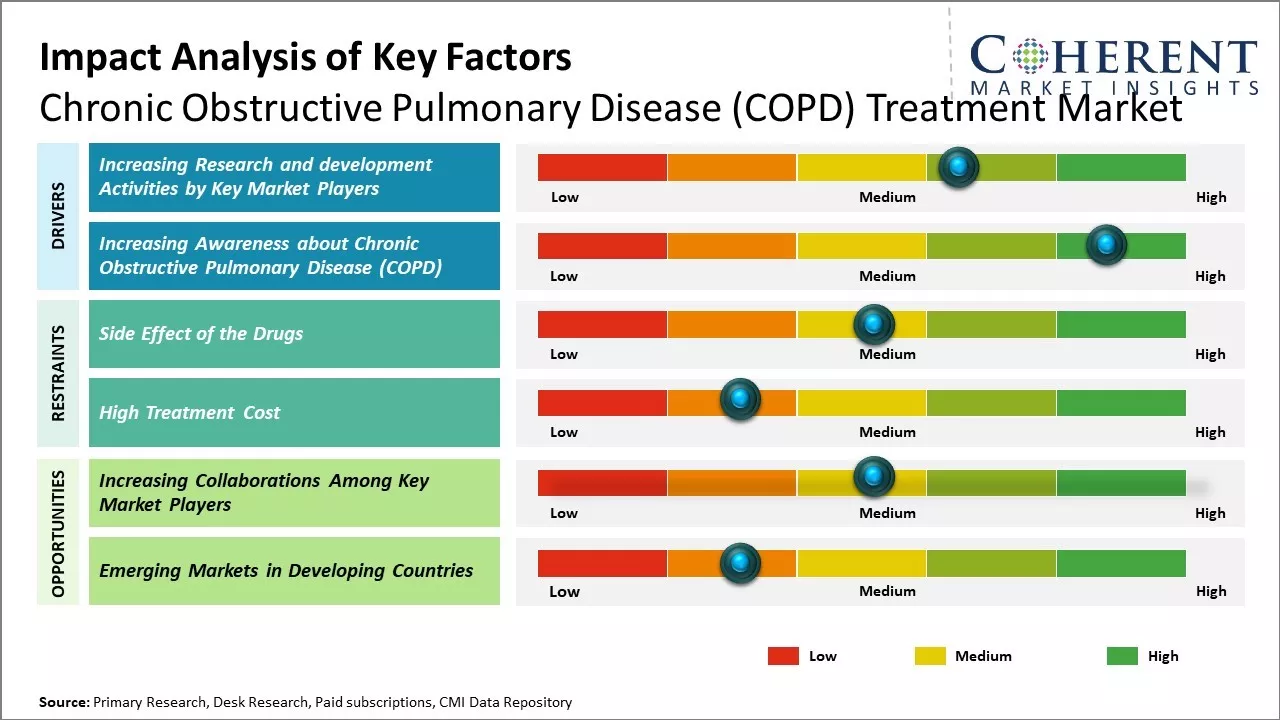
Increasing Research and development Activities by Key Market Players
Increasing research and development activities by key market players is expected to drive the market growth over the forecast period. For instance, on March 14, 2026, AstraZeneca, a global pharmaceutical company, announced that its triple combination inhaled therapy- Breztri Aerosphere (budesonide/glycopyrrolate/formoterol fumarate) will be evaluated in a new phase 3 trial in patients with chronic obstructive pulmonary disease (COPD) and elevated cardiopulmonary risk.
Get actionable strategies to beat competition: Request Free Sample
Increasing Awareness about Chronic Obstructive Pulmonary Disease (COPD)Increasing adoption of inorganic growth strategies such as investments and fundings is expected to drive the market growth over the forecast period. For instance, in November 2021, according to the data published by the National Heart, Lung and Blood Institute, each November, the chronic obstructive pulmonary disease (COPD) community comes together to promote better understanding of chronic obstructive pulmonary disease (COPD), a progressive lung disease that affects millions across the US. Increasing awareness about chronic obstructive pulmonary disease (COPD) and its symptoms is important because early diagnosis and treatment can improve quality of life.
Key Takeaways from Analyst:
Global chronic obstructive pulmonary disease (COPD) treatment market growth is primarily driven by increasing prevalence of COPD worldwide. Rising geriatric population, which are at a higher risk of developing COPD, further aids the market growth. Growing number of smokers, along with rising air pollution levels in cities, are major drivers boosting demand for COPD treatments.
The market faces challenges from limited awareness regarding COPD symptoms in developing nations. This acts as a restraint as many cases remain undiagnosed. Moreover, the lack of disease-modifying drugs for COPD treatment poses a challenge. Treatment options are focused on managing symptoms rather than curing the condition.
The Asia Pacific region has significant growth opportunities attributed to its large population base and growing pollution levels in major economies. China and India have a high prevalence of COPD cases and show great potential. North America currently dominates the market but is expected to lose shares to Asia due to faster adoption and increasing healthcare expenditure in emerging nations.
Market Challenge – Side Effect of the Drugs
The side effect of the drugs used in treating chronic obstructive pulmonary disease can hamper the market growth over the forecast period. For instance, according to the data published by the National Center for Biotechnology Information, there are some severe side effects associated with the drugs used in the treatment of the chronic obstructive pulmonary disease like bruising, oral infections, and hoarseness. These medications are useful for people with frequent exacerbations of chronic obstructive pulmonary disease. Examples of inhaled steroids include Fluticasone (Flovent HFA). Respiratory infections are observed in people with COPD who are more likely to catch colds, flu and pneumonia. Chronic obstructive pulmonary disease can cause many complications like high blood pressure in lung arteries, depression, heart problems, and lung cancer in some cases.
Market Opportunity – Increasing Collaborations among Key Market Players
For instance, in March 2023, UC Davis Health, an health center in the U.S., and Propeller Health announced a new collaboration that will offer personalized treatment for high-risk patients with asthma and chronic obstructive pulmonary disease (COPD). UC Davis Health will provide the Propeller Health an remote monitoring program to eligible patients. It includes sensors, a mobile app, web portal, and personalized support. The sensors attach to a patient’s inhaler to capture unique signals that record events, such as medication usage or respiration. This data will be transmitted to UC Davis Health’s electronic health record system to support patient enrollment and remote patient monitoring via single sign-on. The remote monitoring system may be expanded to patients in other University of California health systems.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
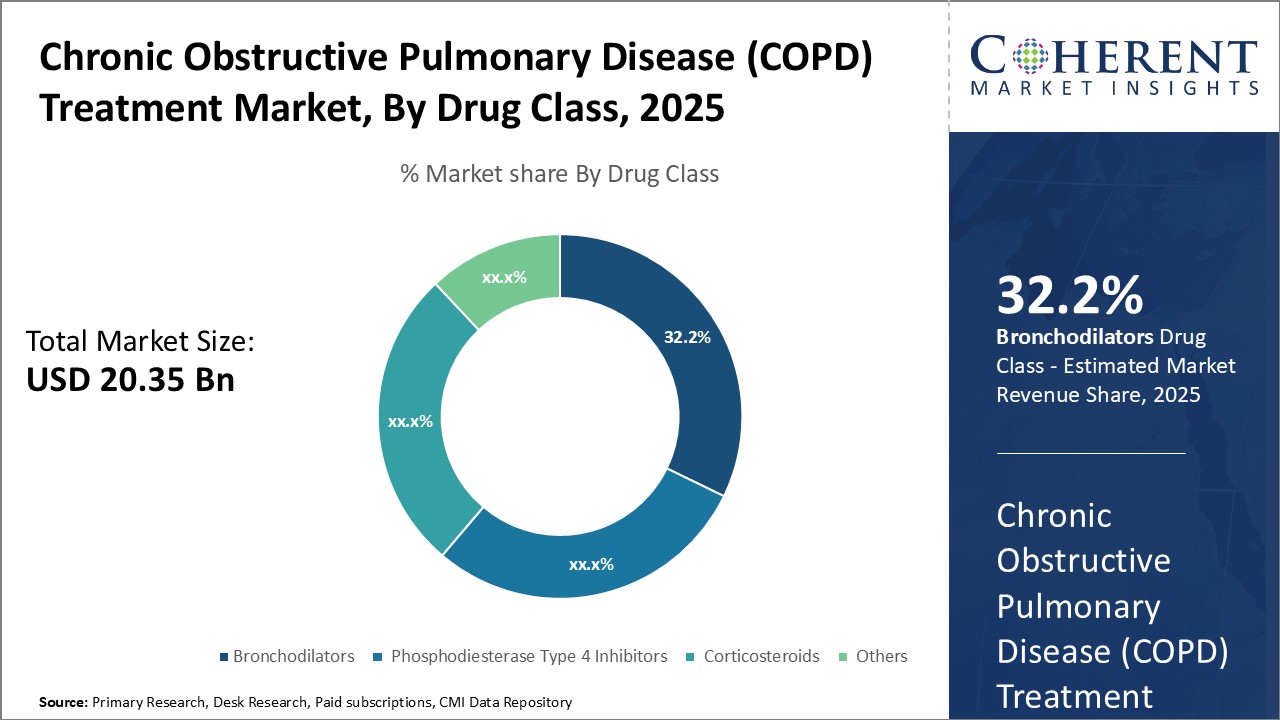
Insights, By Drug Class: Bronchodilators segment leads market share due to effectiveness and prevalence of symptomsDrug Class segment is sub-segmented into bronchodilators, phosphodiesterase type 4 inhibitors, corticosteroids, and others. Bronchodilators sub-segment is estimated to hold 33.2% of the market share in 2026, owing to their effectiveness in relieving bronchospasms and preventing exacerbations. Bronchodilators work by relaxing and opening the airways, thus, providing rapid symptom relief from breathing difficulties that many COPD patients experience on a daily or weekly basis. The prevalence and impact of such symptoms among COPD populations has led bronchodilators to become a frontline therapeutic option. Both short-acting and long-acting bronchodilators are widely prescribed depending on the severity of symptoms and frequency of exacerbations. The convenience of inhaled delivery also enhances patient compliance as compared to other drug classes. With the progression of COPD lung function decline, bronchodilators remain a mainstay treatment approach to controlling respiratory symptoms and related quality of life impacts.
-treatment-market-by-route-of-administration.webp)
To learn more about this report, Request Free Sample
Insights, By Route of Administration: Inhalation segment dominates due to targeted delivery and rapid onset
Route of Administration segment is sub-segmented into oral and inhalation. Inhalation sub-segment is estimated to hold 53.9% of the market share in 2026, owing to its non-invasive and targeted delivery directly to the lungs. Inhaled therapies bypass the gastrointestinal system, allowing for a much more rapid onset of therapeutic effects compared to oral medications. This makes inhalers very well-suited for providing fast relief during COPD exacerbations characterized by acute symptoms like shortness of breath, coughing, and wheezing. Their localized delivery also allows inhaled drugs to use lower doses than would be required systemically through oral administration, thus, reducing the risk of side effects. Inhalers have also witnessed significant device innovations to improve accuracy and ease-of-use, enhancing patient preferences and compliance over oral therapies.
Insights, By Distribution Channel: Hospital pharmacies lead in complex cases and exacerbation management
Distribution Channel segment is sub-segmented into hospital pharmacies, retail pharmacies, and online pharmacies. Hospital pharmacies sub-segment is estimated to hold 37.1% of the market share in 2026, owing to their central role in acute exacerbation treatment and managing advanced COPD patients. When COPD symptoms worsen to a point requiring medical intervention or hospitalization, specialized care and monitoring is usually provided in-hospital under medical supervision. This involves personalized exacerbation treatment plans defined by a pulmonologist or respiratory therapist that often include multiple drug therapies, supplemental oxygen, nebulizers, or ventilator support - making hospital pharmacies pivotal in coordinating such complex regimens. For patients with very severe COPD, the additional layer of support from hospital-based specialists and programs also helps optimize therapeutic outcomes. Post-hospitalization as well, coordinated care through hospital outpatient services enhances medication adherence which is critical to prevent readmissions.
Need a Different Region or Segment? Request Free Sample
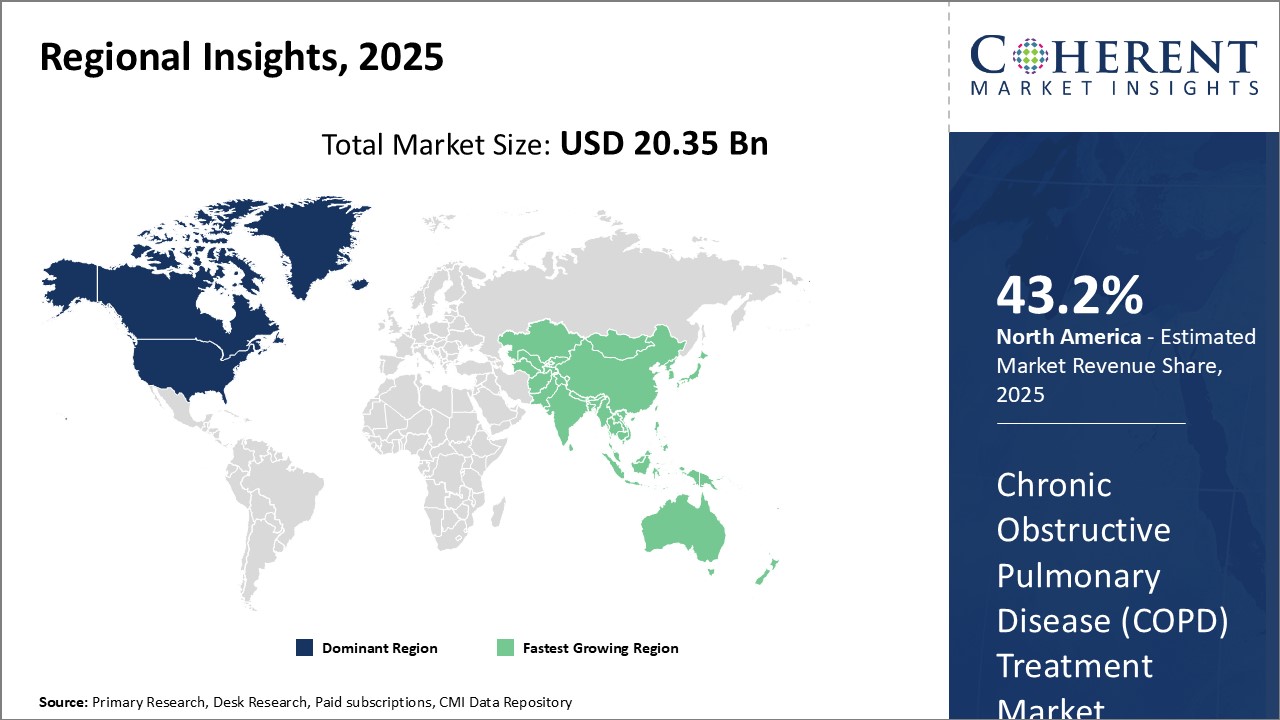
North America remains the dominant region in the global chronic obstructive pulmonary disease (COPD) treatment market and is estimated to hold 43.2% of the market share in 2026. The region is home to some of the largest pharmaceutical companies researching and developing innovative drugs and devices for COPD treatment. With high acceptance of new treatment options and diagnostic methods among patients and healthcare providers, North America provides ample growth opportunities for novel molecules and combination therapies. The strong presence of private and public healthcare systems in the U.S. and Canada ensures widespread insurance coverage and affordability of COPD medications. This high demand and availability of funds have encouraged significant investments by manufacturers in the area of COPD.
The Asia Pacific region is emerging as the fastest growing market for COPD treatment worldwide. Factors such as growing geriatric population, rising air pollution levels in urban cities, and increasing healthcare expenditures are driving the market growth. Countries like China and India have a huge patient population suffering from COPD due to rampant smoking habits and exposure to biomass fuel emissions. This significant disease prevalence attracts several multinational pharmaceutical giants to focus on the Asia Pacific region. These are launching generic and low-cost specialty drugs while strengthening local distribution networks to cater to the massive unmet needs. The improvements in the diagnosis rate through improved diagnostic infrastructure and availability of pulmonary function tests in major hospitals are aiding early detection and treatment-seeking behavior. Rising income levels and awareness about COPD are expected to drive the market growth.
Global Chronic Obstructive Pulmonary Disease (COPD) Treatment Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 21.59 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.1% | 2033 Value Projection: | USD 32.68 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
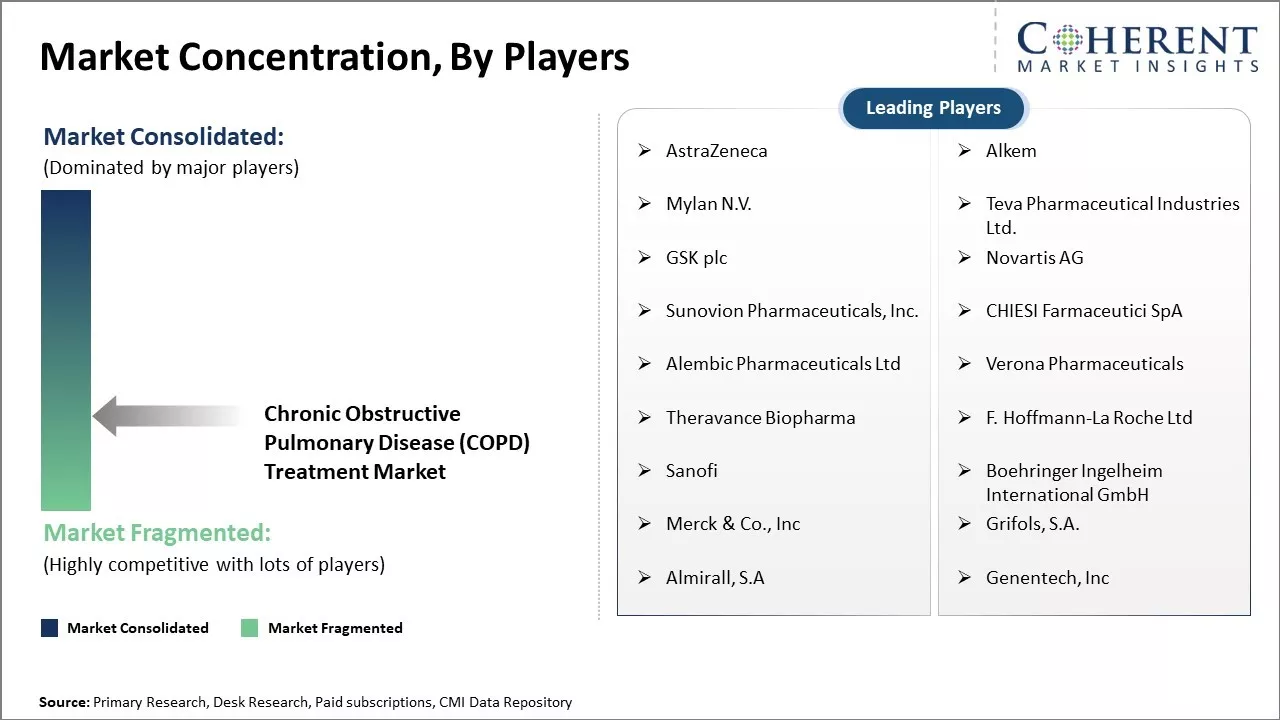
| Companies covered: |
AstraZeneca, Alkem, Mylan N.V., Teva Pharmaceutical Industries Ltd., GSK plc, Novartis AG, Sunovion Pharmaceuticals, Inc., CHIESI Farmaceutici SpA, Alembic Pharmaceuticals Ltd, Verona Pharmaceuticals, Theravance Biopharma, F. Hoffmann-La Roche Ltd, Sanofi, Boehringer Ingelheim International GmbH, Merck & Co., Inc, Grifols, S.A., Almirall, S.A, Genentech, Inc |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: Chronic obstructive pulmonary disease includes chronic bronchitis, in which the bronchi (large air passages) are inflamed and scarred, and emphysema, in which the alveoli (tiny air sacs) are damaged. Chronic obstructive pulmonary disease (COPD) presents itself with mild, moderate, and severe symptoms. Treatment of chronic obstructive pulmonary disease (COPD) involves stepwise non-pharmacological and pharmacological interventions. Non-pharmacological interventions include risk reduction (smoking cessation) and preventing and treating co-morbidities (cardiovascular disease, anxiety, depression, and others). Pharmacological interventions include prescribing short acting bronchodilators, long acting bronchodilators, and others.
Primary Research Interviews:
Databases:
Magazines:
Journals:
Newspapers:
Associations:
Public Domain Sources:
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients