Cardiopulmonary Bypass Equipment Market is estimated to be valued at USD 217.2 Mn in 2026 and is expected to reach USD 267.2 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 3.0% from 2026 to 2033.
To learn more about this report, Request Free Sample
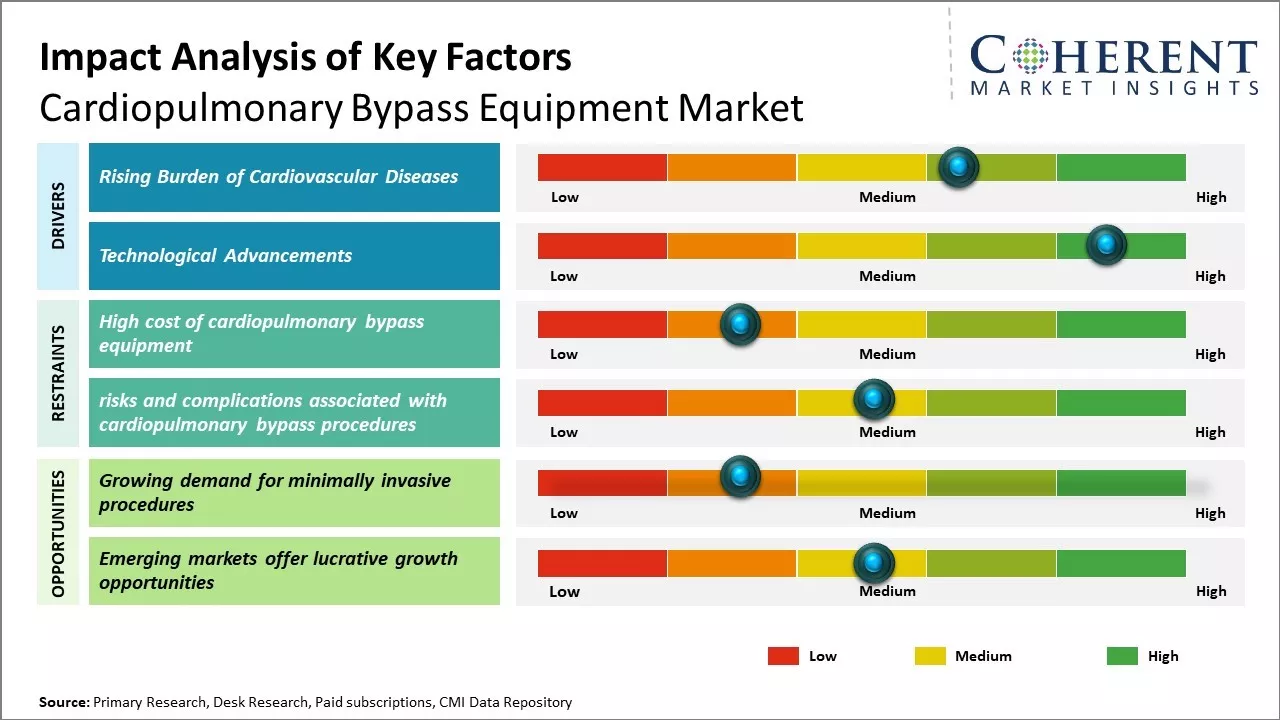
Global cardiopulmonary bypass equipment market is expected to witness a steady growth over the forecast period due to increasing prevalence of cardiovascular diseases and growing demand for minimally invasive surgeries. Moreover, advancement in clinical research for improving bypass equipment and rising healthcare expenditure can also drive the market growth during this period. However, high costs associated with cardiopulmonary bypass surgeries and lack of awareness in developing regions are expected to hinder the market growth.
For instance, in October 2024, Prime Minister of India virtually inaugurated new facilities at AIIMS-Kalyani in North 24 Parganas district of West Bengal, which includes a cardiac laboratory and a generic medicine outlet. The cardiac Cath laboratory has imaging equipment to visualize the heart’s chamber’s arteries and other parts, while a new cardiopulmonary bypass (CPB) machine can temporarily take over the functions of the heart and lungs during a surgery.
|
Current Events |
Description and its Impact |
|
Shift Toward Outpatient Cardiac Surgeries |
|
|
Regulatory and Reimbursement Shifts |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
By Product Type, Single Roller Pump Segment is estimated to account for the highest market share of 61.6% in 2026 because cardiac surgeons have been using it for a long time and because it is more affordable than other pump designs. For the majority of operations, the single roller pump is the preferred option as it was one of the first pump designs created for cardiopulmonary bypass treatments. Surgeons can utilize it with ease due to its straightforward roller based.
Compared to more intricate multi-roller pumps, the design of a single roller pump results in lower production and cardiopulmonary bypass equipment maintenance procedures costs. Healthcare establishments can now afford single roller pumps due to this cost advantage.
For instance, in February 2025, Ellesmere Town Council is thrilled to announce the launch of a crowdfunding campaign aimed at building a pump track in the town, at Berwyn View. This innovative project, designed to provide a safe and fun environment for riders of all ages and skill levels, will promote active lifestyles, enhance community engagement, and support local youth. The pump track will feature a series of rollers, berms, and jumps, allowing cyclists, skateboarders, and scooter riders to build skills and have fun in a controlled setting.
To learn more about this report, Request Free Sample
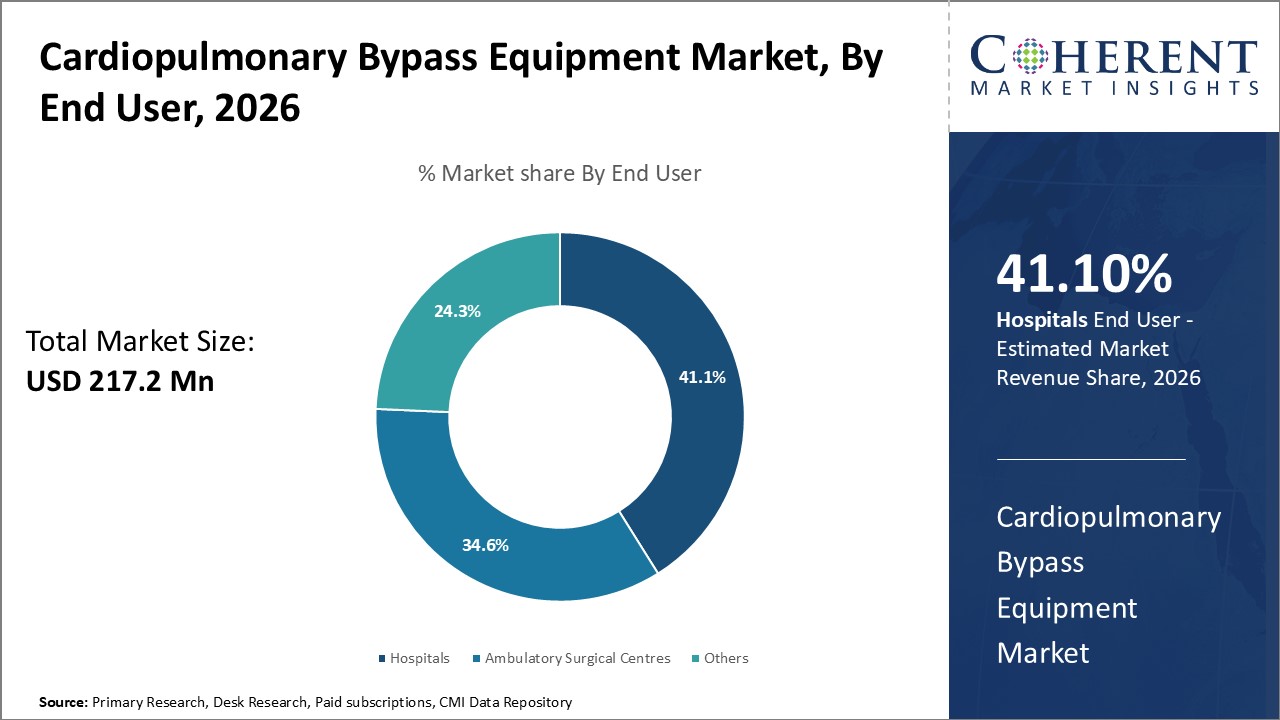
Among end user, hospitals segment is estimated to contribute the highest market share of 41.1% in 2026 due to a significant number of cases are managed in hospital settings. Hospitals perform the most cardiopulmonary bypass surgeries annually because they are the default caregivers for life-threatening illnesses like cardiovascular disease. Hospitals will very certainly have the greatest need for cardiopulmonary bypass equipment due to the size of their cardiac programs. Another benefit hospitals possess is established infrastructure for complex surgeries. Operating rooms, specialist staff, and postoperative care units allow hospitals to comfortably accommodate cardiopulmonary bypass procedures. Resources for equipment purchase and maintenance are also more readily available to hospitals as compared to other end users.
As large urban hospitals, in particular, have sizable capital budgets, purchasing high-end cardiopulmonary bypass devices is an affordable investment. Long-term support for follow-up service agreements is also possible. Hospitals are viewed by equipment makers as a low risk customer sector that is less susceptible to financial limitations. Consequently, cardiopulmonary bypass equipment manufacturers give hospital customers specific needs top priority, solidifying their position as industry leaders.
For instance, in June 2024, A new, robotic approach to heart bypass surgery is now being offered to patients at University Hospitals (UH) with great success. Two UH Harrington Heart & Vascular Institute providers, are currently the only surgeons in Ohio performing this specific type of procedure using the Da Vinci surgical robot. This robot assists surgeons in performing coronary artery bypass grafting (CABG) through a minimally invasive approach which leaves patients with less pain and a shorter recovery after surgery.

To learn more about this report, Request Free Sample
North America has been leading the cardiopulmonary bypass equipment market, accounting for over 40.7% of total transplants worldwide last year. Its cutting edge transplant facilities, encouraging reimbursement practices, and growing awareness are the reasons for the region’s dominance. The market in the U.S. alone is twice as large as that of any other nation. North American medical device businesses make significant investments in R&D to ensure ongoing innovation in organ.
For instance, in September 2024, Vesalio, a privately held medical device company focused on advancing patient care in vascular diseases has announced the U.S. commercialization of its pVasc Thrombectomy System. The mechanical device is designed for the non-surgical removal of peripheral occlusions, which are blockages in the arteries of the arms and legs caused by blood clots, inflammation, and narrowing blood vessels.
Asia Pacific has emerged as the fastest-growing market due to rising chronic kidney disease incidences, growing incomes, and government initiatives to boost organ donation. Transplants have increased, especially in China, as a result of initiatives to improve local production, alter donation regulations, and lower costs. Due to the region’s promising growth prospects, this has drawn international medical technology companies to invest in partnerships and local manufacturing in China.
For instance, in April 2024, the civic-run KEM Hospital in Parel is poised to start its heart transplant programme, as its cardiac surgery department has enrolled two patients for heart transplants. The hospital will become the first public hospital in the state to offer this programme by securing the license to conduct heart transplants.
The European cardiopulmonary bypass accessor equipment industry growth is driven by the increasing prevalence of cardiovascular diseases, a growing geriatric population, and ongoing technological advancements in bypass equipment. Major product categories include oxygenators, ECMO machines, pumps, cannula, temperature monitors, and various accessories. The adult patient segment dominates the market which reflects the high incidence of cardiac disease in this demographic.
The U.S. cardiopulmonary bypass (CPB) equipment market is experiencing significant growth which is driven by technological advancements, a rising prevalence of cardiovascular diseases, and an increasing demand for minimally invasive surgical procedures. Innovations such as compact and portable CPB machines, improved oxygenators, and integration with digital monitoring systems have enhanced the safety and efficiency of cardiac surgeries.
India faces a significant burden of cardiovascular diseases, including heart attacks and strokes due to factors like obesity, diabetes, and lifestyle changes. The adoption of minimally invasive surgical (MIS) procedures is increasing, offering benefits such as reduced postoperative pain and faster recovery times.
China is aggressively incentivizing local production to reduce dependence on imports and improve accessibility in secondary cities. Increasing across the country, with a rise in procedures such as CABG (coronary artery bypass grafting) and value replacements. CPB systems are incorporating real-time monitoring, data analytics, and AI for better intraoperative decision-making.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 217.2 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 3.0% | 2033 Value Projection: | USD 267.2 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Medtronic, LivaNova (formerly Sorin Group), Terumo Cardiovascular Systems Corporation, Maquet Holding BV & Co. KG, Braun Melsungen AG, Medica s.p.a., Eurosets, Fresenius Medical Care AG & Co. KGaA, Lepu Medical Technology, Elite Life Care, OriGen Biomedical, Tianjin Medical, Soma Technology, Inc., Braile Biomédica, CardioGenesis Corporation, Century Medical, Inc., Nonin Medical, Inc., Xijian Medical |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: Cardiopulmonary bypass equipment-commonly known as a heart-lung machine-is a medical device used during certain surgeries to temporarily take over the function of the heart and lungs. It diverts blood away from the heart and lungs, oxygenates and removes carbon dioxide from the blood, and then pumps the oxygen-rich blood back into the body, ensuring continuous circulation and oxygen delivery during procedures like open-heart surgery.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients