The Sarcoma Biopsy Market is anticipated to grow at a CAGR of 7.2% with USD 13.8 Bn in 2026 and is expected to reach USD 19.3 Bn in 2033. Rising incidence of sarcoma cases, Growing demand for early and accurate diagnosis and Technological advancements in biopsy techniques.
|
Current Event |
Description and its Impact |
|
Strengthening Medical Device Regulations in India (2024–2026) |
|
|
Uniform Code for Marketing Practices in Medical Devices (UCMPMD) – 2024 |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Disposable Device holds the largest market share of 53.7% in 2026 owing to the Growing Preference for Minimally Invasive Procedures. Healthcare providers are increasingly adopting disposable devices in the sarcoma biopsy market to enhance infection prevention and patient safety, as single-use instruments eliminate cross-contamination risks. Clinicians improve procedural efficiency by using ready-to-use devices that remove the need for sterilization and shorten turnaround time. The growing preference for minimally invasive, needle-based biopsies further drives demand. Additionally, facilities reduce costs by avoiding maintenance and reprocessing, while expanding use in outpatient and diagnostic settings and adhering to hygiene-focused regulations that support wider adoption.
To learn more about this report, Request Free Sample
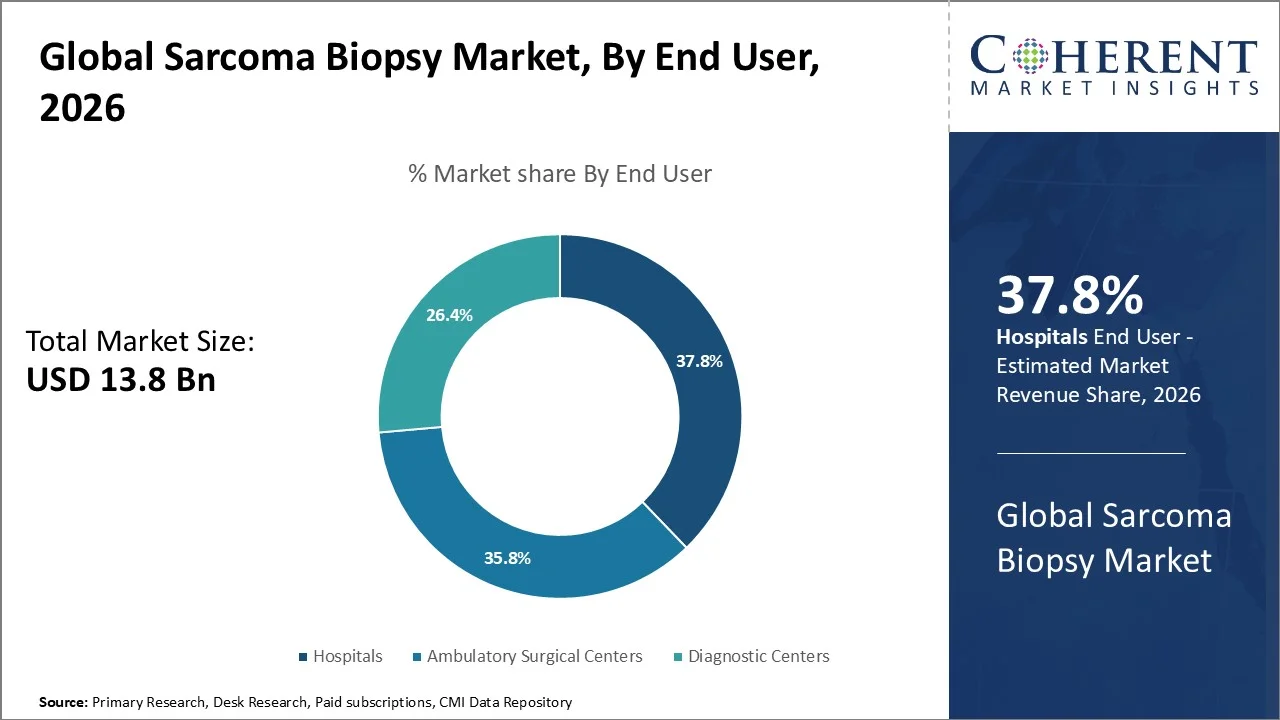
Hospitals acquired the prominent market share of 37.8% in 2026 owing to its availability of advanced diagnostic infrastructure. Hospitals actively drive the sarcoma biopsy market by leading cancer diagnosis and treatment, using advanced imaging systems and in-house pathology services to deliver accurate and timely results. They handle high patient volumes and manage complex cases, which increases biopsy demand. Hospitals continue to invest in minimally invasive and image-guided technologies to improve outcomes and efficiency. They also leverage multidisciplinary oncology teams, secure funding, and participate in clinical research, reinforcing their role as primary centers for sarcoma biopsy procedures.
To learn more about this report, Request Free Sample
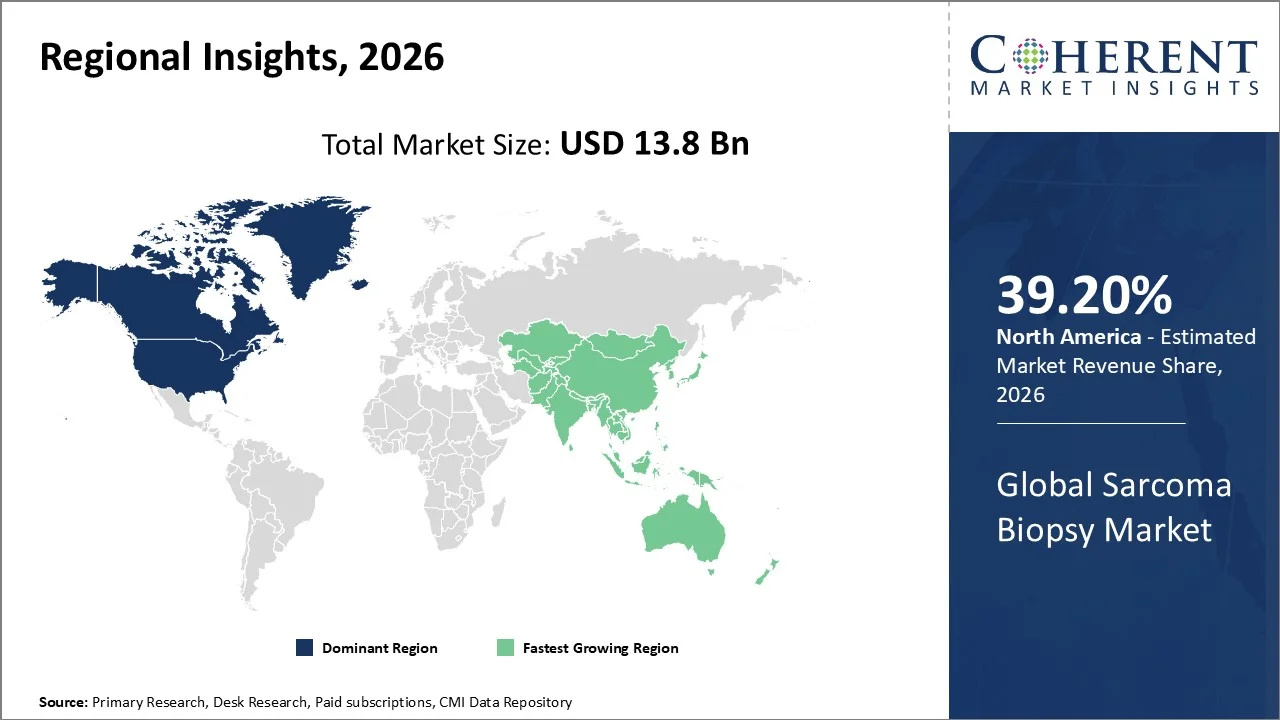
Healthcare providers in North America actively drive the sarcoma biopsy market by leveraging advanced infrastructure, addressing a high cancer burden, and utilizing strong diagnostic capabilities. In 2025, an estimated 2,041,910 new cancer cases were diagnosed in the United States, and 618,120 people died from the disease. They widely adopt image-guided and minimally invasive biopsy techniques to improve accuracy and patient outcomes. Payers and institutions support access through substantial healthcare spending and favorable reimbursement systems. The region further accelerates adoption by investing in research and development, embracing innovative technologies early, and benefiting from the strong presence of leading medical device companies. For instance, in August 2025, Haystack Oncology, a Quest Diagnostics company (NYSE: DGX), has announced a research collaboration with investigators at Mass General Brigham, including Massachusetts Eye and Ear and Massachusetts General Hospital, both globally recognized for expertise in head and neck and advanced cutaneous oncology.
Governments and healthcare providers across Asia Pacific actively drive the sarcoma biopsy market by addressing the expanding healthcare infrastructure, and improving access to diagnostic services. They invest in advanced imaging and minimally invasive biopsy technologies to enhance early detection and treatment outcomes. Increasing healthcare spending, rising awareness of cancer diagnosis, and the growth of medical tourism further support demand. Additionally, a large patient population and ongoing healthcare reforms continue to strengthen the adoption of biopsy procedures across the region.
Healthcare providers in the United States actively drive the sarcoma biopsy market by leveraging advanced healthcare infrastructure, addressing high cancer incidence, and utilizing strong diagnostic capabilities. They widely adopt image-guided and minimally invasive biopsy techniques to improve accuracy and patient outcomes. Strong healthcare spending and supportive reimbursement systems expand access to advanced procedures. The country further strengthens market growth by investing in research and development, adopting innovative technologies early, and supporting leading medical device companies that accelerate biopsy use in oncology care. For instance, in August 2025, Atrium Health Foundation has named Dr. Joshua C. Patt as the inaugural holder of the Paula Takacs Foundation Endowed Chair for Sarcoma Research during a private investiture on August 14. The position is supported by a $2 million endowment funded through donations from The Paula Takacs Foundation for Sarcoma Research to advance sarcoma research at Atrium Health.
Healthcare providers in India actively drive the sarcoma biopsy market by responding to a rising cancer burden, expanding healthcare infrastructure, and improving access to diagnostic services. They increasingly adopt minimally invasive and image-guided biopsy techniques to improve early detection and treatment outcomes. Rising healthcare spending, supportive government initiatives, and growing awareness of cancer diagnosis further increase demand. In addition, a large patient population, the growth of private hospitals, and the expansion of diagnostic centers continue to strengthen biopsy adoption across the country.
Some of the major key players in Sarcoma Biopsy are C. R. Bard, Inc., Becton, Dickinson and Company, Argon Medical Devices, InVivo Therapeutics Corporation, Cook Medical Incorporated, UROMED, and Sterylab
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 13.8 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.2% | 2033 Value Projection: | USD 19.3 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
C. R. Bard, Inc., Becton, Dickinson and Company, Argon Medical Devices, InVivo Therapeutics Corporation, Cook Medical Incorporated, UROMED, and Sterylab |
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients