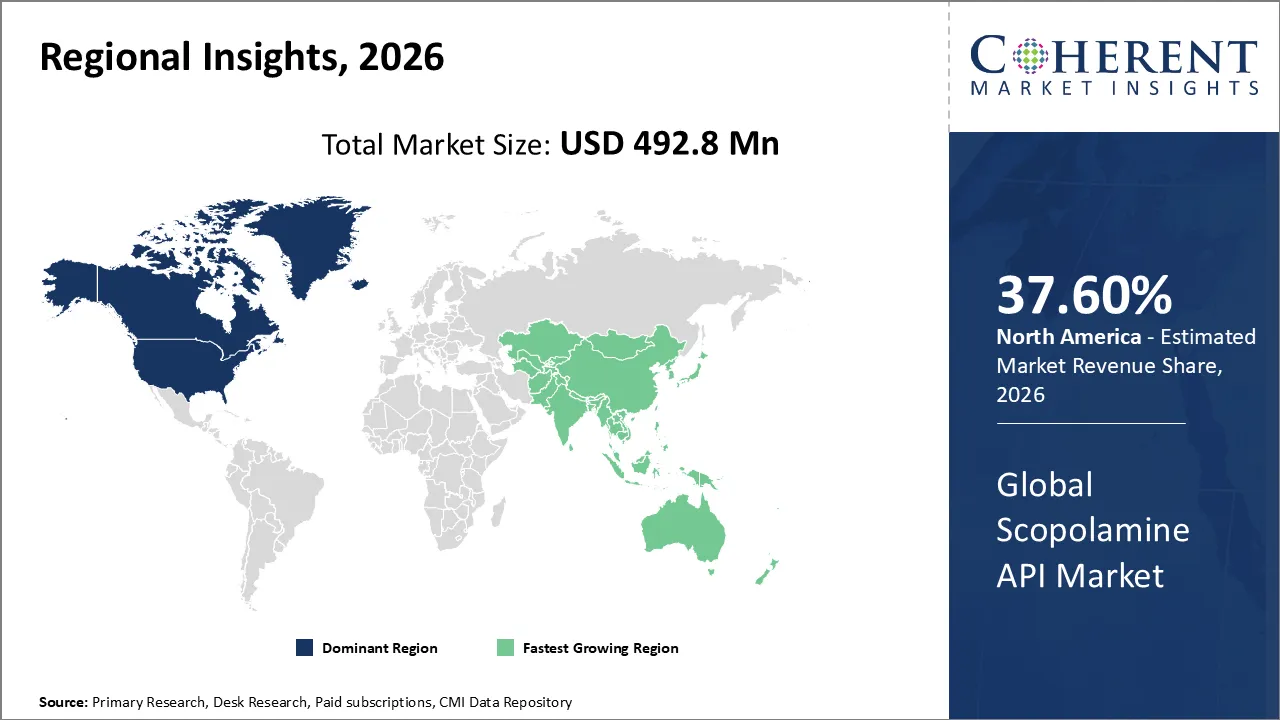
The Scopolamine API Market is estimated to be valued at USD 492.8 Mn in 2026 and is expected to reach USD 741.7 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 6.2% from 2026 to 2033.
Rising demand for antiemetic and anticholinergic therapies drives the Scopolamine API market, particularly for treating motion sickness, postoperative care, and gastrointestinal disorders. Pharmaceutical companies expand manufacturing capacity, while healthcare providers perform more surgical procedures and adopt transdermal drug delivery systems, further supporting growth. Manufacturers prioritize high-purity APIs to meet strict regulatory standards and ensure safety. Advancements in extraction and synthesis technologies enhance production efficiency, and strong demand from developed healthcare systems as well as emerging markets continues to strengthen global supply chains.
|
Current Events |
Description and its impact |
|
Geopolitical and Regulatory Developments |
|
|
Economic and Supply Chain Dynamics |
|
|
Macroeconomic Health and Demand Drivers |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Powder holds the largest market share of 53.8% in 2026. The powder segment drives growth in the Scopolamine API market by offering versatility in pharmaceutical formulation and enabling easy integration into transdermal patches, injectables, and oral drugs. Manufacturers choose powder APIs because they provide greater stability, extended shelf life, and simpler storage and transport. They also ensure precise dosing and maintain consistent quality during large-scale production. Moreover, rising demand for standardized, high-purity inputs and efficient processing methods strengthens the preference for powder-based scopolamine APIs.
≥99% expected to hold largest market share of 52.8% in 2026. Pharmaceutical companies drive the expansion of the ≥99% purity segment in the Scopolamine API market by demanding highly refined compounds that ensure drug safety and consistent therapeutic performance. Manufacturers prioritize the production of ultra-pure APIs to meet strict regulatory requirements and support sensitive formulations such as transdermal systems and injectables. They leverage advanced purification technologies and improved quality control processes to deliver reliable high-purity output. Additionally, the growing focus on precision medicine and tighter impurity limits increases the preference for ≥99% purity scopolamine APIs worldwide.
Industrial Scale acquired the prominent market share of 43.3% in 2026. Manufacturers drive growth in the industrial-scale segment of the Scopolamine API market by meeting rising global demand for bulk pharmaceutical production. They operate large-scale facilities to deliver consistent, high-volume output while maintaining quality and regulatory compliance. Companies implement advanced extraction and synthesis methods to boost efficiency and lower production costs. Expanding surgical procedures, widespread use of antiemetic therapies, and increased outsourcing to contract manufacturers further reinforce industrial-scale operations, enabling the reliable supply of large-quantity scopolamine APIs to pharmaceutical markets worldwide.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 37.60% in 2026. Healthcare infrastructure and high demand for antiemetic and anticholinergic therapies drive strong growth in the North America Scopolamine API market. Hospitals and clinics increase API consumption through widespread surgical procedures and the adoption of transdermal and injectable drug delivery systems. Pharmaceutical companies focus on producing high-purity, standardized APIs to comply with strict FDA regulations and ensure patient safety. Robust research and development, along with a mature manufacturing ecosystem, fosters innovation and reliable supply, strengthening North America’s role as a leading scopolamine API market.
Expanding healthcare infrastructure and rising pharmaceutical manufacturing drive growth in the Asia Pacific Scopolamine API market. Increased awareness of motion sickness and gastrointestinal treatments, along with a rise in surgical procedures, fuels regional API consumption. Manufacturers produce high-purity powder and standardized APIs to meet quality standards for transdermal, injectable, and oral formulations. By investing in advanced extraction and synthesis technologies and leveraging supportive regulatory frameworks, companies enhance production efficiency and supply chains, establishing Asia Pacific as a significant emerging market for scopolamine APIs.
Healthcare providers drive expansion in the United States Scopolamine API market by increasing the use of antiemetic and anticholinergic therapies for motion sickness, postoperative care, and gastrointestinal disorders. Pharmaceutical companies focus on producing high-purity APIs to meet strict FDA regulations and ensure patient safety. Rising adoption of transdermal patches, injectables, and other advanced drug delivery systems further boosts demand. Strong research and development efforts, together with established manufacturing infrastructure, improve production capacity and supply reliability, solidifying the United States as a leading market for scopolamine API use and innovation.
Expanding pharmaceutical manufacturing and increasing healthcare access drive growth in China’s Scopolamine API market. Rising awareness of motion sickness, postoperative nausea, and gastrointestinal disorders boosts regional API consumption. Manufacturers produce high-purity, standardized powder APIs for transdermal, injectable, and oral formulations. By investing in advanced extraction and synthesis technologies and following supportive regulatory measures, companies enhance production efficiency and maintain quality compliance. Strong industrial-scale capabilities and increasing export opportunities establish China as a major contributor to the global scopolamine API market.
The market witnesses increasing consumption of scopolamine APIs due to the rising prevalence of motion sickness, postoperative nausea, and gastrointestinal disorders. Hospitals and clinics are expanding the use of antiemetic drugs in surgical and travel-related treatments. Pharmaceutical manufacturers focus on producing high-purity APIs compatible with transdermal patches, injectables, and oral tablets, ensuring safe and effective therapeutic outcomes for diverse patient populations across global markets.
Increasing global demand for bulk scopolamine APIs creates opportunities for industrial-scale production and partnerships with contract development and manufacturing organizations (CDMOs). Large-scale facilities allow companies to produce consistent, high-quality APIs while reducing costs. Collaborations with pharmaceutical firms for outsourced production enable stable revenue streams and strengthen market presence in both domestic and international markets.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 492.8 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.2% | 2033 Value Projection: | USD 741.7 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Phytex Australia., Aspen Holdings, LGM Pharma, Midas Pharma GmbH, Transo-Pharm Handels-GmbH, Aktin Chemicals,Inc, Merck KgaA, Alchem International Pvt. Ltd., Tiefenbacher API + Ingredients GmbH & Co. KG, Spectrum Chemical, Vital Laboratories Pvt Ltd, Clearsynth, Prism Industries Ltd, Alkaloids Corporation., and Manus Aktteva Biopharma LLP |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients