Self-Care Medical Devices Market is estimated to be valued at USD 28.22 Bn in 2026 and is expected to reach USD 49.32 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 8.3% from 2026 to 2033.
Analysts’ Views on the Global Self-care Medical Devices Market:
The global self-care medical devices market growth can be hindered due to product recalls, thus increasing product launches by key market players are expected to drive the market growth over the forecast period. For instance, in May, 2021, Biobeat, a medical device company, announced the launch of its new wearable and continuous Ambulatory Blood Pressure Monitoring (ABPM) chest-monitoring device. Biobeat's disposable wearable chest-monitor includes an intuitive patient self-set-up, requiring no assistance from a clinician or technician. The device measures diastolic and systolic blood pressure, heart rate, mean arterial pressure, cardiac output, and systemic vascular resistance.
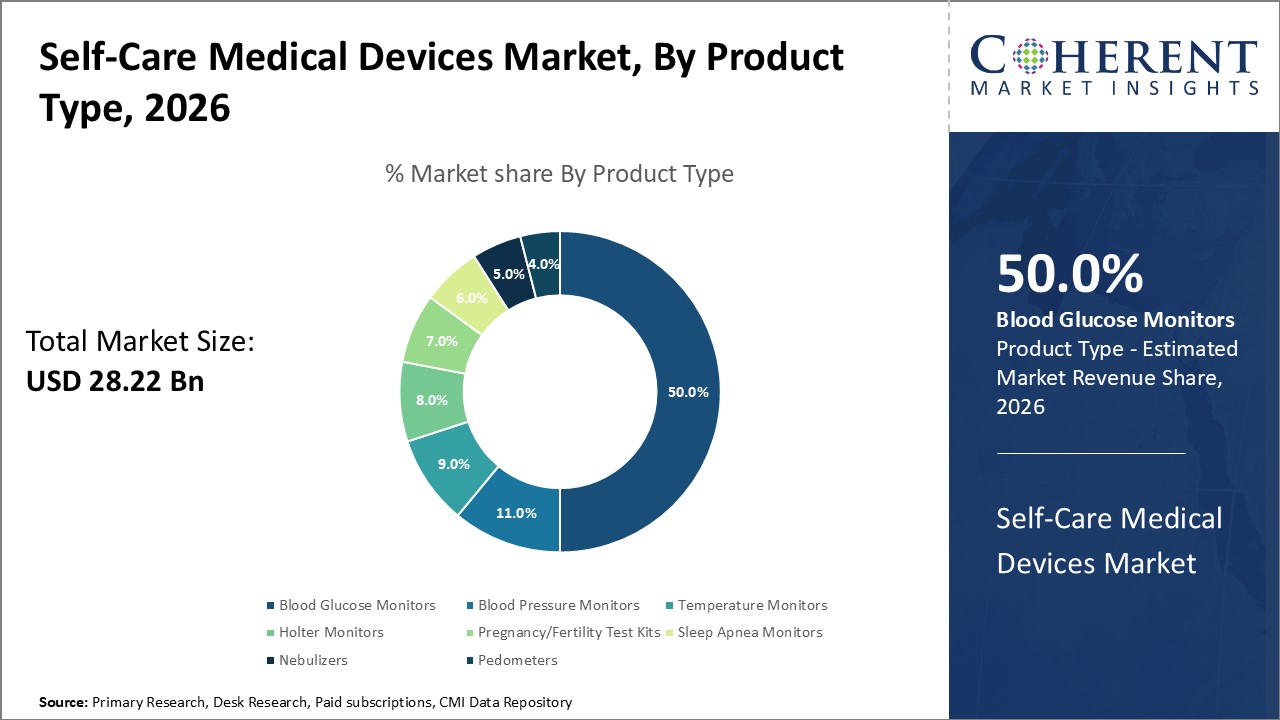
Figure 1. Global Self-care Medical Devices Market Share (%), by Product Type, 2026
To learn more about this report, Request Free Sample
Global Self-care Medical Devices Market - Driver
Increasing product launches by key market players
Increasing product launches by key market players are expected to aid the growth of the global self-care medical devices market over the forecast period. For instance, in February 2022, InBody Pvt. Ltd., a medical technology company, launched InBody BP 170 blood pressure monitor, which provides patients with the option to self-record and track key health metrics at home.
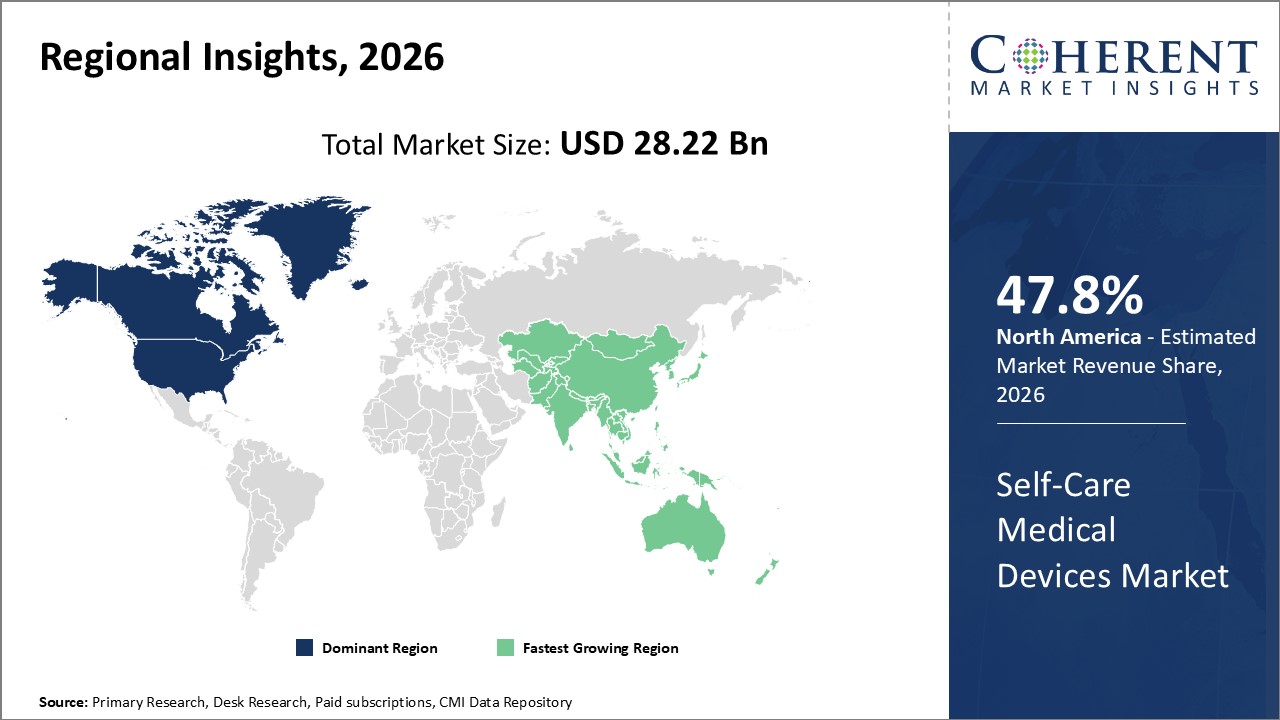
Figure 2. Global Self-care Medical Devices Market Share (%), by Region, 2026
To learn more about this report, Request Free Sample
Global Self-care Medical Devices Market - Regional Analysis
Among regions, North America is estimated to hold a dominant position in the global self-care medical devices market over the forecast period. North America holds 47.8% of the market share due to increasing product launches, which is expected to drive the global self-care medical devices market growth over the forecast period. For instance, on June 6, 2026, Acurable, a U.K.-based medical device company, announced that it will launch its AcuPebble device for obstructive sleep apnea diagnosis in the U.S. The device is designed for remote and efficient home sleep apnea testing that allows earlier detection, faster treatment, and better management of sleep apnea.
For instance, in July 2020, the U.S. In addition, some home-use blood glucose meters have built-in wireless data transmission capabilities, which can facilitate remote patient monitoring. Therefore, the U.S. Food and Drug Administration encourages hospitals and long-term care facilities to consider policies to allow patients to self-test using home-use blood glucose meters, which may include leveraging patients' own home-use blood glucose meters or dispensing a home-use blood glucose meter to patients upon admission to the hospital or to those residing in long-term care facilities.
Self-care Medical Devices Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 28.22 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.3% | 2033 Value Projection: | USD 49.32 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
3M, Abbott, Bayer AG, F. Hoffmann-La Roche Ltd, Inc., GE Healthcare, Johnson & Johnson Services, Inc., Medtronic, OMRON Healthcare, Inc., ResMed, Advin Health Care, Koninklijke Philips N.V., NeuroPace, Inc., Natus Medical Incorporated, and Fisher & Paykel Healthcare Limited |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Self-care Medical Devices Market Segmentation:
The global self-care medical devices market report is segmented into product type, end user, and region.
Based on product type, the market is segmented into blood glucose monitors, blood pressure monitors, temperature monitors, holter monitors, pregnancy/fertility test kits, sleep apnea monitors, nebulizers, and pedometers. Out of which, the blood glucose monitors segment is expected to hold a dominant position in the global self-care medical devices market during the forecast period, and this is attributed to the increasing product launches of blood glucose monitors.
Based on end user, the market is segmented into home care setting and rehabilitation center. Out of which, the rehabilitation center segment is expected to dominate the market over the forecast period, and this is attributed to the rising number of rehabilitation centers in U.S.
Among all segmentation, the product type segment has the highest potential due to the increasing launch of blood glucose monitors by key market players.
Global Self-care Medical Devices Market Cross Sectional Analysis:
In the product type segment, blood glucose monitors hold a dominant position in North America due to increasing collaborations among key market players. For instance, in February 2020, Insulet Corporation, manufacturer of medical devices and Abbott, a medical device company announced that they had collaborated to expand Abbott’s product portfolio. Under this collaboration, Abbott’s market-leading Freestyle Libre continuous glucose monitor will be incorporated into Insulet Corporation’s Omnipod Horizon automated delivery system, comprising a wearable insulin pump and software to glucose control.
Global Self-care Medical Devices Market: Key Developments
Increasing adoption of organic strategies
Increasing adoption of organic strategies, such as the collaborations, by key market players to expand its product portfolios is expected to drive the global self-care medical devices market growth over the forecast period. For instance, in November 2022, Intuity Medical, Inc., a medical technology company, and Embecta Corp., a medical device company, entered into a co-promotional agreement under which Embecta Corp. will be able to promote Intuity Medical Inc.’s innovative POGO Automatic Blood Glucose Monitoring System to healthcare professionals to expand its product portfolio.
Global Self-care Medical Devices Market: Restraint
Increasing product recalls by regulatory authorities.
Increasing product recalls by regulatory authorities are expected to hinder the market growth over the forecast period. For instance, in August 2021, Abbott, a medical technology company, announced that the U.S. Food and Drug Administration (FDA) recalled FreeStyle and FreeStyle Flash blood glucose meters, as they produce low blood sugar results incorrectly when used with FreeStyle or FreeStyle Lite blood glucose test strips.
Global Self-care Medical Devices Market - Key Players
The major players operating in the global self-care medical devices market include 3M, Abbott, Bayer AG, F. Hoffmann-La Roche Ltd, Inc., GE Healthcare, Johnson & Johnson Services, Inc., Medtronic, OMRON Healthcare, Inc., ResMed, Advin Health Care, Koninklijke Philips N.V., NeuroPace, Inc., Natus Medical Incorporated, and Fisher & Paykel Healthcare Limited.
*Definition: Self-care medical devices are designed to monitor and treat regular health conditions such as blood glucose level and others. General medical conditions, such as hypertension, diabetes, fever, and other such conditions, require regular monitoring. These devices support patients to monitor and manage the illness. Self-care medical devices are used for measuring and monitoring various vital parameters. These devices play an important role in monitoring blood sugar level, heart rate, hypertension, and other conditions.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients