Spinal Cord Injury Therapeutics Market is estimated to be valued at USD 8.68 Bn in 2026 and is expected to reach USD 12.71 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 5.6% from 2026 to 2033.
Analysts’ Views on the Global Spinal Cord Injury Therapeutics Market:
Spinal cord injuries are the injuries caused to the 31 pairs of spinal nerves, which carry nerve signals from the brain to various parts of the body and also controls the arm and leg movements. The main cause of spinal cord injuries include trauma, loss of blood supply to spinal cord, and compression in spinal cord due to tumors or infections. Spinal cord injuries can be diagnosed with the help of various techniques such as X-ray, Magnetic Resonance Index (MRI), and Computed Tomography (CT). The increasing prevalence of traumatic spinal cord injuries is expected to drive the market growth over the forecast period. For instance, in 2020, according to the National Center for Spinal Cord Injury Statistical Center, the annual incidence of spinal cord injuries is approximately 54 cases in a million population.
Figure 1. Global Spinal Cord Injury Therapeutics Market Share (%), By Indication, 2026
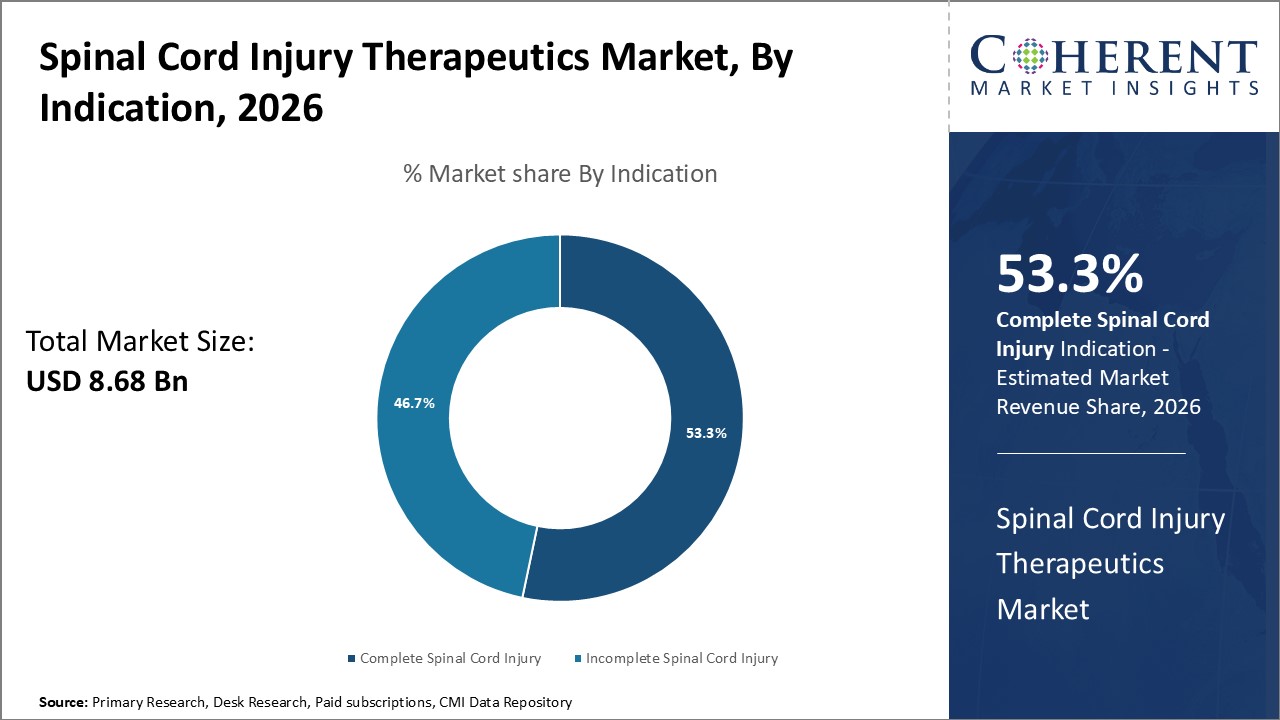
To learn more about this report, Request Free Sample
Global Spinal Cord Injury Therapeutics Market - Drivers
Figure 2. Global Spinal Cord Injury Therapeutics Market Share (%), By Region, 2026
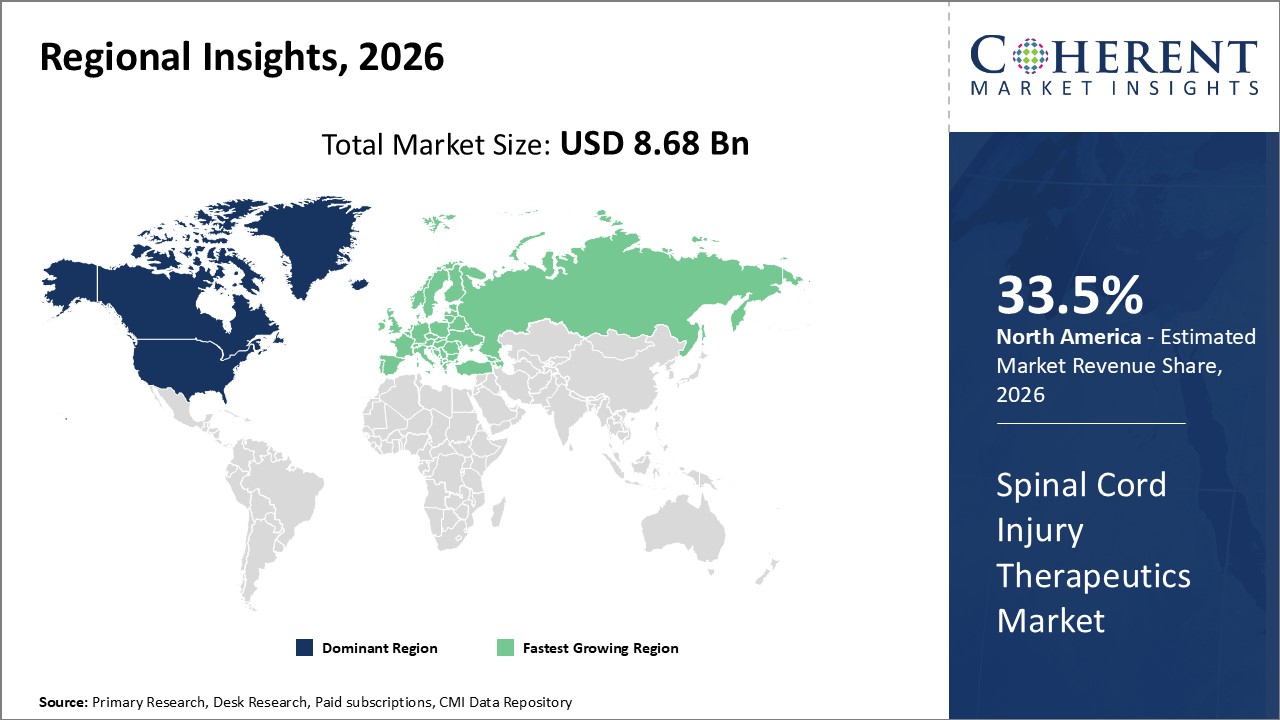
To learn more about this report, Request Free Sample
Global Spinal Cord Injury Therapeutics Market - Regional Analysis
Among regions, North America is estimated to hold a dominant position in the global spinal cord injury therapeutics market over the forecast period. North America is estimated to hold 33.5% of the market share in 2026. The global spinal cord injury therapeutics market is expected to witness significant growth in the coming years, driven by the high prevalence of trauma injury cases, favorable health reimbursement, and increased awareness. The increasing prevalence of neurological disorders is contributing to the growth of the spinal cord injury therapeutics market in the North America region. For instance, according to the American Neurological Association (ANA), in March 2021, neurological diseases affect around 100 billion people in the U.S. This significant number of patients with a neurological disorder is expected to increase the number of neurosurgeries in the U.S., thereby driving the demand for spinal cord injury therapeutics in the U.S.
Spinal Cord Injury Therapeutics Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 8.68 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.6% | 2033 Value Projection: | USD 12.71 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Teva Pharmaceutical Industries Ltd., AbbVie Inc, Pfizer Inc., Reddy's Laboratories Ltd, Zydus Cadila, ReNetX Bio, Inc, InVivo Therapeutics Holdings, Lineage Cell Therapeutics, Inc, Kringle Pharma, Inc., Acorda Therapeutics, Inc., Bioaxone Biosciences, Inc, RespireRx Pharmaceuticals Inc., NervGen, Rising Pharmaceuticals, Inc, ScieGen Pharmaceuticals, Inc, MSN Laboratories Private Limited, and Lannett Co Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Spinal Cord Injury Therapeutics Market Segmentation:
The global spinal cord injury therapeutics market report is segmented based on drug type, indication, application, route of administration, distribution channel, and region.
Global Spinal Cord Injury Therapeutics Market - Cross Sectional Analysis:
Key players are making spinal cord injury therapeutics with advanced technologies in emerging economies, which is also expected to boost the demand for spinal cord injury therapeutics market in the North America region. For instance, on 12 September 2023, Teva Pharmaceuticals, a U.S. affiliate of Teva Pharmaceuticals Industries Ltd., a biopharmaceutical company; announced 16 data presentations across its neuroscience portfoliohappening at Psych Congress on September 6-10 in Nashville. Once-daily AUSTEDO XR, a new formulation of twice-daily AUSTEDO was approved by the U.S. Food and Drug administration (FDA) in February 2023 for adults with chorea associated with Huntington’s disease (HD), and UZEDY, a long-acting formulation of risperidone for subcutaneous use, was approved by the U.S. FDA in April 2023 for adults with schizophrenia. TV-44749 is an investigational formulation of olanzapine currently being studied in adults with schizophrenia.
Global Spinal Cord Injury Therapeutics Market: Key Developments
Global Spinal Cord Injury Therapeutics Market: Key Trends
Introduction of spinal cord injury therapeutics with support of rehabilitation centers
Technological development over the last several decades have created new possibilities for patients with spinal cord injury, offering them various donations and milestones. For instance, in July 2023, Spaudling Rehabilitation, a rehabilitation center recently received a milestone US$ 6 million gift, it is the second largest in Spaulding Rehabilitation’s history from an anonymous donor, which will support Spaulding Rehabilitation’s commitment to improving the lives of patients with Spinal Cord Injury (SCI).
Global Spinal Cord Injury Therapeutics Market: Restraint
Potential side effects of drugs used in the treatment of spinal cord injury
Global Spinal Cord Injury Therapeutics Market - Key Players
The major players operating in the global spinal cord injury therapeutics market include Teva Pharmaceutical Industries Ltd., AbbVie Inc, Pfizer Inc., Reddy's Laboratories Ltd, Zydus Cadila, ReNetX Bio, Inc, InVivo Therapeutics Holdings, Lineage Cell Therapeutics, Inc, Kringle Pharma, Inc., Acorda Therapeutics, Inc., Bioaxone Biosciences, Inc, RespireRx Pharmaceuticals Inc., NervGen, Rising Pharmaceuticals, Inc, ScieGen Pharmaceuticals, Inc, MSN Laboratories Private Limited, and Lannett Co Inc.
Definition: Spinal cord injury treatment involves a combination of medical interventions and rehabilitative therapies. Immediate medical care focuses on stabilizing the spine and preventing further damage. Surgical procedures may be performed to relieve pressure on the spinal cord or repair damaged structures. Rehabilitation aims to improve function, mobility, and quality of life through physical therapy, occupational therapy, and assistive devices. Ongoing research and advancements continue to explore potential therapies such as stem cell transplantation and neural prosthetics.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients