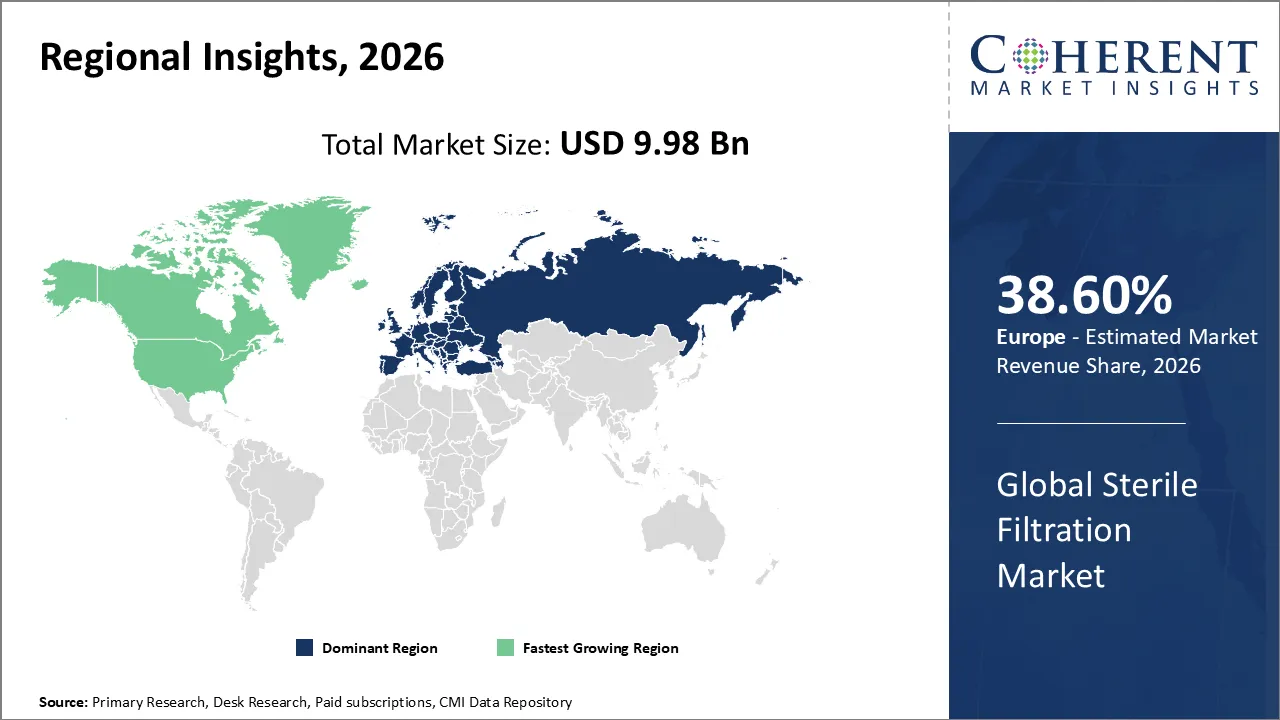
The Sterile Filtration Market is estimated to be valued at USD 9.98 Bn in 2026 and is expected to reach USD 16.25 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 10.24% from 2026 to 2033.
The sterile filtration market ensures product safety and quality across pharmaceutical, biotechnology, healthcare, food and beverage, and laboratory applications. Growing demand for contamination control in biologics, vaccines, injectable drugs, and advanced therapies actively drives the market. Strict regulatory requirements, increased adoption of single-use systems, and ongoing innovation in membrane materials and filtration technologies continue to strengthen market expansion and encourage widespread use in manufacturing and research processes.
|
Current Events |
Description and its impact |
|
Geopolitical and Regulatory Developments |
|
|
Technological Innovations and Industry Advances |
|
|
Regional Infrastructure and Capacity Expansion |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Membrane Filters hold the largest market share of 36.6% in 2026. The sterile filtration industry is powered by membrane filters. They get rid of germs while keeping fragile items safe. Pharmaceutical and biotech businesses utilize them to keep biologics, injectables, and vaccines sterile. Using it often for clarifying, pre-filtration, and final filtering makes the process more efficient and guarantees that the result is always of high quality. Improvements in membrane chemistry, flow performance, and compatibility with single-use items make it easier to employ in regulated manufacturing, laboratory, and quality control environments.
Bioprocess expected to hold the largest market share of 43.4% in 2026. Bioprocessing plays a key role in sterile filtration. Manufacturers follow strict guidelines to prevent contamination in upstream and downstream processes. Biologics, cell cultures, and fermentation products are becoming more complex, requiring reliable filtration to protect purity and yield. Demand grows as single-use systems expand, production volumes increase, and companies adopt flexible manufacturing. Sterile filtration is essential in bioprocess workflows to meet strict quality standards and support continuous process improvement.
For instance, in October 2025, Avantor launched a next-generation sterile sampling platform with PUPSIT assemblies, offering modular, closed-path, and supplier-agnostic setups that streamline bioprocessing and reduce line breaks and operator intervention.
0.5um acquired the prominent market share of 46.6% in 2026. In the sterile filtration sector, manufacturers are the ones who want 0.5 µm filters. These filters keep high flow rates while getting rid of bigger particles and lowering bioburden. Companies utilize them as pre-filters or to clean things up before finer sterile filtration. This keeps membranes downstream safe and makes the process work better. Companies that want reliable performance, operational efficiency, and consistent quality use them. They work in labs to do research, for practical purposes, and with systems that can only be used once.
Pharmaceutical and Biotechnology Companies capture the largest market share of 53.3% in 2026. The sterile filtration sector is mostly made up of pharmaceutical and biotechnology industries. They make sure that vaccinations, biologics, and injectables stay safe and clean. They employ modern filtration to keep everything clean, follow the rules, and keep the quality high. To make manufacturing easier and better, many organizations are using single-use systems and novel membrane technologies. As they grow their business, invest in research and development, and work with contract manufacturers, sterile filtering becomes even more important. For instance, in December 2024, UK-based Amazon Filters launched SupaPore TMB, a high-temperature-resistant membrane for vent applications in pharmaceutical and biotech processes like sterile air venting and large-scale fermentation.
To learn more about this report, Request Free Sample
Europe dominates the overall market with an estimated share of 38.6% in 2026. Europe has rigorous rules for the sterile filtration business. These guidelines make sure that bioprocess and pharmaceutical goods are always safe and of high quality. sophisticated filtration is in high demand because of the growth of biologics, vaccines, and sophisticated medicines. More and more, manufacturers are using single-use technology and automated systems to make things more efficient and less dirty. Research and development, investments in infrastructure, and biotech clusters are what make new things happen. Sustainability programs and partnerships with contract manufacturers also have an impact on market trends and the use of new technologies. For instance, in October 2025, ABN Cleanroom Technology launched CYTONIX, a turnkey rental solution for cytostatics production, ahead of new 2026 pharmaceutical inspection regulations in Belgian hospitals.
A robust pharmaceutical and biotech ecosystem helps North America's sterile filtration business. Growth is driven by better healthcare infrastructure and big investments in research and development. To meet rigorous rules and better prevent contamination, businesses use new filtering technologies, such as single-use and automated systems. The US is the most advanced country in terms of adoption because it makes a lot of biopharmaceuticals and focuses on biologics and vaccines. Canada increases demand in the region by increasing biotech development. For instance, in September 2025, Brainlab received FDA 510(k) clearance and launched Spine Mixed Reality Navigation in the US, combining its precise optical navigation system with advanced mixed reality for enhanced visualization in the sterile field.
The UK sterile filtration market is rising because there is a strong biopharmaceutical base and more money is going into biologics and precision medicines. Single-use, scalable filtering solutions help companies be more flexible, follow the rules, and control pollution. Demand is high since there are strict rules on the safety and quality of products. Research hubs and partnerships with contract manufacturers help filtration technologies move forward and find new uses in pharmaceutical production and lab work.
The US market for sterile filtration is growing because of a focus on making vaccines and biopharmaceuticals. This growth is supported by advanced healthcare infrastructure and a lot of money spent on research and development. Companies use single-use and automated filtration technologies to make sure that their products are safe, satisfy FDA criteria, and work better. There is a lot of demand for biologics, cell, and gene therapies because they are leading the way and have a lot of clinical activity. The market is growing even faster because of the widespread usage of both large-scale and small-batch processing. For instance, in July 2024, Parker Bioscience Filtration, part of Parker Hannifin, introduced a new range of sterilising-grade gas filters to enhance microbial safety and lower operational costs in the food and beverage industry.
The sterile filtration market is increasingly embracing single-use systems to reduce contamination risks and simplify operations. These disposable filters eliminate cleaning and sterilization steps, saving time and lowering cross-contamination potential. They are particularly favored in biologics and vaccine production, as well as in flexible and modular manufacturing setups. End-users benefit from faster batch changeovers, operational efficiency, and regulatory compliance, making single-use filtration a key trend in modern pharmaceutical and biotechnology workflows.
Automation is reshaping sterile filtration processes, with manufacturers adopting systems that include real-time monitoring, integrity testing, and process control. Automated filtration minimizes human intervention, reducing the risk of contamination and operational errors. It enhances reproducibility and efficiency, particularly in high-volume production or complex biologics manufacturing. This trend reflects the broader industry movement toward Industry 4.0, where smart, automated, and validated processes are essential to meet regulatory expectations and accelerate production timelines.
The shift toward single-use and modular manufacturing platforms creates opportunities for sterile filtration providers. End-users seek disposable, ready-to-use filters that reduce cleaning, validation, and contamination risks while improving operational flexibility. Companies can capitalize on this trend by offering versatile, easy-to-integrate filtration systems compatible with single-use bioreactors, modular cleanrooms, and flexible production lines, meeting evolving pharmaceutical and biotech demands while streamlining workflows and enhancing process efficiency.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 9.98 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 10.24% | 2033 Value Projection: | USD 16.25 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
3M, Merck & Co., Inc., General Electric Company, Pall Corporation, Parker Hannifin Corporation, Porvair Filtration Group, Sterlitech Corporation, Sartorius, Starlab Scientific Co., Ltd., Sigma Aldrich Corporation, and Thermo Fisher Scientific. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients