Still’s s Disease Treatment Market is estimated to be valued at USD 2.11 Bn in 2026 and is expected to reach USD 2.97 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 5.0% from 2026 to 2033.
Analysts’ views on the Global Still’s Disease Treatment Market:
Adult-onset Still's disease (AOSD) is a rare inflammatory disorder that has recently been classified as a polygenic autoinflammatory disease. The previous classification based on disease progression seems to be quite outdated. Indeed, there is accumulating evidence that AOSD can be divided into two distinct phenotypes based on cytokine profile, clinical presentation, and outcome, a “systemic” pattern and an “articular” pattern. On May 2, 2023, Mayo Clinic, a U.S.-based hospital, explained the major symptoms of Still’s disease and stated that fever can rise to at least 102 degrees celsius. A fever may occur once or twice a day for a week or longer. The rash may come and go with the fever. The rash usually appears on the body, arms, or legs. Ankles, elbows, hands, and shoulders can also hurt. Joint pain usually lasts for at least two weeks. Muscle pain usually comes and goes with fever. The pain can be severe enough to interfere with daily activities.
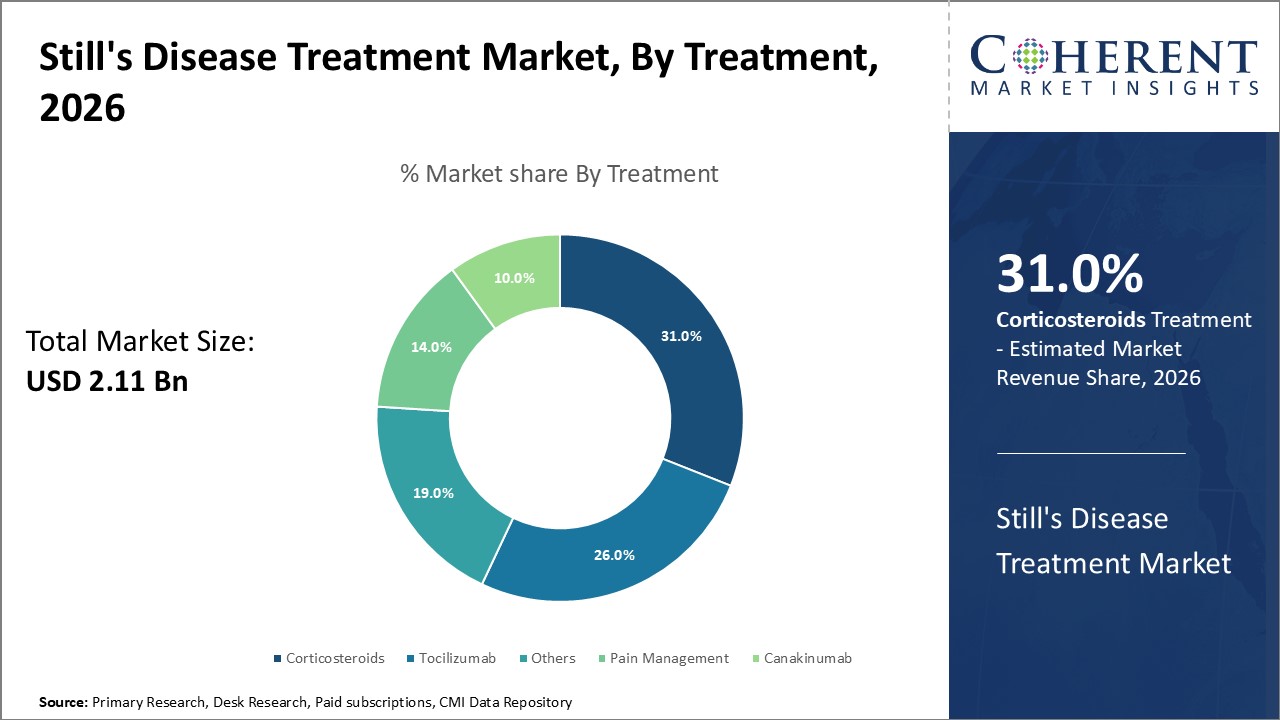
Figure 1. Global Still’s Disease Treatment Market Share (%), By Treatment, 2026
To learn more about this report, Request Free Sample
Global Still’s Disease Treatment Market - Drivers
Increasing awareness about Still’s disease
Increasing awareness campaigns in the field of Still’s disease are expected to drive the global Still’s disease treatment market growth over the forecast period. For instance, on September 7, 2022, AiArthritis, an International foundation for autoimmune and auto inflammatory arthritis, observed Still's Disease Awareness Day which was established by people, patients, and AiArthritis organization. The primary goal of this day is to increase awareness and education about Still's Disease (which includes both systemic Juvenile Idiopathic Arthritis/SJIA and Adult-Onset Still's Disease/AOSD) so an expedite diagnosis, improve disease management, and help patients explain their disease to others.
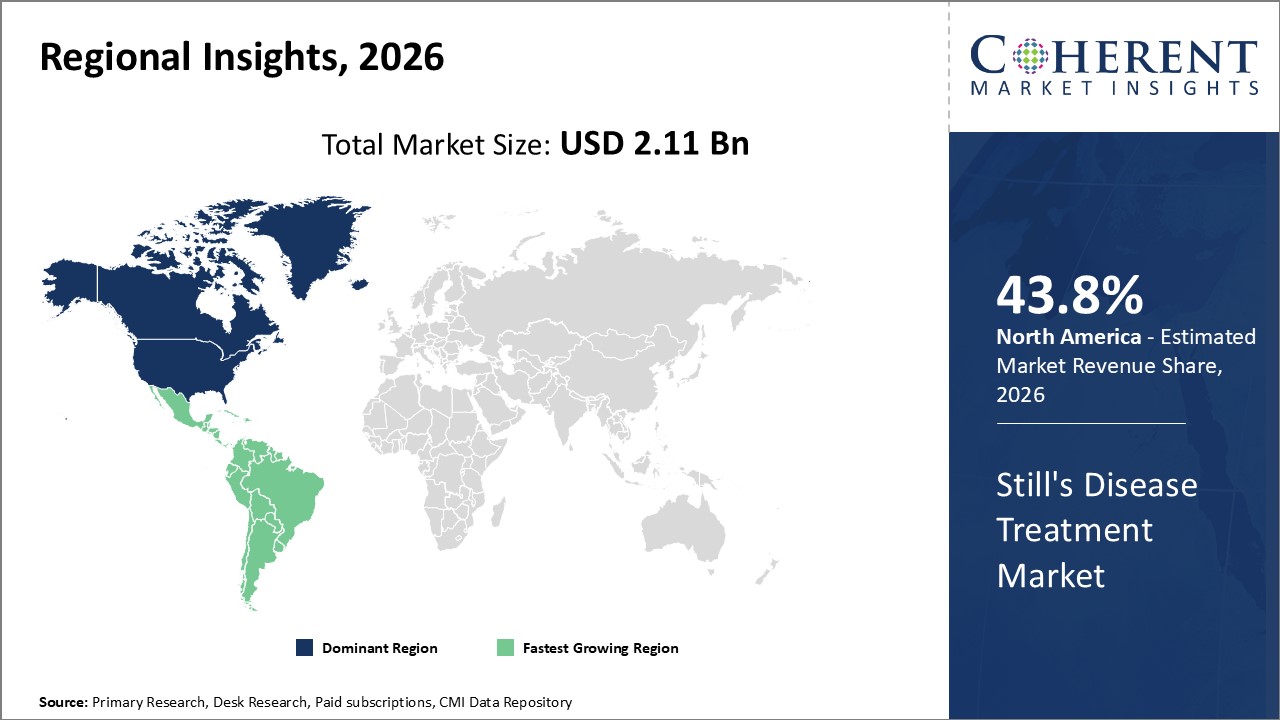
Figure 2. Global Still’s Disease Treatment Market Value (US$ Billion), By Region, 2026
To learn more about this report, Request Free Sample
Global Still’s Disease Treatment Market - Regional Analysis
Among region, North America is estimated to hold a dominant position in the global Still’s disease treatment market over the forecast period, owing to increasing clinical trials of the drugs for the treatment of Still’s diseases. For instance, on May 5, 2021, Avalo Therapeutics, a U.S.-based biotechnology company, announced that it has dosed its first patient in a Phase 1b open-label dose-escalation clinical trial of CERC-007 in patients with Adult Onset Still’s Disease (AOSD). CERC-007 is a high affinity, fully human anti-IL-18 monoclonal antibody (mAb). IL-18 has been demonstrated to have a key role as a marker of disease activity and its concentration correlates with disease severity in AOSD patients. Proof-of-concept of anti-IL-18 therapy has been demonstrated, establishing the clinical utility of this mechanism for patients with AOSD.
Global Still’s Disease Treatment Market
Still’s Disease Treatment Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2.11 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.0% | 2033 Value Projection: | USD 2.97 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
AB2 Bio Ltd , F.Hoffmann-La Roche Ltd, Novartis AG, Swedish Orphan Biovitrum AB, Jubilant Life Sciences Ltd, Vintage Labs, Hikma Pharmaceuticals PLC, Teva Pharmaceutical Industries Ltd, Mylan N.V, Horizon Therapeutics plc, Sun Pharmaceutical Industries Ltd, Zydus Cadila, Amneal Pharmaceuticals LLC, Dr Reddy's Laboratories Ltd, and Fresenius Kabi AG. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Still’s Disease Treatment Market Segmentation:
The global Still’s disease treatment market report is segmented into treatment, route of administration, distribution channels, and region.
Based on Treatment, the global Still’s disease treatment market is segmented into pain management, corticosteroids, tocilizumab, canakinumab, and others. Out of which, the corticosteroids segment is expected to dominate the market due to increasing research and development in the still’s disease treatment drugs.
Based on route of administration, the global Still’s disease treatment market is segmented into injectables and oral. Among these, the oral segment is expected to dominate the market over the forecast period due to ease in administration offered by it.
Based on distribution channels, the global Still’s disease treatment market is segmented retail pharmacies, hospital pharmacies, and online pharmacies. Out of which, the hospitals pharmacies segment is expected to dominate the market over the forecast period due to advanced treatments and immediate access to the patients.
Based on region, the global Still’s disease treatment market is segmented into North America, Latin America, Europe, Asia Pacific, Middle East, and Africa. Among these, North America is expected to dominate the market over forecast period due to increased research and development activities.
Global Still’s Disease Treatment Market: Key Developments
On January 6, 2022, Avalo Therapeutics, Inc., a clinical-stage precision medicine company that discovers, develops, and commercializes targeted therapeutics for patients with significant unmet clinical need in immunology and rare genetic diseases, provided a comprehensive update on the company’s growth opportunities and mid-stage development portfolio. AVTX-007 is being evaluated in a multicenter, Phase 1b study in 12 refractory or steroid-dependent patients with Adult Onset Still’s Disease in two cohorts. Top-line data for both cohorts of the trial were obtained by mid-year 2022.
On December 22, 2020 Cerecor Inc., a biopharmaceutical company focused on development and commercialization of treatments for rare and orphan diseases, announced that its Investigational New Drug Application (IND) to study the use of CERC-007 to treat Still’s disease has been accepted by the U.S. Food and Drug Administration (FDA) and is now open. CERC-007 is a high affinity, fully human anti-IL-18 monoclonal antibody (mAb). The first study will be a global multicenter Phase 1b clinical trial in adult onset Still’s disease and started in the first quarter of 2021. Initial data is expected in the second quarter of 2021.
Global Still’s Disease Treatment Market: Key Trends
Introduction of new research and treatments for Still’s disease
The introduction of new research and treatments for Still’s disease can drive the market growth. On December 10, 2021, Journal of Rare Disease Research and Treatment, U.S. published an article explaining ankylosing spondylitis associated with adult Still's disease as the glycosylated fraction was reduced (10%) in patients. Infectious tests were negative of the patients. These included thick blood drop, blood cultures, throat swabs, urine cultures, viral serology tests (hepatitis B, hepatitis C and HIV), and syphilis. Immunological tests including rheumatoid factor, anti-cyclic citrullinated peptide antibodies, and antinuclear antibodies were also negative. The diagnosis of Adult-onset Still's Disease (AOSD) was thus made in accordance with the criteria of Fautrel9, and with those of Yamaguchi10. Non-steroidal anti-inflammatory drug was replaced by prednisone 1 mg/kg/day. Methotrexate, which was still being used before the onset of Still's disease, was continued. The course of treatment was favorable, with gradual reduction of the corticosteroids after 4 weeks.
Global Still’s Disease Treatment Market: Restraints
Complications associated with Still’s disease
Complications, such as Macrophage Activation Syndrome (MAS), are expected to hamper the global Still’s disease treatment market growth. For instance, according to an article published by MDPI, a Switzerland-based healthcare publisher on October 24, 2022, explained the complications as the most severe complication of adult-onset Still’s disease is MAS. The prevalence ranges between 10% and 15% and is associated with a high mortality rate. Infections or medications, in combination with uncontrolled and prolonged inflammation in patients with a genetic predisposition, may cause this potentially fatal condition. MAS can occur either at the time of diagnosis or later on. Specific predictive or diagnostic factors are lacking for still’s disease. High fever, hepatosplenomegaly, cytopenias, coagulopathy, extreme hyperferritinemia, and hemophagocytosis on bone marrow aspirates are the most common symptoms of MAS.
To counterbalance this restraint, a treatments on side effects and MAS should be introduced.
Global Still’s Disease Treatment Market - Key Players
The major players operating in the global Still’s disease treatment market include AB2 Bio Ltd, F.Hoffmann-La Roche Ltd, Novartis AG, Swedish Orphan Biovitrum AB, Jubilant Life Sciences Ltd, Vintage Labs, Hikma Pharmaceuticals PLC, Teva Pharmaceutical Industries Ltd, Mylan N.V, Horizon Therapeutics plc, Sun Pharmaceutical Industries Ltd, Zydus Cadila, Amneal Pharmaceuticals LLC, Dr Reddy's Laboratories Ltd, and Fresenius Kabi AG.
Global Still’s Disease Treatment Market - Definition
Adult-onset Still's disease is a rare inflammatory arthritis condition. It common symptoms include fever, rash, and joint pain. In some people, the condition may appear as a single episode that goes away. In other people, the condition does not go away or goes away but comes back. Adult Still's disease can affect the joints, especially the wrists. It treatment includes medications that reduce pain and help control the disease. Prednisone is often used when pain relievers such as ibuprofen (Advil, Motrin IB, and others) are not enough.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients