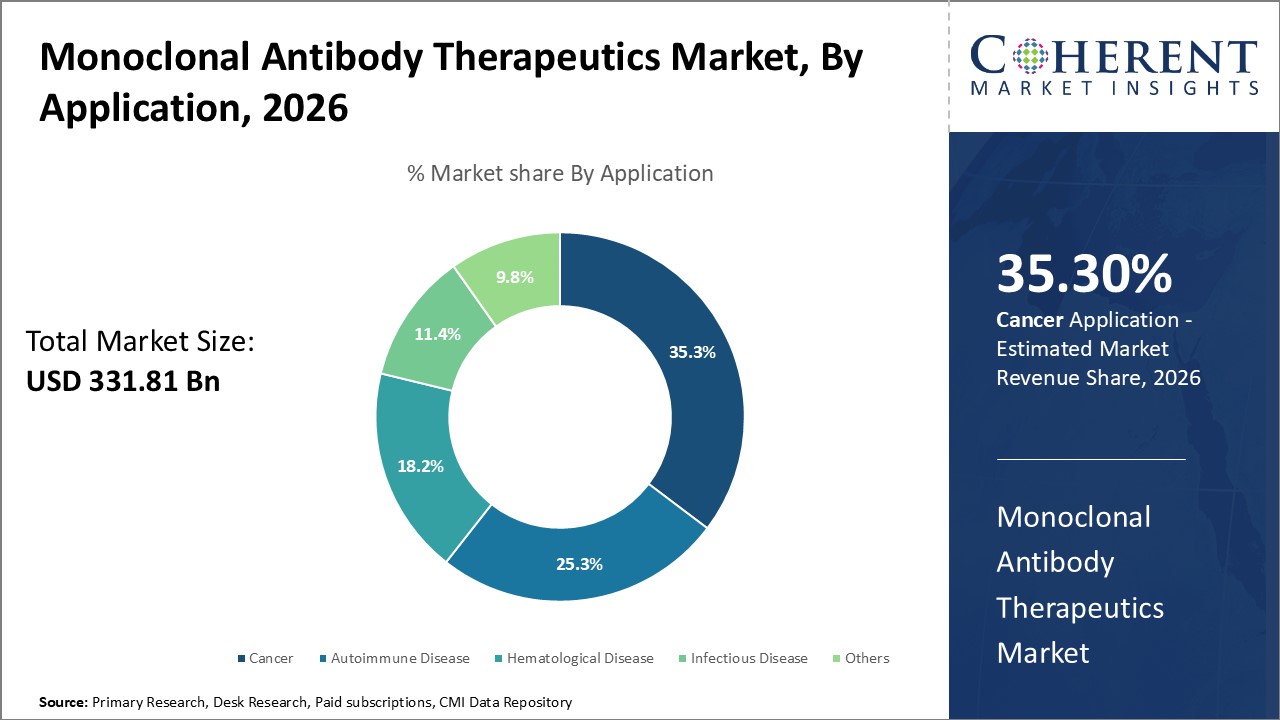
Monoclonal Antibody Therapeutics Market is estimated to be valued at USD 331.81 Bn in 2026 and is expected to reach USD 775.31 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 12.9% from 2026 to 2033.
Advances in biotechnology and rising demand for targeted treatments in diseases like cancer, autoimmune disorders, and infectious diseases are rapidly evolving the Monoclonal Antibody Therapeutics Market. These therapies provide high specificity and improved safety profiles, making healthcare providers prefer them for personalized medicine. Innovations like biosimilars and antibody-drug conjugates are expanding access to and effectiveness of treatments. Strong regulatory frameworks and increased healthcare investments actively support the market’s robust growth and widespread global adoption.
|
Current Events |
Description and its impact |
|
US-China Trade War and Supply Chain Disruptions |
|
|
Biosimilar Market Expansion and Patent Cliffs |
|
|
Geopolitical Shifts in Clinical Trials |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Humanized Segment acquired the prominent share of 50.0% in 2026. Researchers and developers are driving the demand for humanized monoclonal antibodies by focusing on their reduced immunogenicity and enhanced compatibility with the human immune system. Advances in genetic engineering and antibody design now allow scientists to create highly specific therapies for cancer, autoimmune diseases, and infectious conditions. Compared to murine or chimeric antibodies, humanized versions deliver improved safety and effectiveness. The growing focus on personalized medicine and positive clinical outcomes continues to promote their use in research, diagnostics, and treatments.
In April 2025, the NIH officially launched a Phase 2 human challenge trial for AV-1 — an investigational monoclonal antibody designed to reduce dengue virus symptoms when administered both before and after infection.
To learn more about this report, Request Free Sample
The increasing prevalence of cancer is driving growth in the monoclonal antibody therapeutics market, as healthcare providers turn to these targeted treatments for their precision and lower risk of side effects. Researchers are advancing oncology by developing monoclonal antibodies that accurately recognize and destroy cancer cells. These therapies have proven effective in treating cancers such as breast, lung, and blood-related types. Continued innovation and clinical integration are strengthening their role in cancer treatment and advancing the field of immunotherapy. For instance, Swiss drugmaker Roche Pharma unveiled its breast cancer antibody cocktail drug, PHESGO, in India. The new treatment is priced 20% lower and offers greater convenience in administration compared to the current therapy.
Hospital pharmacies play a critical role in the monoclonal antibody therapeutics market by ensuring safe procurement, storage, compounding, dispensing, and administration of these complex biologic therapies. They provide clinical and pharmacological expertise to physicians and nurses, guiding proper dosing, formulation, and drug monitoring. Equipped to handle sterile injectable drugs, hospital pharmacies manage administration of mAbs for serious conditions like cancer and autoimmune diseases, where careful supervision is essential. Their central position in hospital infrastructure makes them the primary distribution channel and a cornerstone for patient access to these advanced treatments. For instance, Onco360®, the nation’s leading independent specialty pharmacy, serve as a pharmacy provider for Elrexfio™ (elranatamab-bcmm). ELREXFIO is a bispecific CD3 T-cell engager that targets B-cell maturation antigen (BCMA) and is approved for treating adult patients with relapsed or refractory multiple myeloma (RRMM) who have undergone at least four prior lines of therapy, including a proteasome inhibitor, an immunomodulatory agent, and an anti-CD38 monoclonal antibody.
To learn more about this report, Request Free Sample
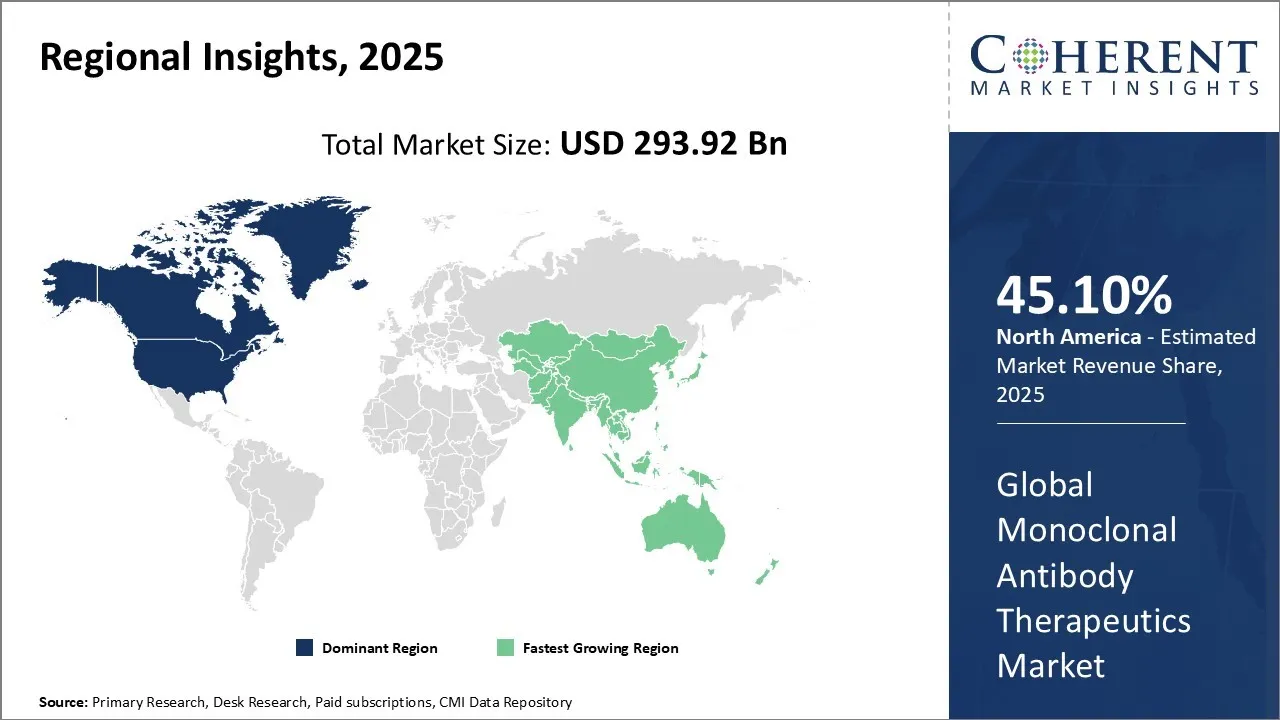
North America acquires the dominant share of 45.10%. Healthcare providers in North America are rapidly expanding the use of monoclonal antibody therapies to treat cancer, autoimmune disorders, and infectious diseases. They are prescribing antibodies targeting RSV and influenza more often in response to increasing infection rates and recent FDA approvals. Hospitals and specialty pharmacies are adopting biosimilars to lower treatment costs and enhance accessibility. Biotech companies and major pharmaceutical firms are actively advancing bispecific antibodies and antibody-drug conjugates to drive progress in precision immunotherapy across the region. For instance, in January 2026, Star Therapeutics, a clinical-stage biotechnology company focused on developing best-in-class antibodies, announced today that the U.S. Food and Drug Administration (FDA) has granted Fast Track designation to VGA039 for von Willebrand disease (VWD). VGA039 is an investigational monoclonal antibody designed to target Protein S and restore blood clotting balance as a universal hemostatic therapy for various bleeding disorders.
Governments in the Asia-Pacific region are actively increasing healthcare R&D funding and building biomanufacturing capacity, particularly in China, India, Singapore, and South Korea. These efforts are driving the growth of monoclonal antibody and ADC pipelines while advancing clinical trial activity. Companies like Biocon, Samsung Bioepis, and Henlius are leading the development and adoption of biosimilars for key antibodies such as rituximab and trastuzumab. Regulatory reforms and enhanced local production are making these therapies more accessible and affordable across the region. For instance, in May 2024, Glenmark, a subsidiary of Glenmark Pharmaceuticals Ltd., has partnered with oncology company BeiGene to market and distribute Tislelizumab and Zanubrutinib in India. Tislelizumab, a novel anti-PD-1 monoclonal antibody, has received approval from the NMPA, EMA, and FDA for treating multiple types of cancer.
China’s National Medical Products Administration has actively streamlined approval processes for monoclonal antibody therapies, significantly cutting drug lag. These reforms have enabled local producers to obtain faster domestic licenses for innovative mAbs, including PD-1/PD-L1 antibodies, allowing them to compete more effectively with global companies. Leading contract development and manufacturing organizations such as WuXi Biologics have expanded bioreactor capacity across several facilities, boosting large-scale production of antibodies and ADCs while strengthening China’s biologics export capabilities. For instance, in July 2024, China developed the monoclonal antibody HANQUYOU as one of its first mAb biosimilars to enter international markets. The EU granted initial approval, and over the past four years, HANQUYOU has demonstrated consistent international quality. It now offers breast and gastric cancer patients around the world access to affordable, high-quality treatment options.
AbbVie and Pfizer are actively developing antibody-drug conjugates (ADCs) and bispecific antibodies to treat cancers like multiple myeloma and non-small cell lung cancer. The FDA has recently approved therapies such as ELREXFIO and TALVEY, showcasing the U.S.’s leadership in oncology innovation. Following patent expirations of major biologics like Humira, companies like Biocon and Sandoz are launching biosimilars, improving patient access and increasing competition in the market. For instance, in October 2024, City Therapeutics, Inc. launched today with a mission to lead the advancement of RNA interference (RNAi)-based medicine. The company envisions RNAi becoming the next major class of high-impact therapies, potentially matching or surpassing the success of monoclonal antibodies.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 331.81 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 12.9% | 2033 Value Projection: | USD 775.31 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pfizer Inc., Novartis AG, Bayer AG, Sanofi, F. Hoffmann-La Roche Ltd, Eli Lilly and Company, Merck KGaA, GSK plc., AbbVie Inc., Bristol-Myers Squibb Company, Regeneron Pharmaceuticals Inc., AstraZeneca, AbCellera Biologics Inc., Molecular Depot LLC, DAIICHI SANKYO COMPANY, LIMITED., Abbott, Biogen, Thermo Fisher Scientific, Inc., Cell Signaling Technology, Inc, Intas Pharmaceuticals Ltd., Avantor Inc., UCB S.A., Merus N.V., GENEXTGENOMICS.COM, Biogenuix |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The market is shifting towards highly targeted monoclonal antibodies that selectively bind specific antigens, improving treatment precision for diseases like cancer, autoimmune disorders, and infectious diseases. This trend enhances therapeutic effectiveness while minimizing damage to healthy tissues. Advances in antibody engineering have enabled development of bispecific antibodies and antibody-drug conjugates (ADCs), which offer dual targeting or combined therapeutic effects, driving monoclonal antibody market demand for more personalized and effective treatment options globally.
There is strong potential in innovating next-generation monoclonal antibodies, such as bispecific antibodies, antibody-drug conjugates (ADCs), and immune checkpoint inhibitors. These advanced therapies offer improved efficacy and the ability to target multiple disease pathways simultaneously. Companies investing in cutting-edge research and development can address unmet clinical needs, especially in oncology and autoimmune diseases, driving differentiated product offerings and competitive advantage. This is further accelerating the monoclonal antibody therapeutics market revenue.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients