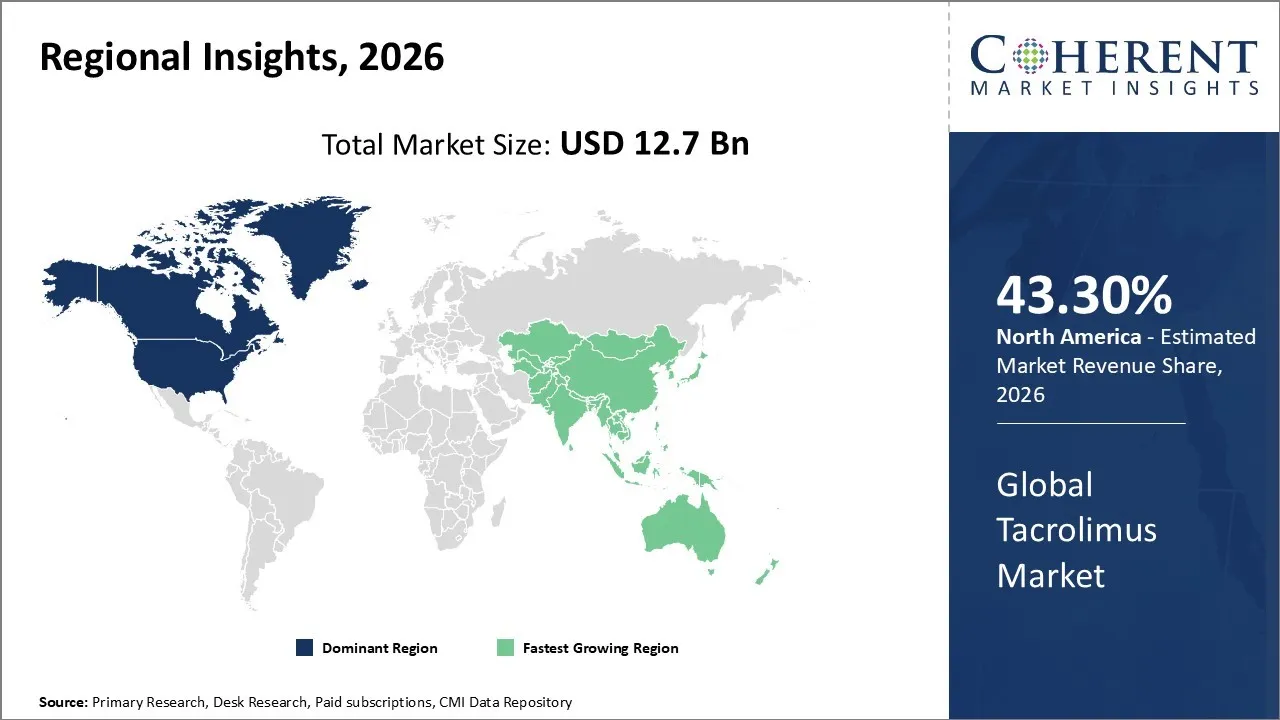
The Tacrolimus Market is estimated to be valued at USD 12.7 Bn in 2026 and is expected to reach USD 16.6 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 7.2% from 2026 to 2033.
The Tacrolimus market grows primarily because healthcare providers rely on it as a first-line immunosuppressant in organ transplantation and increasingly use it to manage autoimmune disorders. Physicians prescribe tacrolimus to prevent graft rejection after kidney, liver, and heart transplants, making it essential for long-term post-transplant care. Rising transplant procedures, expanding healthcare infrastructure, greater awareness of immunosuppressive therapies, and the introduction of generic formulations actively support market expansion.
|
Current Events |
Description and its impact |
|
Macroeconomic and Policy Shifts |
|
|
Technological and Pharmaceutical Innovations |
|
|
Public Health and Demographic Trends |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Immunosuppression expected to hold largest market share of 34.4% in 2026. Clinicians drive the Tacrolimus market by actively using immunosuppressive therapy to regulate immune responses after organ transplantation and in severe immune-mediated conditions. Increasing cases of organ failure, higher transplant success rates, and the ongoing need to protect grafts throughout patients’ lives sustain steady demand for tacrolimus-based treatments. Physicians choose tacrolimus for its proven effectiveness and manageable therapeutic monitoring. Expanding transplant programs, enhanced diagnostic tools, and structured long-term patient management across hospitals and specialty clinics further boost global adoption of tacrolimus. For instance, in October 2025, NATCO Pharma launched 1mg Everolimus tablets, a generic version of Novartis’ Zortress, under the immunosuppressant category.
Tacrolimus Capsules and Tablets hold the largest market share of 42.2% in 2026. The Tacrolimus market for capsules and tablets expands as healthcare providers prioritize their convenience, proven clinical effectiveness, and suitability for long-term outpatient care. Physicians actively choose oral solid formulations for maintenance immunosuppression after organ transplantation because they enable precise dose adjustment and improve patient adherence compared with injectable options. Improving transplant survival rates, increasing availability of generic products, smoother transitions from hospital to home care, and expanding pharmacy networks collectively drive stronger demand for capsules and tablets across global healthcare systems. For instance, Dr. Reddy’s Laboratories launched generic lenalidomide capsules in the US. Lupin received FDA approval for its generic azilsartan medoxomil tablets. Lupin also markets generics such as emtricitabine and tenofovir disoproxil fumarate for HIV, tacrolimus capsules for organ transplant immunosuppression, and empagliflozin for diabetes.
Hospitals acquired the prominent market share of 43.6% in 2026. Hospitals actively drive the Tacrolimus market by performing organ transplants and providing critical care where immunosuppressive therapy is crucial. Medical teams administer tacrolimus during surgeries and in early post-operative care, closely monitoring patients and adjusting doses more effectively than outpatient settings allow. The expansion of specialized transplant programs, multidisciplinary care teams, and hospital procurement policies, along with established treatment protocols, strengthens hospitals’ role as primary drivers of tacrolimus use. Growing hospital infrastructure and higher patient volumes further boost market demand. For instance, Biocon launched generic tacrolimus capsules in the US, following FDA approval. Lupin introduced tacrolimus capsules (0.5 mg, 1 mg, and 5 mg) after its partner Concord Biotech received FDA ANDA approval.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 43.30% in 2026. Healthcare providers in North America actively drive Tacrolimus market trends by adopting the drug due to advanced medical infrastructure, high organ transplant volumes, and established clinical guidelines. Hospitals and specialty centers increasingly administer tacrolimus for maintenance immunosuppression, leveraging effective therapeutic monitoring and physician expertise. Widespread use of generic formulations enhances accessibility and cost-efficiency. Continuous research, innovative drug delivery methods, and the expansion of outpatient transplant care further influence market dynamics, while growing awareness of immunosuppressive therapies among patients and clinicians reinforces tacrolimus as a standard treatment across the United States and Canada. For instance, Veloxis Pharmaceuticals announced that the FDA approved a new de novo indication for Envarsus XR (tacrolimus extended-release tablets) to prevent organ rejection in kidney transplant patients when used with other immunosuppressants.
Healthcare providers in the Asia Pacific actively expand the Tacrolimus market by using the drug for organ transplantation and autoimmune treatments. Governments promote transplant programs, hospitals enhance infrastructure, and patients increase awareness, all driving greater usage. Physicians rely on tacrolimus for its proven effectiveness and manageable post-transplant monitoring. The availability of affordable generic formulations improves access across emerging markets, while growing specialty centers, enhanced training for medical professionals, and broader insurance coverage further strengthen tacrolimus adoption throughout the region.
Hospitals and transplant centers in the United States actively drive the Tacrolimus market by relying on the drug for organ transplants and immune-related conditions. Physicians prescribe tacrolimus widely because of its proven effectiveness, reliable monitoring, and established treatment protocols. The increasing availability of generic versions enhances affordability and patient access, while ongoing clinical research and innovations in drug delivery improve therapeutic outcomes. Expanding outpatient transplant programs, advanced specialty centers, and rising awareness of immunosuppressive therapies among patients and healthcare professionals further boost the drug’s adoption nationwide. For instance, Astellas Pharma Inc. announced that the FDA approved its supplemental New Drug Application (sNDA) for PROGRAF® (tacrolimus) to prevent organ rejection in adult and pediatric lung transplant recipients.
Hospitals and specialty clinics in India actively drive the Tacrolimus market by using the drug for organ transplants and autoimmune disorders. Physicians rely on tacrolimus for its proven effectiveness and manageable post-transplant monitoring. Government initiatives supporting transplant programs, improving hospital infrastructure, and increasing patient awareness further expand its use across the country. The growing availability of affordable generic formulations improves access in both urban and rural areas, while the development of transplant centers, physician training, and wider insurance coverage continue to strengthen tacrolimus adoption nationwide.
Tacrolimus continues to dominate as a first-line immunosuppressant in kidney, liver, heart, and lung transplants. Hospitals and transplant centers increasingly rely on it for post-operative care due to its proven effectiveness in preventing graft rejection. Physicians favor tacrolimus for its predictable therapeutic monitoring, dose flexibility, and long-term safety profile. The trend toward higher transplant volumes, coupled with expanding specialty care programs and outpatient transplant management, reinforces the consistent demand for tacrolimus in global healthcare settings.
The demand for extended-release tablets, controlled-release capsules, and oral bioavailability-enhanced formulations opens opportunities for innovation. Pharmaceutical companies can create products that improve patient adherence, reduce side effects, and simplify post-transplant care. Hospitals and specialty clinics increasingly prefer these formulations for long-term outpatient therapy. By investing in research and development, companies can differentiate their products, enhance therapeutic outcomes, and capture a larger share of the global tacrolimus market.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 12.7 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.2% | 2033 Value Projection: | USD 16.6 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Novartis AG, Mylan N.V., Astellas Pharma Inc., Dr. Reddy's Laboratories Ltd., Pfizer Inc., Abbott, Senju Pharmaceutical Co. Ltd., Glenmark Pharmaceuticals Ltd., Lupin Limited, and Vibcare Pharma Pvt. Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients