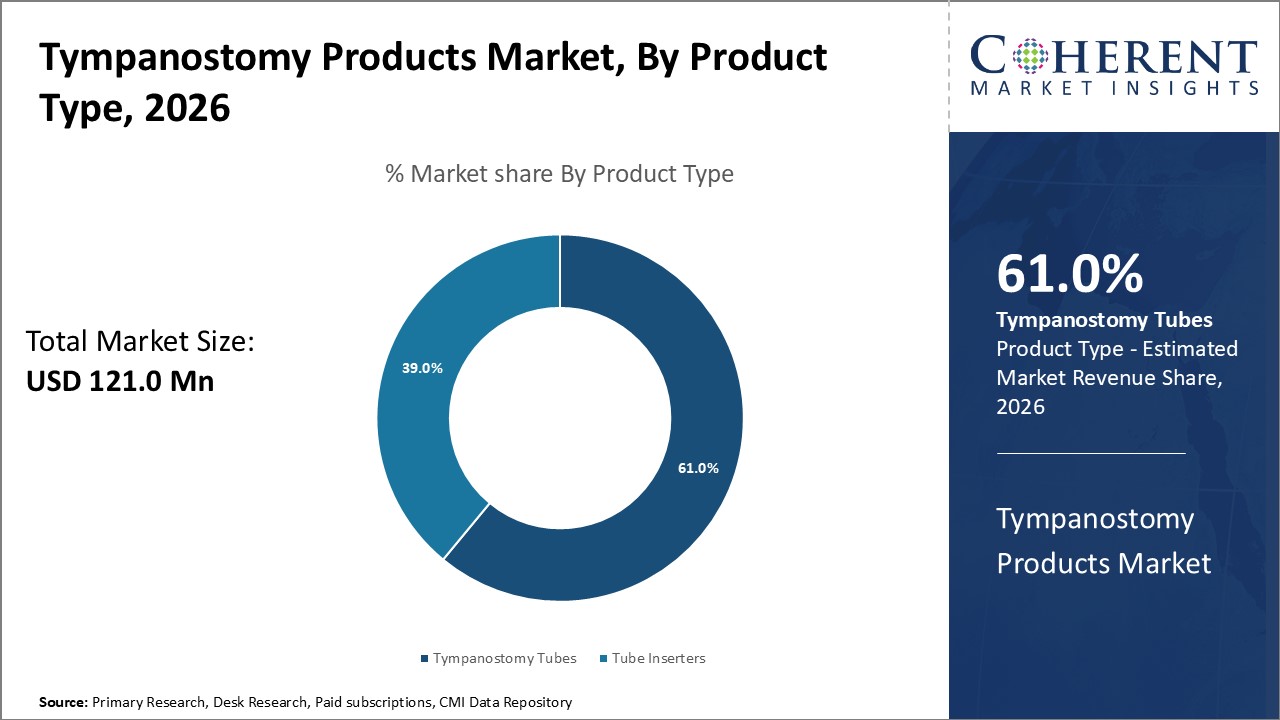
Tympanostomy Products Market is estimated to be valued at USD 121.0 Mn in 2026 and is expected to reach USD 156.1 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 3.7% from 2026 to 2033.
Analysts’ Views on Global Tympanostomy Products Market:
The tympanostomy products are designed to relieve pressure and promote ventilation in the middle ear. The market for tympanostomy products is expected to grow significantly in the coming years due to the increasing prevalence of ear infections and related conditions. In addition, advancements in medical technology have led to the development of more efficient and minimally invasive tympanostomy products, further driving market growth. Furthermore, the rising awareness among healthcare professionals and parents about the benefits of early intervention for ear infections is also contributing to the increasing demand for these products. Moreover, the growing geriatric population, which is more susceptible to ear infections, is expected to fuel the demand for tympanostomy products. Additionally, government initiatives and favorable reimbursement policies are likely to support market growth by making these products more accessible and affordable for patients.
Figure 1. Global Tympanostomy Products Market Share (%), By Product Type, 2026
To learn more about this report, Request Free Sample
Global Tympanostomy Products Market– Drivers
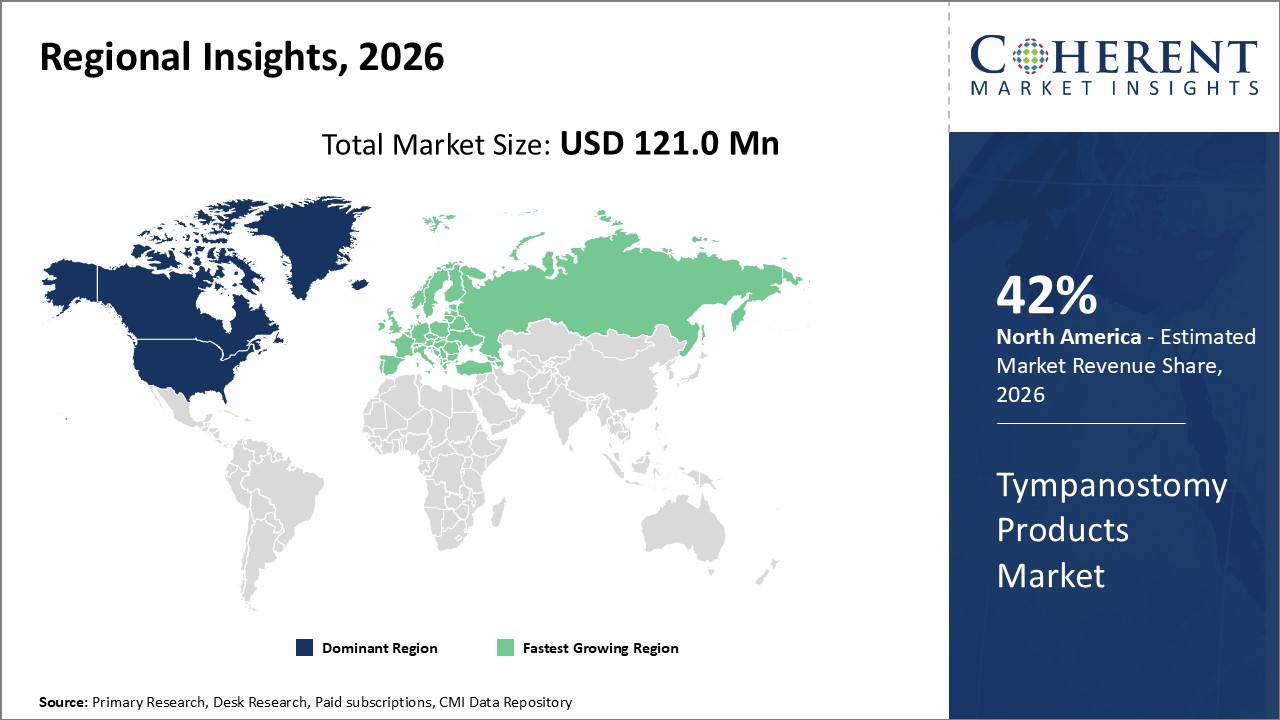
Figure 2. Global Tympanostomy Products Market Share (%), By Region, 2026
To learn more about this report, Request Free Sample
Global Tympanostomy Products Market- Regional Analysis
Among regions, North America is estimated to hold a dominant position in the global tympanostomy products market over the forecast period. North America is estimated to hold 42% of the market share in 2026. The global tympanostomy products market is expected to witness significant growth in the coming years due to the approval of tympanostomy products by the U.S. FDA in the North America region.
Tympanostomy Products Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 121.0 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 3.7% | 2033 Value Projection: | USD 156.1 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Medtronic plc., Olympus Corporation, Medasil Surgical Limited, Preceptis Medical, Acclarent Inc., Summit Medical, Adept Medical, Teleflex Medical Ltd, AdventaMed, and Smith+Nephew . |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Tympanostomy Products Market Segmentation:
The global tympanostomy products market report is segmented into product type, material, application, end user, and region.
Among all the segmentation, the material segment is expected to dominate the market over the forecast period, and this is attributed to the increasing demand for the steel material in surgical instruments.
Global Tympanostomy Products Market- Cross Sectional Analysis:
Tympanostomy products are being made by major firms using cutting-edge technologies. Emerging markets are anticipated to increase demand, which will benefit the North American market for tympanostomy products. The use of tympanostomy products in North America is significantly increasing due to the high prevalence of ear infections and related conditions. This rise in demand for tympanostomy products is also attributed to the growing awareness among healthcare professionals about the benefits of these products in managing ear infections and improving patient outcomes. Additionally, advancements in technology have led to the development of more effective and user-friendly tympanostomy products, further driving their adoption in North America.
Global Tympanostomy Products Market: Key Developments
Global Tympanostomy Products Market: Key Trends
Global Tympanostomy Products Market: Restraint
As a result, businesses in this sector are increasingly concentrating on creating products that are efficient for patients.
Global Tympanostomy Products Market - Key Players
Major players operating in the global tympanostomy products market include Medtronic plc., Olympus Corporation, Medasil Surgical Limited, Preceptis Medical, Acclarent Inc., Summit Medical, Adept Medical, Teleflex Medical Ltd, AdventaMed, and Smith+Nephew.
*Definition: A tympanostomy is a procedure in which a myringotomy (an incision through the tympanic membrane) is made and a tube is placed through the myringotomy to maintain the patency of the defect. The tube allows for the drainage of fluid from the middle ear and helps to equalize pressure, preventing the buildup of fluid and reducing the risk of ear infections. This procedure is commonly performed in children who experience recurrent ear infections or have chronic fluid buildup in the middle ear. Tympanostomy tubes are typically made of silicone or other biocompatible materials and are designed to stay in place for a certain period of time before naturally falling out on their own. In addition to reducing the risk of ear infections, tympanostomies can also improve hearing and speech development in children with chronic fluid buildup.
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients