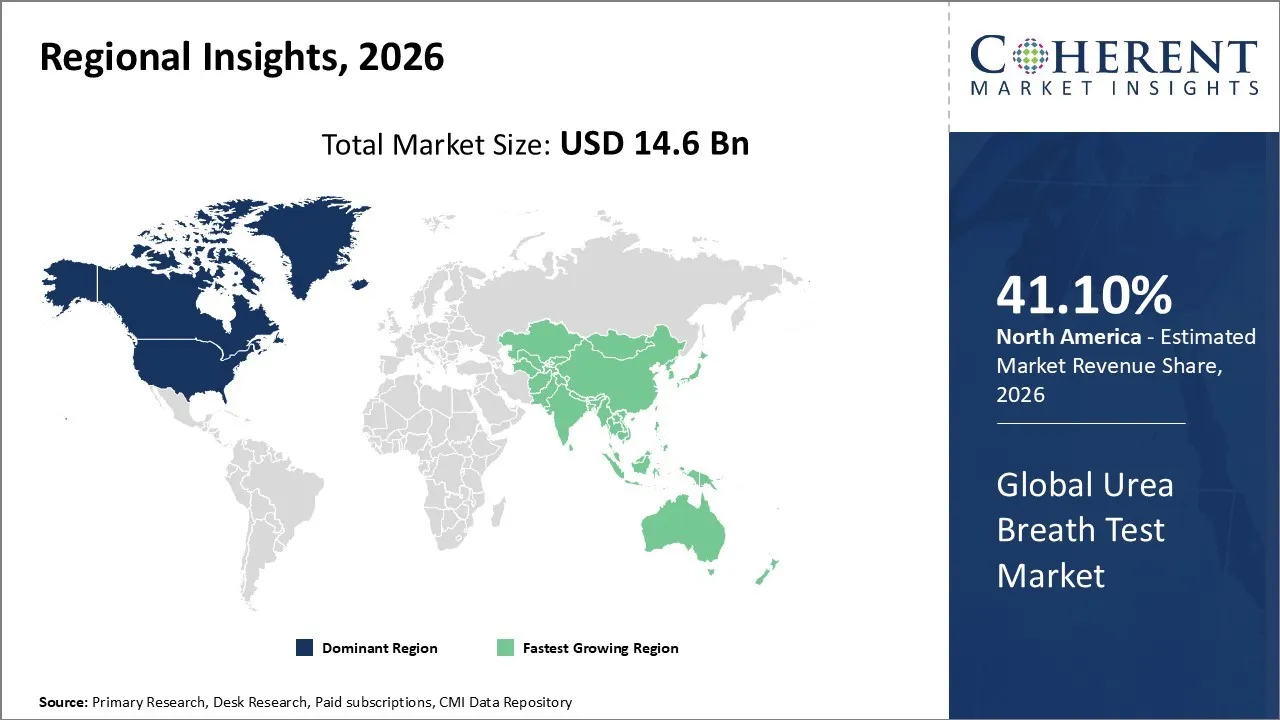
The Urea Breath Test Market is estimated to be valued at USD 14.6 Bn in 2026 and is expected to reach USD 25.8 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 7.3% from 2026 to 2033.
The global Urea Breath Test (UBT) market is growing as more people contract Helicobacter pylori and awareness of gastrointestinal diseases increases. Hospitals, diagnostic laboratories, and clinics actively use UBT for accurate diagnosis and post-treatment monitoring because it offers a non-invasive, patient-friendly option. Companies are improving test accuracy and efficiency through technological advancements such as portable analyzers and mass spectrometry systems. Strong healthcare infrastructure, supportive reimbursement policies, and seamless integration into clinical workflows continue to drive widespread adoption of UBT worldwide.
|
Current Events |
Description and its impact |
|
Geopolitical and Regulatory Developments |
|
|
Economic and Healthcare Infrastructure Trends |
|
|
Competitive and Industry Dynamics |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Breath Analyzer expected to hold largest market share of 51.6% in 2026. The Urea Breath Test (UBT) market is increasingly adopting breath analyzers as healthcare providers seek rapid, non-invasive, and patient-friendly methods to detect Helicobacter pylori. Clinicians actively use these devices for their portability, ease of operation, and ability to deliver reliable results quickly at point-of-care settings. Ongoing technological advancements, such as automated analysis and improved sensor accuracy, boost clinical confidence. Growing awareness of gastrointestinal health and the integration of these devices into hospitals and outpatient clinics further drive their widespread use for effective diagnosis and monitoring. For instance, Meridian Bioscience, Inc., a global provider of diagnostic solutions and life science materials, announced that the FDA has cleared Premier HpSA FLEX to diagnose H. pylori in preserved and unpreserved stools. This product complements Meridian’s H. pylori portfolio, including BreathID urea breath tests, Curian® HpSA, and ImmunoCard® Stat! HpSA.
Laboratory Based Test hold the largest market share of 52.2% in 2026. The demand for laboratory-based Urea Breath Tests (UBT) is rising as healthcare providers seek more precise and reliable Helicobacter pylori diagnostics. Laboratories deliver high-throughput testing with exceptional sensitivity and accuracy, enabling clinicians to confirm infections and monitor treatment outcomes effectively. Expanding laboratory infrastructure, advancing analytical technologies, and integrating UBT into standard diagnostic protocols actively drive adoption. Hospitals and research centers rely on lab-based tests for their reproducibility and detailed quantitative results, reinforcing their central role in comprehensive gastrointestinal care. For instance, in February 2026, Zandra Healthcare has opened a Metabolic Health Centre in India to tackle obesity, diabetes, and related complications. The centre uses an integrated diagnostic approach, offers Cryolipolysis for fat reduction, and performs Urea Breath Tests to detect H. pylori infections.
Mass Spectrometer acquired the prominent market share of 43.3% in 2026. The Urea Breath Test (UBT) market increasingly relies on mass spectrometers as healthcare providers and laboratories seek highly accurate and sensitive detection of Helicobacter pylori. Hospitals and diagnostic labs use these instruments for precise isotope analysis and consistent quantitative results, supporting both diagnosis and treatment monitoring. Advances in automation, faster data processing, and seamless integration into laboratory workflows improve operational efficiency. Research centers and specialized clinics actively adopt mass spectrometry to meet the demand for reliable, reproducible results, cementing its role as a critical tool in gastrointestinal diagnostics. For instance, in June 2025, Thermo Fisher Scientific introduced two advanced mass spectrometers and highlighted its multi-omics strategy at the American Society for Mass Spectrometry (ASMS) conference in Baltimore, Maryland.
Hospitals capture the largest market share of 37.7% in 2026. Hospitals actively drive growth in the Urea Breath Test (UBT) market by adopting non-invasive, patient-friendly diagnostics to detect Helicobacter pylori. They apply UBT to manage large patient volumes efficiently, streamline gastrointestinal diagnostic workflows, and monitor treatment progress. Hospitals leverage the test’s high accuracy, seamless integration into clinical protocols, and support for multidisciplinary care teams. Expanding infrastructure, availability of advanced diagnostic equipment, and a focus on early detection motivate hospitals to establish UBT as a standard tool for comprehensive gastrointestinal care.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 41.10% in 2026. The North America Urea Breath Test (UBT) market is expanding as healthcare providers respond to growing awareness of gastrointestinal health and the increasing prevalence of Helicobacter pylori infections. Hospitals and outpatient clinics actively adopt non-invasive, rapid, and accurate UBT methods, integrating them into routine diagnostic workflows. Companies introduce technological innovations, such as portable analyzers and advanced mass spectrometry systems, to improve testing efficiency and reliability. Strong healthcare infrastructure, supportive reimbursement policies, and adherence to clinical guidelines drive widespread UBT adoption across medical facilities in the region. For instance, Phathom Pharmaceuticals, launched by Takeda and Frazier Healthcare Partners, developed VOQUEZNA, a first-in-class oral treatment combining vonoprazan with antibiotics to treat H. pylori infection in adults.
The Asia Pacific Urea Breath Test (UBT) market is growing rapidly as healthcare providers respond to the high prevalence of Helicobacter pylori infections. Hospitals and diagnostic centers actively adopt non-invasive, accurate, and patient-friendly UBT methods to enhance diagnosis and monitor treatment outcomes. Increasing investments in healthcare infrastructure, modernization of laboratories, and greater awareness of gastrointestinal diseases drive adoption. Companies introduce technological improvements in portable analyzers and automated systems, boosting efficiency and enabling broader implementation across both urban and emerging healthcare markets throughout Asia Pacific.
The United States Urea Breath Test (UBT) market is growing as healthcare providers focus on early and accurate detection of Helicobacter pylori infections. Hospitals, clinics, and diagnostic laboratories actively use non-invasive, patient-friendly UBT methods to improve diagnosis and track treatment outcomes. Companies continually introduce technological innovations, such as portable analyzers and advanced mass spectrometry systems, to boost test speed and reliability. Strong healthcare infrastructure, supportive insurance coverage, and adherence to clinical guidelines drive widespread adoption, making UBT a central diagnostic tool in U.S. gastrointestinal care.
The China Urea Breath Test (UBT) market is growing as hospitals and diagnostic centers actively address the high prevalence of Helicobacter pylori infections. Healthcare providers increasingly use non-invasive, accurate, and patient-friendly UBT methods to improve diagnosis and track treatment outcomes. Expanding investments in healthcare infrastructure, laboratory modernization, and rising public awareness of gastrointestinal health drive adoption. Companies introduce technological advancements, including portable analyzers and automated systems, to boost testing efficiency and enable wider deployment across both urban hospitals and regional healthcare facilities throughout China.
Healthcare providers increasingly favor non-invasive testing methods like UBT over traditional invasive procedures such as endoscopy. Patients benefit from a painless, quick, and convenient diagnostic experience. This trend reflects a broader shift toward patient-centered care, where minimizing discomfort, improving compliance, and reducing procedure-related risks are prioritized. Clinicians also value the efficiency of UBT for screening, diagnosis, and post-treatment monitoring, particularly in outpatient and high-volume hospital settings.
The UBT market is evolving with innovations in breath analyzers and mass spectrometry systems. Portable analyzers, automated sample processing, and enhanced sensor accuracy improve diagnostic speed and reliability. Integration of digital tools and software for data management allows laboratories and clinics to streamline workflows, reduce human error, and provide actionable results faster. Continuous technological development strengthens clinician confidence in UBT and supports wider adoption across hospitals, outpatient centers, and research institutions.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 14.6 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.3% | 2033 Value Projection: | USD 25.8 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Kibion AB (Mayoly Spindler), Avanos Medical, Inc.,Quest Diagnostics, Beijing Richen-Force Science & Technology Co., Ltd., Paladin Labs Inc., Beijing Binal Health Bio- Sci & Tech Co., Ltd., AB ANALITICA s.r.l., Otsuka Holdings Co., Ltd., Sercon Group, Campro Scientific GmbH, Shenzhen Zhonghe Headway Bio-Sci & Tech Co., Ltd., Avisa Diagnostics, INFAI GmbH, Meridian Bioscience, Inc., Metabolic Solutions, Inc., Laboratory Corporation of America Holdings, x, Gulf Coast Scientific, Inc., FAN GmbH, and Kizlon Medical |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In January 2026, AIG Hospitals launched PYtest, India’s first clinically validated breath test for detecting Helicobacter pylori infection, developed by Nobel Laureate Barry Marshall.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients