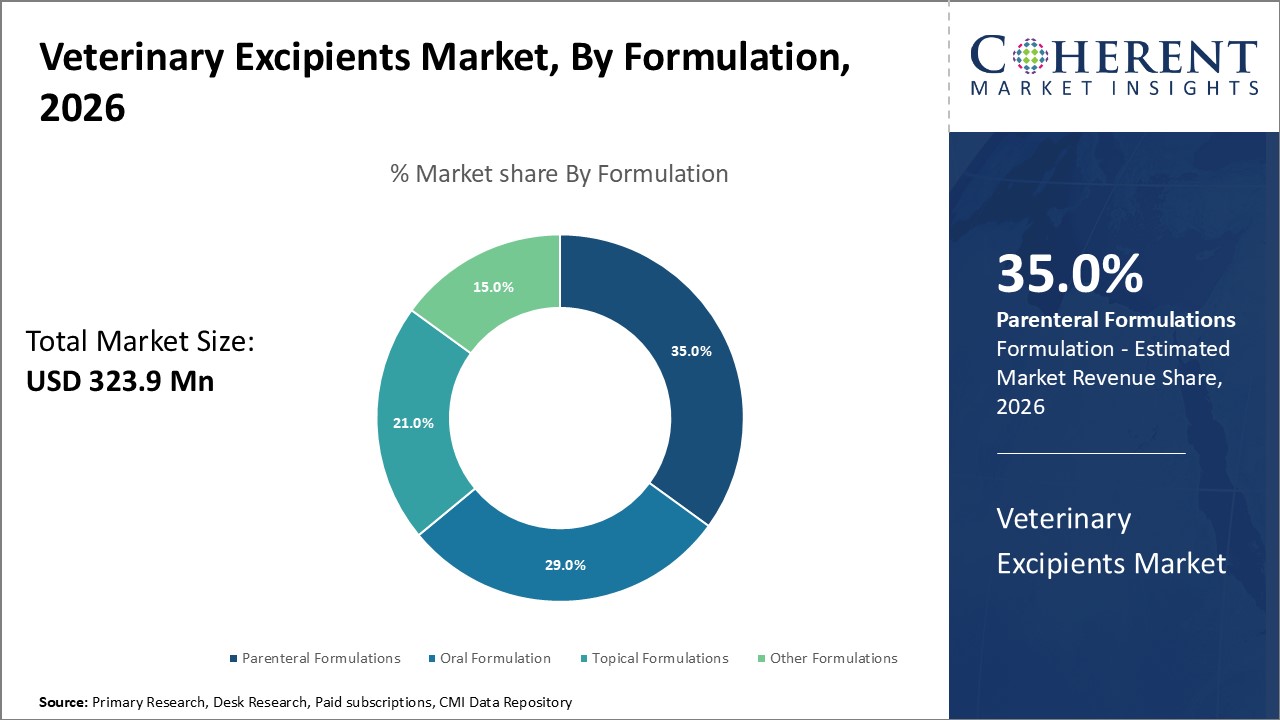
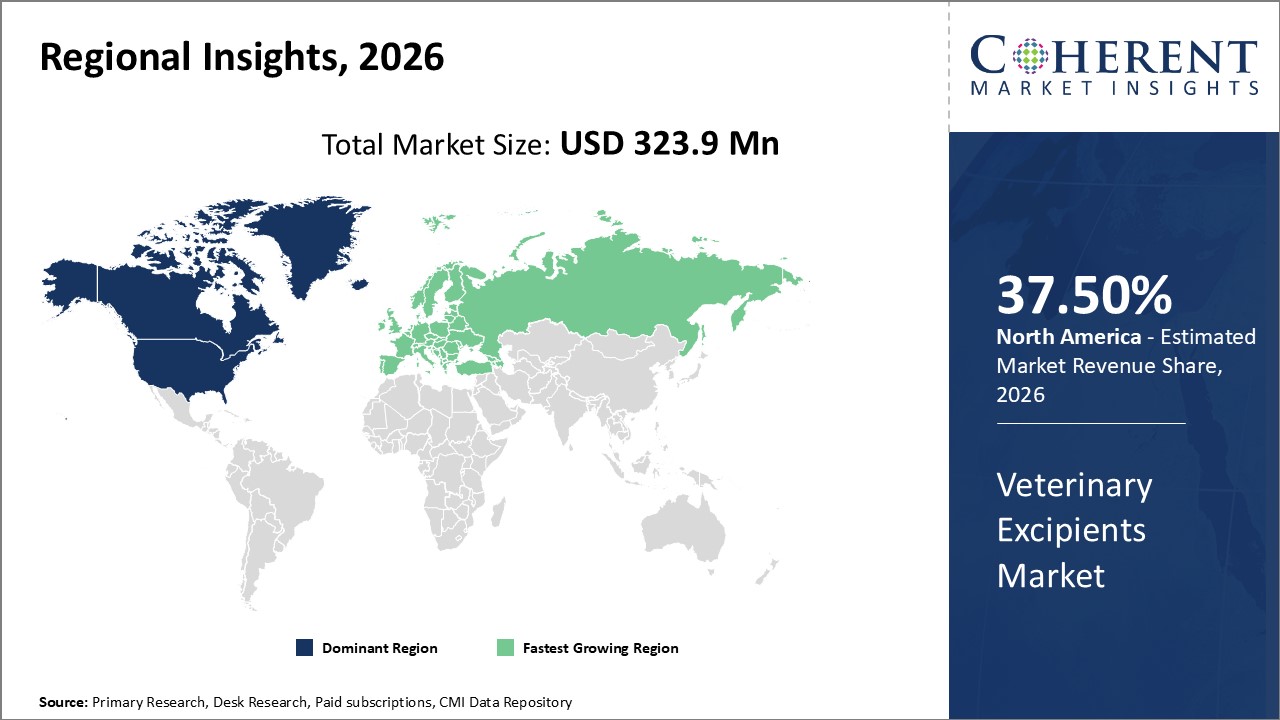
Veterinary Excipients Market is estimated to be valued at USD 323.8 Mn in 2026 and is expected to reach USD 544.3 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 7.7% from 2026 to 2033.
Figure 1. Global Veterinary Excipients Market Share (%), by Formulation, 2026
To learn more about this report, Request Free Sample
Global Veterinary Excipients Market – Key Driver
Increasing demand for animal healthcare products is expected to drive the market over the forecast period.
An increasing in the number of pet ownership along with increasing livestock population is driving the need for better treatment options for animals, which is resulting in increasing demand for animal healthcare products. This is expected to drive the market growth over the forecast period. For instance, according to the 2021-2022 American Pet Products Association (APPA) National Pet Owners Survey, 70% of the U.S. households own a pet, which equates to 90.5 million homes. Furthermore, according to the data published by the American Pet Products Association (APPA), 45.3 million in U.S. households owns cat and 69.0 million households own a pet.
According to the National Agricultural Statistics Service (NASS), January 2022, there were around 91.9 million head of cattle and calves on the U.S. farms as of January 1, 2022. Additionally, according to the National Dairy Development Board, the total livestock population in India in the year 2019 was 535.8 Million.
Therefore, as the livestock population increases, the risk of animal infection increases. This will increase the demand for veterinary excipients for the drug development process.
Veterinary Excipients Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 323.8 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.7% | 2033 Value Projection: | USD 544.3 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
BASF SE, Croda Health Care, JRS Pharma, Lipoid GmbH, Azelis, U.K. Vet Chem, Gattefoss, Synergy API, Anzchem, Vantage. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Figure 2. Global Veterinary Excipients Market Share (%), by Region, 2026
To learn more about this report, Request Free Sample
Increasing incidence of zoonotic diseases is expected to drive the market over the forecast period.
Increasing incidence and prevalence of animal diseases is expected to surge the demand for veterinary excipients in order to manufacture drugs for treatment of animals. This is expected to drive the market growth during the forecast period. Anthrax, caused by soil-borne spore-forming bacteria called Bacillus anthracis, is a zoonotic disease that persists worldwide in livestock and wildlife and infects humans. For instance, according to an article published by the National Center for Biotechnology Information (NCBI), 2021, the pooled prevalence of anthrax globally was 28% (95% confidence interval, 26-30%) from 2,452 samples through for the period from 1992 to 2020. Continent-wise subgroup analysis through the random effects model revealed that the pooled prevalence of anthrax was highest in Africa (29%) and least in North America (21%).
Furthermore, foot-and-mouth disease (FMD) or hoof-and-mouth disease (HMD) is an infectious and sometimes fatal viral disease that affects cloven-hoofed animals, including domestic and wild bovids. According to the data published by Frontiers Media S.A., August 2022, the overall seroprevalence (the level of a pathogen in a population) of foot and mouth disease (FMD) in cattle from January to December 2021, in Ethiopia, Africa, was 26.8%. Also, adult animals were 9.01 times more likely to be seropositive for the disease than young animals.
Furthermore, healthcare providers were facing challenges in terms of additional manpower, equipment, consumables, and other resources which are required to ensure safety in hospitals and provide treatment to patients with other diseases. This has also impacted the overall healthcare market negatively.
Global Veterinary Excipients Market: Key Developments
In May 2022, TheraVet, a company in the management of osteoarticular diseases in pets, announced the signing of an exclusive distribution agreement with Nuzoa, a distributor of veterinary products and services, for the distribution of BIOCERA-VET product line in Spain. BIOCERA-VET contains calcium phosphate excipients which acts as binder and filler in solid oral dosage forms.
In April 2022, Boehringer Ingelheim, a pharmaceutical development and manufacturing company, announced plans to invest US$ 57 million to expand its Animal Health Global Innovation Center in Athens, Georgia.
Global Veterinary Excipients Market: Restraint
The major factors that hinder growth of the global veterinary excipients market include strict regulatory hold over the approval of the veterinary excipients and drugs. The studies designed to evaluate a new drug’s safety and efficacy must meet strict standards known as Good Laboratory Practices and Good Clinical Practices. The U.S. FDA inspects the facilities that run the studies to ensure they are performing at the highest standards. Additionally, studies must be geographically diverse and statistically designed. This helps to ensure a veterinarian, farmer, and pet parent can have confidence a medicine is safe and effective regardless of differences in climate, patient characteristics, or disease variations across the country.
Furthermore, the adverse effects associated with veterinary excipients is expected to hinder the growth of the market over the forecast period. Excipients can cause adverse effects when combined with certain active ingredients or other excipients. For example, modified release excipients, if not properly formulated, can lead to “dose dumping” or overdose, or can result in increased side effects. Antioxidants such as BHA (butylhydroxyanisol) appear to cause hyperplasia and carcinoma of the pre-stomach in rats at a dose of 20g/kg diet, while BHT (butyl hydroxytuolene) contributed to lesions of the hepatic cells after chronic administration to laboratory animals.
Key Players
Major players operating in the global veterinary excipients market include BASF SE, Croda Health Care, JRS Pharma, Lipoid GmbH, Azelis, U.K. Vet Chem, Gattefoss, Synergy API, Anzchem, Vantage.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients