Celiac Disease Drugs Market Size and Forecast – 2026 – 2033
The Celiac Disease Drugs Market is forecast to grow from roughly about USD 1.06 Bn in 2026, expanding at a CAGR of 14.9 % through 2033, and is expected to reach around USD 3.7 Bn by 2033.
Global Celiac Disease Drugs Market Overview
Celiac disease drugs are treatments designed to manage or alleviate symptoms of celiac disease, an autoimmune disorder triggered by gluten consumption. Currently, the primary approach is a strict gluten-free diet, but pharmaceutical innovations aim to supplement or replace dietary restrictions. Drug development focuses on enzyme therapies to break down gluten, intestinal permeability modulators to prevent immune reactions, and vaccines to induce immune tolerance. These therapies aim to reduce inflammation, intestinal damage, and associated complications like malabsorption or anemia. With rising celiac prevalence globally, the market is expanding, attracting research and investment into effective, patient-friendly pharmacological solutions.
Key Takeaways
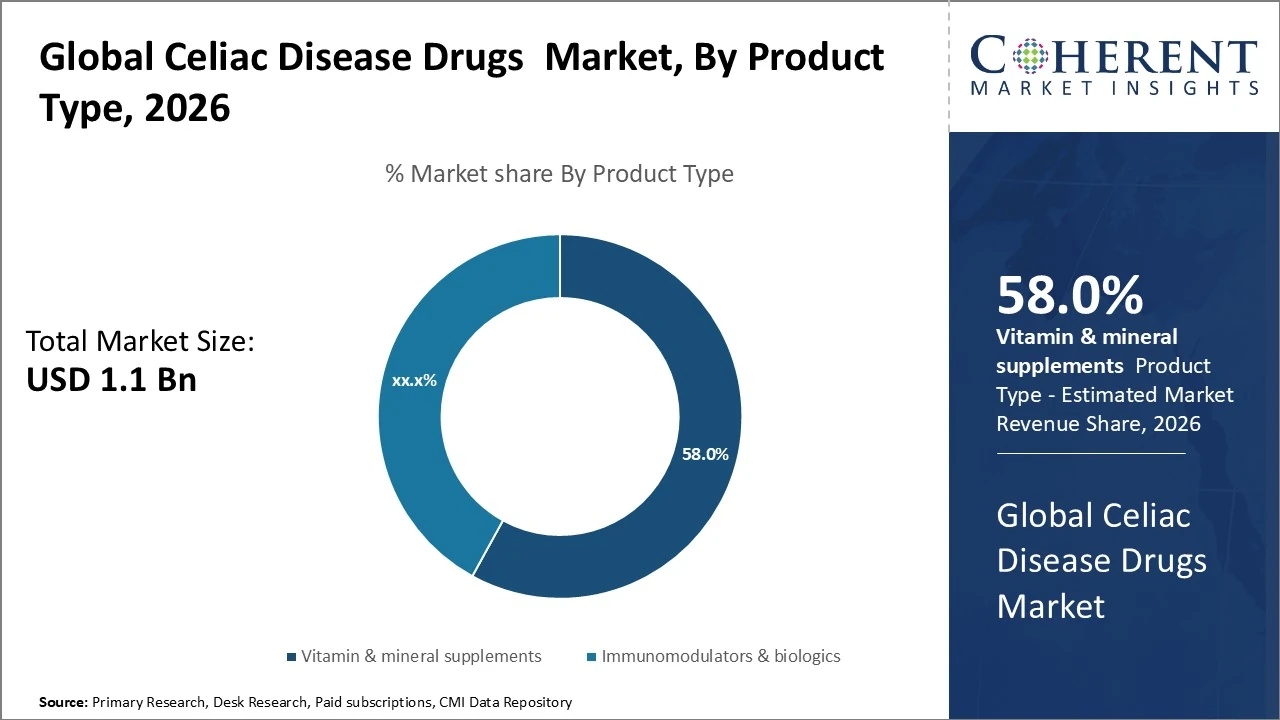
In the global celiac disease drugs market by Product type, Vitamin & mineral supplements currently dominate with about 58%.
In the global celiac disease drugs market, Hospitals and clinics are the dominant end‑user, holding about 50 % of the market.
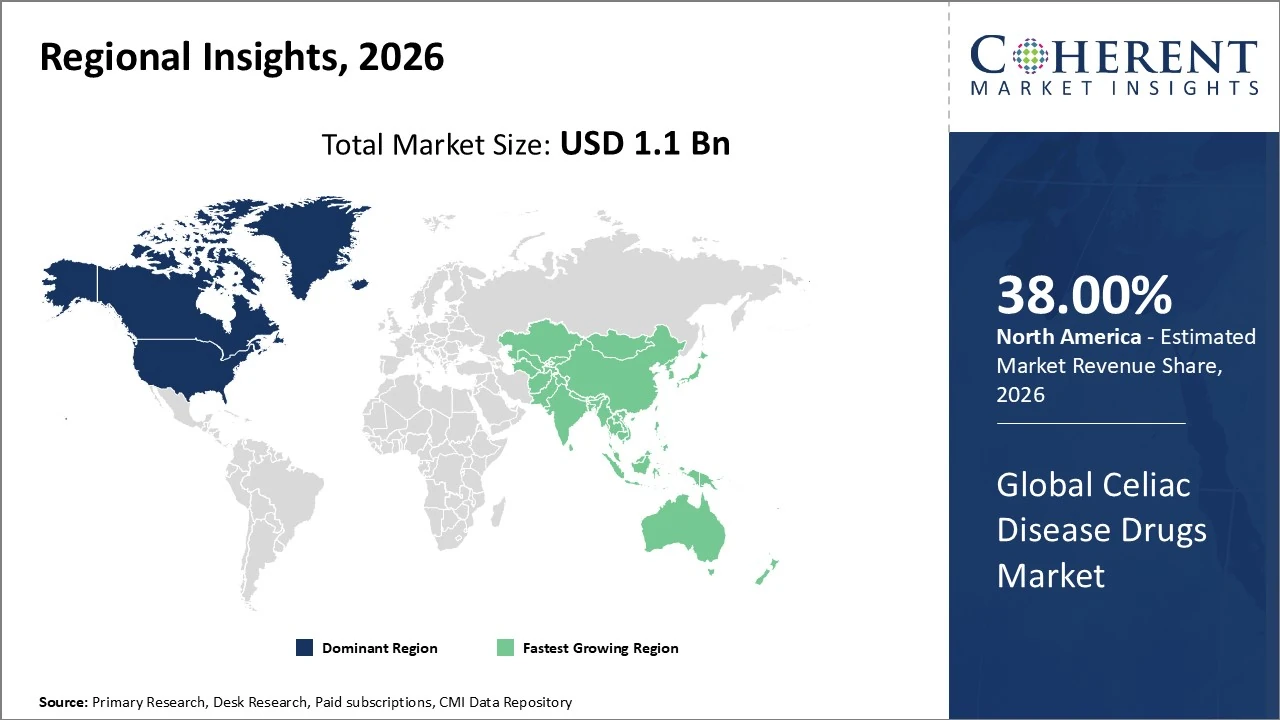
North America dominates the global celiac disease drugs market, holding around 38% share in 2026.
The Asia Pacific celiac disease drugs market is expanding rapidly, holding about 20 % of the global market share.
The Germany celiac disease drugs/treatment market is growing steadily, with revenues around USD 38.4 million in 2026
Celiac Disease Drugs Market Segmentation Analysis
To learn more about this report, Request Free Sample
Celiac Disease Drugs Market Insights, By Product Type
In the global celiac disease drugs market, Vitamin & mineral supplements currently dominate with about 58% due to their routine use for nutrient deficiencies. Enzyme‑based therapies are emerging and growing fast but hold a smaller current base share. Immunomodulators & biologics represent the largest active pharmaceutical segment with roughly 42 % share in drug‑focused reports, reflecting investment in immune‑targeted treatments. Other drug classes like corticosteroids and immunosuppressants form the remainder of the market but with lower individual shares vs biologics and supplements.
Celiac Disease Drugs Market Insights, By End-User
In the global celiac disease drugs market, Hospitals and clinics are the dominant end‑user, holding about 50 % of the market in 2026 because they provide diagnosis, treatment initiation, and access to advanced therapies like enzyme drugs and biologics. Specialty clinics are growing quickly as more focused care improves adherence and outcomes. Home healthcare settings are also rising with telemedicine and remote diet management support. Retail pharmacies and other outpatient providers contribute smaller shares but support ongoing patient access to oral medications and supplements outside hospital environments.
Celiac Disease Drugs Market Trends
While gluten-free diets remain standard, enzyme therapies, biologics, and immune-modulating drugs are gaining traction, offering more effective symptom management and disease modification.
Pharmaceutical R&D focuses on biologics, vaccines, and intestinal barrier modulators, targeting underlying autoimmune mechanisms rather than just symptom control.
Increased awareness, improved diagnostic methods, and higher prevalence detection are expanding the patient base, driving demand for both prescription drugs and adjunctive supplements.
Celiac Disease Drugs Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Celiac Disease Drugs Market Analysis and Trends
North America dominates the global celiac disease drugs market, holding around 38% share in 2026, driven by advanced healthcare infrastructure, high awareness, and strong pharmaceutical presence. The U.S. accounts for most regional share, supported by high diagnosis rates and rapid adoption of innovative therapies like enzyme and biologic candidates in clinical pipelines. Canada and Mexico contribute smaller portions of the regional market but are growing steadily with increasing access to diagnostics and treatments. Growth is propelled by rising celiac prevalence, robust R&D investment, and supportive reimbursement frameworks, while early diagnosis and patient education further expand drug demand.
Asia Pacific Celiac Disease Drugs Market Analysis and Trends
The Asia Pacific celiac disease drugs market is expanding rapidly, holding about 20 % of the global market share in 2026 as diagnostic awareness and healthcare access improve in China, India, Japan, and Southeast Asia. APAC is one of the fastest‑growing regions, with CAGRs around 12 % forecast through 2033, driven by rising disease awareness, increasing healthcare expenditure, and growing demand for therapeutic options beyond gluten‑free diets. Urbanization and expanding middle‑class populations further fuel drug uptake, while ongoing clinical trials and local biotech involvement boost regional innovation and treatment availability.
Celiac Disease Drugs Market Outlook for Key Countries
United States Celiac Disease Drugs Market Analysis and Trends
The USA celiac disease drugs market is growing steadily as diagnosis rates climb and unmet therapeutic needs drive demand for treatments beyond the gluten-free diet. The U.S. market is forecast to expand at an ~11–12% CAGR through 2030, with revenues rising significantly as enzyme-based therapies, vitamins/minerals, and novel pharmacological options gain traction alongside dietary management. Market growth is supported by advanced healthcare infrastructure, strong clinical research activity, and increasing patient awareness. Emerging therapies targeting immune modulation and gluten degradation are fueling innovation, making the U.S. the largest contributor to the North American celiac drugs landscape.
Germany Celiac Disease Drugs Market Trends
Germany’s celiac disease drugs market is poised for steady growth, reflecting broader European trends toward pharmaceutical management beyond gluten-free diets. The growth is driven by rising diagnosis rates, increasing patient awareness, and strong healthcare infrastructure that supports adoption of advanced therapies and supportive treatments like vitamins, minerals, and gluten-degrading enzymes. Germany’s robust reimbursement policies and clinical research activity further fuel uptake of both established and emerging therapies. Enhanced integration of pharmacological options with patient care, and growing demand for evidence-based, convenient treatments, underpin this positive trend.
Analyst Opinion
Therapeutic development is accelerating, with enzyme therapies, immune‑modulating biologics, and barrier modulators gaining clinical momentum, signaling a shift beyond gluten‑free diet reliance.
Persistent symptoms despite dietary adherence and underdiagnosis fuel market growth and pharmaceutical interest.
Complex endpoints, long studies, and recruitment hurdles increase development risk and time to approval.
Most candidates are designed to complement gluten‑free diets, serving as first‑line adjuncts rather than standalone cures.
Personalized medicine, refractory disease solutions, and biotech collaborations offer strategic growth avenues.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1.06 Billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 14.9% | 2033 Value Projection: | USD 3.7 Billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Takeda Pharmaceutical Company Limited, ImmunogenX, Cour Pharmaceuticals, ImmusanT, Teva Pharmaceutical Industries Ltd., Pfizer Inc. | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Celiac Disease Drugs Market Growth Factors
The celiac disease drugs market is expanding due to rising disease prevalence, increasing awareness, and improved diagnostic capabilities globally. The limitations of a strict gluten-free diet create demand for pharmacological therapies, including enzyme treatments and immune-modulating biologics. Advancements in R&D and clinical trials are accelerating the development of innovative therapies targeting autoimmune responses and intestinal barrier repair. Government initiatives and favorable reimbursement policies in developed regions support patient access. Additionally, growing urbanization, lifestyle changes, and higher healthcare expenditure in emerging markets are driving adoption. The market is further fueled by increasing patient preference for convenient, effective treatment options beyond dietary management.
Celiac Disease Drugs Market Development
In May 2025, the FDA granted fast track designation to Teva’s investigational anti-IL-15 antibody TEV-53408 for treating celiac disease in patients on a gluten-free diet, according to a Teva news release.
Key Players
Leading Companies of the Market
Takeda Pharmaceutical Company Limited
ImmunogenX
Cour Pharmaceuticals
ImmusanT
Teva Pharmaceutical Industries Ltd.
Pfizer Inc.
Major companies driving the celiac disease drugs market include Takeda Pharmaceutical Company Limited, ImmunogenX, Cour Pharmaceuticals, ImmusanT, Teva Pharmaceutical Industries Ltd., Pfizer Inc. focusing on enzyme therapies, immune modulation, biologics, and novel treatment development.
Celiac Disease Drugs Market Future Outlook
The future of the celiac disease drugs market is promising, driven by innovative therapies beyond gluten-free diets, including enzyme treatments, biologics, vaccines, and intestinal barrier modulators. Rising disease prevalence, improved diagnostics, and greater patient awareness will expand the addressable market globally. Technological advancements and personalized treatment approaches are expected to enhance efficacy and adherence. Emerging markets in Asia Pacific and Latin America offer new growth opportunities, while collaborations between biotech and pharmaceutical companies accelerate pipeline development. Overall, the market is projected to grow at a double-digit CAGR, shifting toward targeted pharmacological interventions and comprehensive disease management solutions.
Celiac Disease Drugs Market Historical Analysis
Historically, the celiac disease drugs market was limited, with gluten-free diets and nutritional supplements as the primary management approach. Awareness of celiac disease was low, resulting in underdiagnosis and minimal pharmaceutical intervention. Over the past decade, increasing recognition of refractory and non-classical cases prompted research into enzyme therapies, immunomodulators, and biologics. Early clinical trials focused on safety and efficacy, while dietary supplementation remained dominant. Developed regions like North America and Europe led market growth due to advanced healthcare systems and patient awareness. This historical trajectory set the foundation for rapid innovation, emerging therapies, and market expansion in recent years.
Sources
Primary Research Interviews:
Pharmaceutical & Biotech Companies
Healthcare Professionals
Distributors & Wholesalers
Regulatory & Policy Experts
Databases:
PubMed / Medline
ClinicalTrials.gov
World Health Organization
Journals:
The American Journal of Gastroenterology
Journal of Pediatric Gastroenterology and Nutrition
Alimentary Pharmacology & Therapeutics
Clinical Gastroenterology and Hepatology
Newspapers:
The Wall Street Journal
Financial Times
The Guardian
The New York Times
Associations:
Celiac Disease Foundation (CDF)
Beyond Celiac
European Society for the Study of Coeliac Disease (ESsCD)
American College of Gastroenterology (ACG)
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients