The Clomiphene Citrate Market size is anticipated to grow at a CAGR of 7% with USD 800 Mn share in 2026 and is expected to reach USD 1,280 Mn in 2033. Rising global infertility prevalence (nearly 10–15 % of couples), delayed childbearing, expanding reproductive healthcare services, and enhanced access to fertility treatments are key contributors to growth in the Clomiphene Citrate market.
PCOS is projected to account for the largest clomiphene citrate market share of 61.7% in 2026. The increasing incidence of PCOS is the main factor that contributes to anovulatory infertility globally thus affecting an estimated 11–13% of women of reproductive age worldwide.
PCOS is closely linked to infertility, with recent clinical data indicating that it contributes to the majority of anovulatory infertility cases, as many as 80% in affected women. Clomiphene Citrate remains one of the most widely prescribed first‑line pharmacological agents for induction of ovulation in PCOS patients, often before moving to alternatives like aromatase inhibitors or gonadotropins.
Recent clinical studies demonstrate that Clomiphene Citrate continues to achieve meaningful ovulation and pregnancy outcomes. For example, in a recent comparative clinical assessment, about 64% of PCOS patients ovulated and nearly 32% conceived with Clomiphene treatment. Although newer agents (e.g., letrozole) may show higher efficacy in certain populations, Clomiphene’s entrenched role in treatment protocols, especially where alternative agents are restricted or less accessible, supports its sustained usage.
To learn more about this report, Request Free Sample
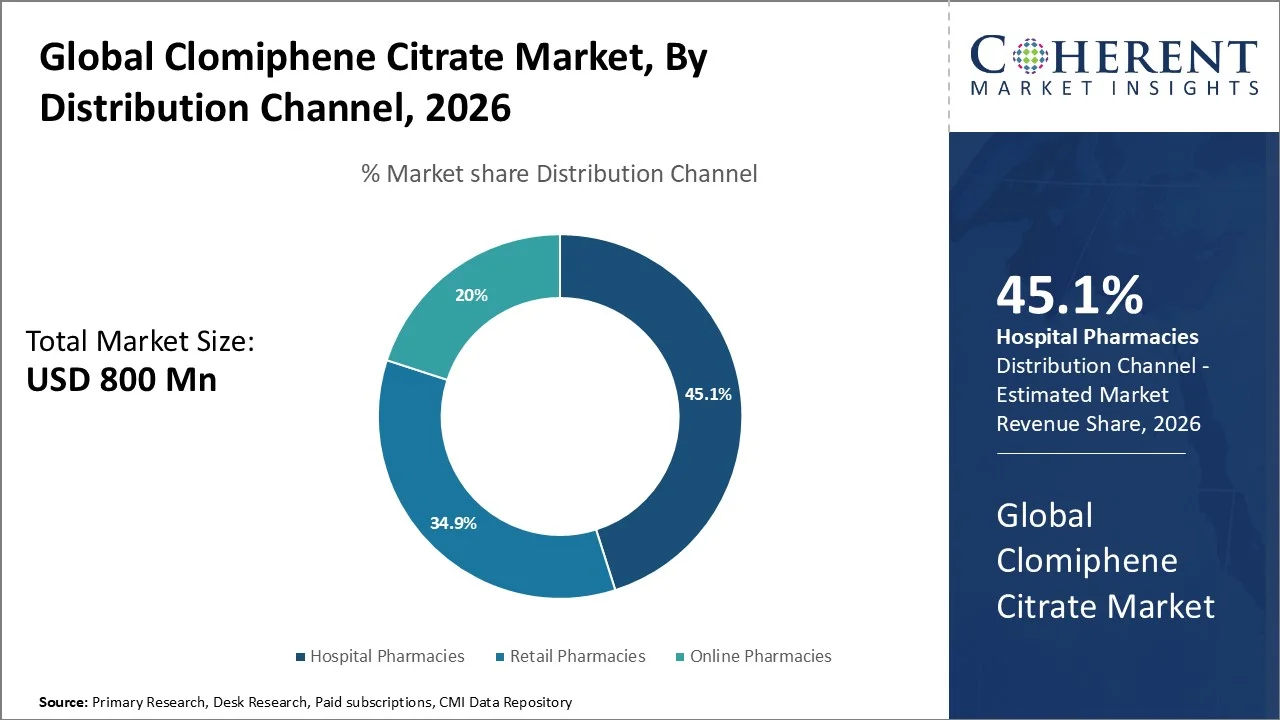
Based on distribution channel, hospital pharmacies lead with a major 45.1% share in 2026. The segment’s growth is attributed to their central role in fertility evaluation, ovulation induction, and monitored dispensing of prescription therapies. The rising infertility incidence continues to boost demand for Clomiphene Citrate, especially in supervised reproductive care settings. According to WHO, around 1 in 6 people of reproductive age experience infertility globally during their lifetime.
Hospital pharmacies benefit further because treatment initiation commonly occurs under specialist oversight. NICE recommends ultrasound monitoring during at least the first clomifene citrate cycle, while recent NHS fertility guidance notes that around 70 in every 100 patients treated with oral ovulation induction agents ovulate and about 40 in 100 conceive. This is further propelling the Clomiphene Citrate market demand.
The increased adoption of Assisted Reproductive Technologies (ART) is substantially contributing to the growth of the clomiphene citrate market in the United States. ART, which includes procedures like In-Vitro Fertilization (IVF), surrogacy, and intrauterine insemination (IUI), is transforming fertility treatments. As ART adoption rises, the demand for ovulation-inducing drugs like Clomiphene Citrate, an integral part of ART, is also seeing major growth.
According to the American Society for Reproductive Medicine (ASRM), the number of IVF cycles performed in the U.S. increased considerably, with over 432,000 cycles performed in 2023, thus leading to 95,000+ babies born via IVF. This surge in ART treatments demonstrates increased utilization of fertility drugs like Clomiphene Citrate to support ovulation before or during ART procedures.
The new formulation research is improving clomiphene citrate by addressing one of its key limitations: anti-estrogenic effects on the endometrium that can reduce implantation success. Although clomiphene citrate is still marketed mainly as a 50 mg oral tablet, the recent development work is moving toward better delivery systems and optimized treatment protocols.
An NCBI-indexed study on a sustained-release phospholipid-based nanoformulation reported a particle size of 67 nm, with 29% drug release in 24 hours and 76% release by 120 hours, while also significantly improving implantation-related markers like LIF, LIFR, HOXA10, HB-EGF, and EGF versus conventional clomiphene suspension. Separately, a 2024 meta-analysis found that combined letrozole and clomiphene therapy produced a significantly higher ovulation rate and more dominant follicles than either drug alone, highlighting how optimized delivery and regimen design can improve clinical response.
In addition to this, the FDA revised its product-specific guidance for clomiphene citrate in October 2024, supporting more standardized generic development and bioequivalence pathways for the oral 50 mg formulation. These developments are strengthening the clomiphene citrate sector by improving treatment efficiency, supporting patient access, and promoting next-generation fertility therapy innovation.
|
Current Event |
Description and its Impact |
|
2026 California Infertility Coverage Expansion and New U.S. Fertility-Benefit Guidance |
|
|
WHO’s First Global Infertility Guideline and Stronger PCOS Focus |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report

To learn more about this report, Request Free Sample
North America account 43.7% market share in 2026. The region’s growth is mainly attributable to the increasing adoption of fertility treatments and the prevalence of conditions like PCOS. The region benefits from a strong healthcare infrastructure and broad access to fertility care, which includes the use of Clomiphene Citrate for inducing ovulation. According to the CDC, nearly 13.4% of women in the U.S. aged 15-49 experience infertility while 8.5% of them are classified as infertile. This situation contributes to an increasing demand for fertility treatments, including oral medications like Clomiphene Citrate. This is further backed by a strong presence of fertility clinics, thus making treatments more accessible.
The American Society for Reproductive Medicine (ASRM) reports that fertility treatments continue to rise, with Clomiphene Citrate being one of the most commonly used first‑line medications for women with anovulatory infertility. In addition, the telemedicine market for fertility consultations is growing at a rapid pace in North America. In 2025, innovations like telehealth platforms for remote fertility consultations are expected to boost the accessibility of fertility services, thereby propelling the Clomiphene Citrate market growth.
The Asia Pacific region is poised to be as the fastest-growing region through 2026-2033. The rising infertility rates, particularly in India, China, and Southeast Asia, contribute considerably to the region’s growth. As per the data from the World Health Organization (WHO), approximately 10-15% of reproductive-aged couples in the Asia Pacific region experience infertility. In India, nearly 3.9% to 16.8% people are affected by infertility, while China faces rising infertility issues, with around 18% of couples struggling to conceive.
The rise in investments in healthcare infrastructure, mainly in rural as well as underserved areas, are propelling greater access to fertility treatments. India performs approximately 300,000 IVF cycles annually. The fertility clinics are also growing in the region. The governments are also launching awareness campaigns like the Indian government's National Infertility Awareness Program in order to educate the public about fertility and encourage early treatment. These developments are expected to drive substantial market growth for clomiphene citrate.
The United States Clomiphene Citrate Market is experiencing robust growth. This is due to the increasing infertility concerns and expanding therapeutic use. According to CDC fertility rate data, approximately 3,622,673 live births were recorded in the U.S. in 2024. Thus, showcasing ongoing shifts in reproductive health dynamics and demographic patterns that are influencing the demand for fertility care. The government statistics also highlight persistent fertility challenges, with around 11% of women and 9% of men of reproductive age facing infertility, affecting roughly 15% of heterosexual couples. This has led to a growing dependency on fertility treatments including ovulation induction agents like clomiphene citrate.
Prescribing data reveals that more than 1 million U.S. women are prescribed Clomid (clomiphene citrate) annually, highlighting its significant and entrenched position in fertility therapy.
The China Clomiphene Citrate Market is poised for substantial growth in the coming years. The growth is mainly owing to a sharp decline in birth rates and a growing need for fertility treatments. In 2025, China’s birth rate fell to a historic low, with only 7.9 million babies born, thus marking a 17% decrease from the previous year. This decline shows a bigger change in the population, with the number of births per woman falling to almost 1.0, much lower than the 2.1 births needed to maintain the population. The country is facing a shrinking population, with China’s total population decreasing by about 3.39 million in 2025. As infertility rates rise, with approximately 15% of women of reproductive age affected, the demand for fertility medications like clomiphene citrate is expected to rise at a fast pace.
Some of the major key players in Clomiphene Citrate are Sanofi S.A., Par Pharmaceutical, Merck & Co. Inc., Teva Pharmaceutical Industries Ltd., Cipla Inc., Unichem Laboratories Ltd., Emcure Pharmaceuticals Ltd., Serum Institute of India Pvt. Ltd., Incepta Pharmaceuticals Ltd., and Shanghai Trifecta Pharma Co. Ltd.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 800 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7% | 2033 Value Projection: | USD 1,280 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Sanofi S.A., Par Pharmaceutical, Merck & Co. Inc., Teva Pharmaceutical Industries Ltd., Cipla Inc., Unichem Laboratories Ltd., Emcure Pharmaceuticals Ltd., Serum Institute of India Pvt. Ltd., Incepta Pharmaceuticals Ltd., and Shanghai Trifecta Pharma Co. Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients