Global Biochemistry Analyzers and Consumables Market and Forecast – 2026-2033
The global biochemistry analyzers and consumables market is estimated to be valued at USD 4,823.2 Mn in 2026 and is expected to reach USD 6,335.3 Mn by 2033, growing at a compound annual growth rate (CAGR) of 5.5% from 2026 to 2033. The steady growth reflects increasing adoption of advanced diagnostic technologies and rising demand across healthcare, pharmaceuticals, and research sectors worldwide, driving the expansion of biochemistry analysis capabilities and related consumables.
Key Takeaways of the Global Biochemistry Analyzers and Consumables Market
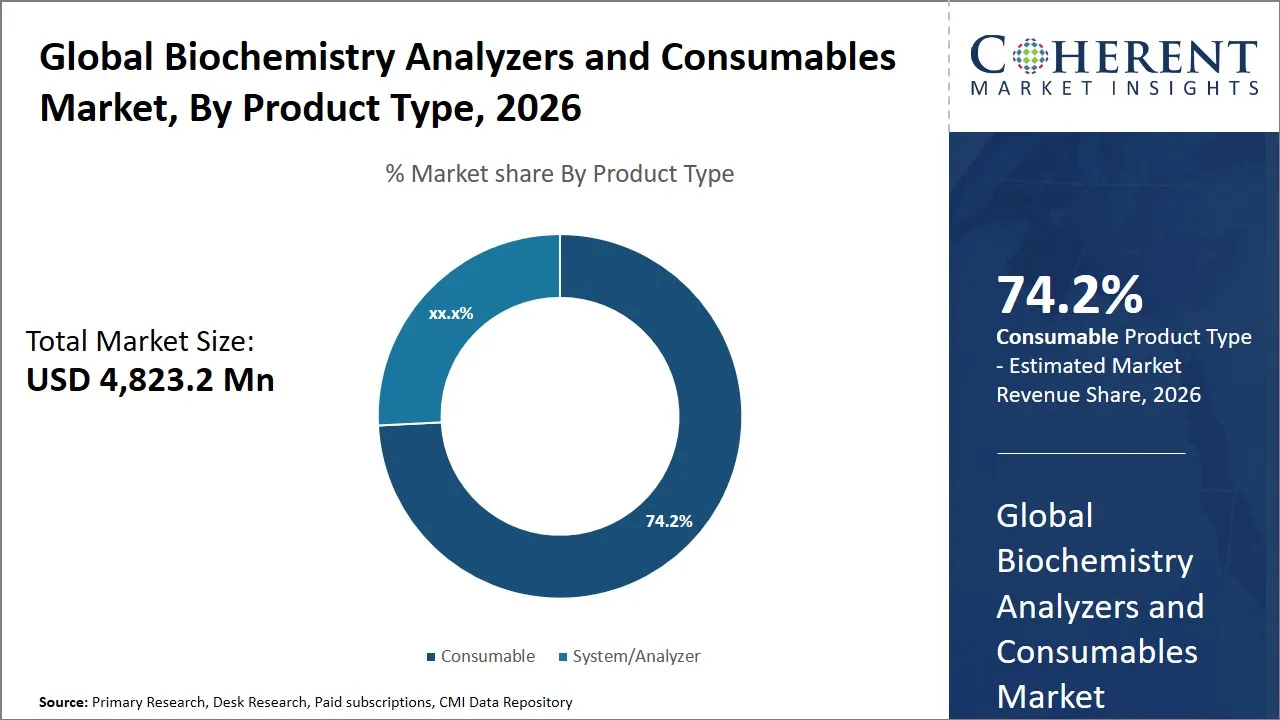
- Consumable segment is expected to lead the global biochemistry analyzers and consumables market, capturing 74.2% share in 2026.
- Fully automatic biochemistry analyzers segment is estimated to represent 66.7% of the global biochemistry analyzers and consumables market share in 2026.
- Blood segment is projected to dominate with 53.0% of the biochemistry analyzers and consumables market share in 2026.
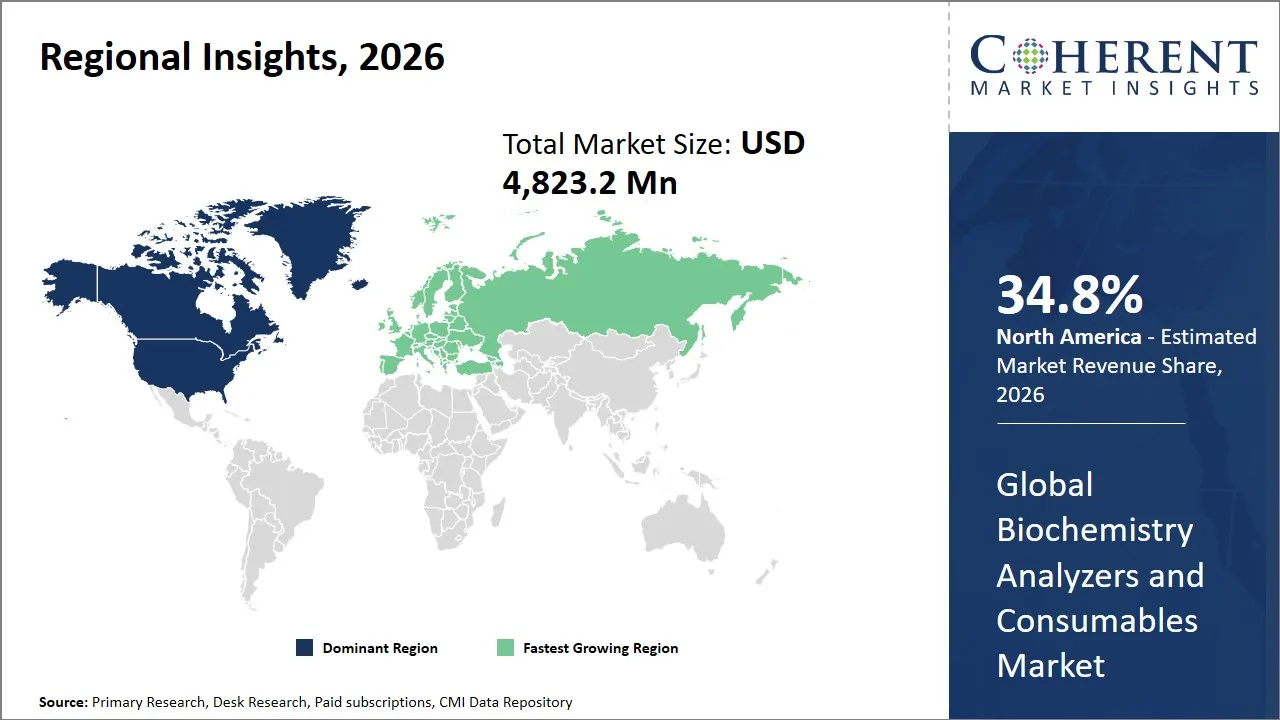
- North America is expected to lead the market, holding a share of 34.8% in 2026. Europe is anticipated to be the fastest-growing region, with 29.3% share in 2026.
Market Overview
- The global biochemistry analyzers and consumables market is expanding on the back of rising test volumes for diabetes, liver function, kidney function, lipid profile, and other routine biochemistry parameters, as chronic disease burden and preventive screening continue to increase globally.
- A core growth driver is laboratory automation, with hospitals and reference labs shifting toward high-throughput and integrated chemistry platforms that improve turnaround time, reagent handling, uptime, and workflow efficiency.
- One major trend is the move toward integrated clinical chemistry plus immunoassay systems, especially for labs looking to consolidate instruments, reduce manual touchpoints, and improve productivity in both mid-volume and low-volume settings.
- Consumables remain the backbone of revenue generation in this market because every installed analyzer drives recurring demand for reagents, calibrators, controls, and other assay-specific materials, making the installed base a long-term commercial advantage for manufacturers.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Launch of high-throughput integrated biochemistry analyzers |
|
|
Shift toward compact integrated systems for decentralized and mid-volume labs |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Consumable Segment Dominate the Global Biochemistry Analyzers and Consumables Market in 2026?
The consumable segment is expected to hold the largest market share of 74.2% in 2026, owing to the essential role these items play in the routine operation and maintenance of biochemistry analyzers. Consumables are also included like reagents, cuvettes, tubes, valves, and pumps among others that are essential to prevent biochemical samples to be processed with precision. The frequent character of the type of products they offer leads to constant replenishment hence becomes a regular stream of revenue in the market. This exorbitant reliance upon consumables is occasioned by the mere or bulk of biochemical testing which is done in clinical laboratories, hospitals, and diagnostic centers at the expense of thousands of tests which require fresh supplies daily in order to maintain precision and reliability.
Besides, there has been a breakthrough in technology of consumables that has enhanced the accuracy of the tests and made them even faster hence strengthening their imperativeness. As an example, disposable cuvettes avoid the possibilities of cross-contamination, and, therefore, high standards of quality control are ensured. In a similar manner, new materials in tubes and valves improve the sample integrity and minimize errors that arise due to handling of the sample. The growth of personalistic medicine and rising rates of chronic illnesses only add to the pressure on a diversified consumable market since the diagnostic tier is increasingly sophisticated and intricate.
For instance, in May 2025, Furuno Electric launched the CA-800 clinical chemistry analyzer capable of performing up to 800 tests per hour and up to 1,200 tests per hour with ion selective electrode modules. The system includes automated workflows and touchscreen interfaces designed to improve laboratory productivity and accuracy. Such product launches illustrate continued technology development and competition among manufacturers to improve analyzer performance and expand diagnostic testing capacity.
(Source- Furuno Electric)
Why Does the Fully Automatic Biochemistry Analyzers Segment Dominate the Global Biochemistry Analyzers and Consumables Market in 2026?
The fully automatic biochemistry analyzers segment is expected to hold 66.7% of the market share in 2026, propelled by the increasing emphasis on efficiency, accuracy, and throughput in clinical diagnostics. Fully automatic analyzers simplify the process of carrying out the entire biochemical testing process which includes handling the samples and production of the result to the minimal human involvement, and minimization of possibilities of error. This automation brings in quality and reproducibility that is essential in high-volume laboratories and a hospital environment where quick diagnostic decisions may have a direct impact on patient care.
The increasing sophistication of the clinical tests are becoming more difficult to manage, requiring systems that can be used to support multiple types of tests at the same time, which fully automatic systems provide with built-in protocols and configurable workflows. Another reason why these analyzers are preferred is the fact that they can be connected with the laboratory information management systems (LIMS) so that real-time data management, reporting, and analytics could be provided. This union will improve the productivity of the lab and speed up patient diagnosis, as part of the digitization of the healthcare industry more generally.
For instance, in June 2024, Roche introduced the cobas c 703 and cobas ISE neo units for its cobas pro integrated platform, designed for high-throughput clinical chemistry testing in large laboratories. The system improves automation and sample processing capacity, reinforcing the growing preference for fully automatic biochemistry analyzers that enable faster results, reduced manual intervention, and higher operational efficiency in diagnostic laboratories.
(Source- Roche)
The Blood Segment Dominates the Global Biochemistry Analyzers and Consumables Market
The blood segment is expected to hold the largest market share of 53.0% in 2026, driven by its widespread use in clinical biochemistry testing due to the rich biochemical information it provides. Blood samples are used as the basic sample in the diagnosis and monitoring of a wide range of diseases such as metabolic disorders, abnormalities in organ functions, infections and cardiovascular diseases. The rich and in-depth composition of blood plasma, serum, and cellular components enables the implementation of a wide range of biochemical tests, which is why blood is essential in both routine and sophisticated diagnostics.
The use of blood as the main type of sample is supported by the fact that the sampling and handling procedures are well-developed and ensure the usage of reliable and uniform testing activities. Furthermore, blood can be simply converted into many different derivatives, such as serum and plasma, and this increases the testing range even in the same biological source. The validity and clinical significance of blood tests support the use of blood tests as a first-line test, which in many cases leads to additional analysis of the sample or confirmation tests.
Global Installed Base of Clinical Chemistry Analyzers Driving Consumables Demand
- The number of clinical chemistry analyzers installed all over the world is significant and is growing as the hospitals, reference laboratories and independent diagnostic chains upgrade routine testing capacity. There is no specific number in the global number of units to offer in all manufacturers that can be found in the public sources, but there remain large diagnostics companies that are still stressing on the strategic significance of the installed base in core laboratory applications. Examples of this include Roche which has recurrently emphasized its extensive worldwide installed base of diagnostics and its commercial coverage such as its core laboratory presence in clinical chemistry and immunoassay processes.
- The installed base is important in the market in that it provides a long-term recurring demand of reagents, calibrators, controls and service contracts through the placements of the analyzers. This is why it is no longer a battle of selling instruments but increasing the number of analyzer footprints at high-volume core labs and mid-volume decentralized locations, and maintaining their business with menu breadth, workflow integration and consumables pull-through. Roche has also attributed high-volume chemistry growth directly to installed-base and revenue growth that indicates the overall trend of global biochemistry analyzers and consumables market.
Regional Insights
To learn more about this report, Request Free Sample
North America Biochemistry Analyzers and Consumables Market Analysis and Trends
The North America region is projected to lead the market with a 34.8% share in 2026, driven by a well-established healthcare infrastructure, substantial investment in research and development, and strong presence of key industry players. The well-developed ecosystem of the market is driven by a blend of both high levels of technology innovation, high rate of automated biochemistry equipment usage, and supportive regulatory climate that facilitates products approval and commercialization. The government programs of encouraging precision medicine and enhancing diagnostic strength further consolidate leadership in the market in this region.
The major players including Tony Abbott Laboratories, Beckman Coulter (Danaher company), and Siemens Healthineers among others have a considerable presence here and are constantly coming up with new products and product lines to suit clinical and research uses. Also, trade has operated in favor of streamlined importation and exportation processes, and intellectual property protection, which enhances long term growth and competition between the local and foreign players.
For instance, in February 2025, the U.S. FDA posted a Class 2 recall for the DxC 500 AU after the company-initiated action on January 10, 2025. The recall is relevant to the North America Biochemistry Analyzers and Consumables Market because it shows how performance and quality issues can directly affect analyzer uptime, laboratory workflow, and consumables usage in routine chemistry testing.
(Source- U.S. FDA)
Europe Biochemistry Analyzers and Consumables Market Analysis and Trends
The Europe region is expected to exhibit the fastest growth in the global biochemistry analyzers and consumables market contributing 29.3% share in 2026, supported by a dense network of hospital laboratories, public healthcare systems, and well-established diagnostic testing standards. Incessant automation of the laboratories, the substitution of the old chemistry systems and the necessity to provide high-throughput platforms capable of managing increasing routine tests are all influencing demand.
Meanwhile, Europe is among the most regulation-oriented areas of the in vitro diagnostics and the IVDR framework is increasing documentation, classification, and compliance-related requirements of analyzers, software, calibrators, and other consumables. That pressure is more towards the benefit of large manufacturers with greater regulatory and service capacities and also throwing more barriers towards smaller players.
For instance, in December 2025, MedTech Europe published new data outlining the scale and trends of the European in vitro diagnostics industry. The report provides market estimates across EU member states and highlights the strong role of laboratory diagnostics in the region’s healthcare systems. Such updates reflect Europe’s established position as a major market for clinical chemistry analyzers and consumables due to advanced healthcare infrastructure and widespread laboratory testing capacity.
(Sources- MedTech Europe)
Global Biochemistry Analyzers and Consumables Market Outlook for Key Countries
How is the U.S. Helping in the Growth of the Biochemistry Analyzers and Consumables Market?
U.S. biochemistry analyzers and consumables market is one of the world standards in adoption of biochemistry analyzers and consumption of biochemistry consumables with the help of a wide network of hospitals, diagnostic labs and research institutes. The major corporations like Abbott, Beckman Coulter and ortho clinical diagnostics have large operations and innovation facilities in this area. The American market enjoys a high regulation standard of product provision that is of high standard coupled with quick reimbursement system that facilitates its adoption.
Moreover, there are strategic partnerships between the industry and academic centers that drive an ongoing product innovation based on diagnostics of chronic diseases, personalized medicine, and point-of-care testing.
How is Germany Helping in the Growth of the Biochemistry Analyzers and Consumables Market?
The biochemistry analyzers industry is leading in Germany within Europe, supported by an advanced healthcare system and a strong medical equipment production industry. There are such companies as Siemens Healthineers and Roche Diagnostics, which are not newcomers in the country, thus investing in the technological process and product diversification. Healthcare innovation policies in the German government, coupled with effective enforcement of intellectual property, encourage growth in the market based on research. Germany is also a strategic market to export medical diagnostics equipment to other European countries with a well-developed logistics and trade networks.
Key Drivers for the Growth of the China Biochemistry Analyzers and Consumables Market
China has remained the dominating player in the market in Asia Pacific through the upsurge of more investments in their healthcare infrastructures and diagnostics capabilities. The local industry like Mindray Medical International is fast gaining grounds owing to its creation of reasonable biochemistry analyzers that are aligned to the varied Chinese healthcare environment.
The government policy of China focuses on the innovation of biotechnology and production within the country by giving incentives and friendly regulations to minimize reliance on imports. The rising middle-income and rising expenditure on healthcare also enhance demand, as well as the expansion of the hospital network and diagnostic laboratories with modern biochemical analyzers to make screening programs popular among the population.
Japan Biochemistry Analyzers and Consumables Market Trends
The market in Japan is characterized by high level of technology and the aging people that enhances the need to have high-quality diagnostic solutions. Such companies as Sysmex Corporation and Toshiba make significant contributions as they introduce advanced and small-size biochemical analyzers in different healthcare facilities such as large hospitals and outpatient clinics. Healthcare innovation is also encouraged by the government by subsidizing and regulating technologies in new diagnostics. The culture of quality and precision medicine in Japan largely affects the development of products and the preferences of the market and thus, Japan is a crucial player in the domestic and global market.
Laboratory Automation Transforming the Global Biochemistry Analyzers and Consumables Market
- Laboratory automation is emerging as a growth driver in the global biochemistry analyzers and consumables market as labs are challenged with increasing samples, decreasing turnaround times and the persistent challenge of doing more with less manual intercession. Advanced automated systems of biochemistry are progressively integrating sample processing with reagent handling, calibration and result processing into a more simplified workflow, which can assist hospitals and diagnostic laboratories to enhance consistency and minimize bottlenecks in their workflows. This is the reason why demand is turning to integrated and scalable systems in analyzers instead of simple standalone.
- In the case of consumables side, automation is of even greater importance since all automated platforms raise the reliance on validated reagents, calibrators, controls, and standardized assay workflows. That gives manufacturers greater recurring revenue and more switching costs to the laboratories once installed with a system. This is observable in both high-volume core labs and smaller decentralized environments, where small automated systems are taken up due to efficiency, menu flexibility, and reduced maintenance load. Roche, as an illustration, has pointed out to extensive adoption of compact chemistry analyzers worldwide and so has ADLM with regard to instrumentation upgrades and automation being a key present lab emphasis.
Market Players, Key Developments, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In November 2025, Abbott announced it would acquire Exact Sciences in a deal valued at up to USD 23 billion including debt, expanding Abbott’s diagnostics presence in cancer screening and precision oncology. This is not a pure biochemistry analyzer deal, but it is relevant as a major diagnostics acquisition showing how large IVD players are broadening test portfolios and strengthening lab diagnostics ecosystems.
- In May 2025, Siemens Healthineers introduced sustainability-focused solutions for clinical laboratories aimed at reducing water, energy, and plastic use in lab testing workflows. While not a standalone analyzer launch, this is directly relevant to the consumables side of the market because labs are increasingly evaluating chemistry platforms based on operating efficiency and consumable footprint.
- In March 2025, Beckman Coulter confirmed U.S. FDA clearance for the DxC 500i, expanding its commercial reach after earlier availability in CE-mark accepting countries. This matters because regulatory clearance in the U.S. opens a larger installed-base opportunity for both analyzer placements and repeat consumables demand.
Top Strategies Followed by Global Biochemistry Analyzers and Consumables Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Established market leaders strengthen their position by investing heavily in R&D to launch faster, more accurate, and more automated biochemistry analyzers. They also use partnerships and OEM collaborations to improve system integration and expand product capabilities, while widening distribution in emerging markets to capture new demand and reduce dependence on mature regions. |
Roche Diagnostics has continued to strengthen its leadership through innovation in integrated clinical chemistry platforms such as its cobas systems, while also expanding its reach across hospital and laboratory networks globally. |
|
Mid-Level Players |
Mid-level players compete by offering reliable biochemistry analyzers and consumables at more affordable price points, targeting smaller hospitals, diagnostic labs, and cost-sensitive markets. They often depend on partnerships with technology providers, contract manufacturers, and regional distributors to improve efficiency, expand reach, and stay competitive without the R&D budgets of larger multinational companies. |
Mindray has built strong market presence by providing cost-effective clinical laboratory systems with essential automation features, making it a competitive alternative in many emerging and mid-tier healthcare markets. |
|
Small-Scale Players |
Small-scale players usually focus on niche opportunities within the biochemistry analyzers and consumables market by offering specialized products, compact systems, or application-specific reagents. They stay relevant by adopting new technologies early and building local partnerships with regional manufacturers and distributors to improve visibility and reach targeted customers. |
DiaSys Diagnostic Systems is a good example of a smaller specialized player that focuses on clinical chemistry reagents and analyzer solutions for specific laboratory needs. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
Biochemistry Analyzers and Consumables Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 4,823.2 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.5% | 2033 Value Projection: | USD 6,335.3 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Roche Diagnostics, Siemens Healthineers, Abbott Laboratories, Beckman Coulter, Mindray, HORIBA, QuidelOrtho, Randox Laboratories, Erba Mannheim, Agappe Diagnostics, DiaSys Diagnostic Systems, HUMAN Diagnostics, Diatron, DIRUI Industrial, and Sysmex Corporation |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Biochemistry Analyzers and Consumables Market Dynamics
To learn more about this report, Request Free Sample
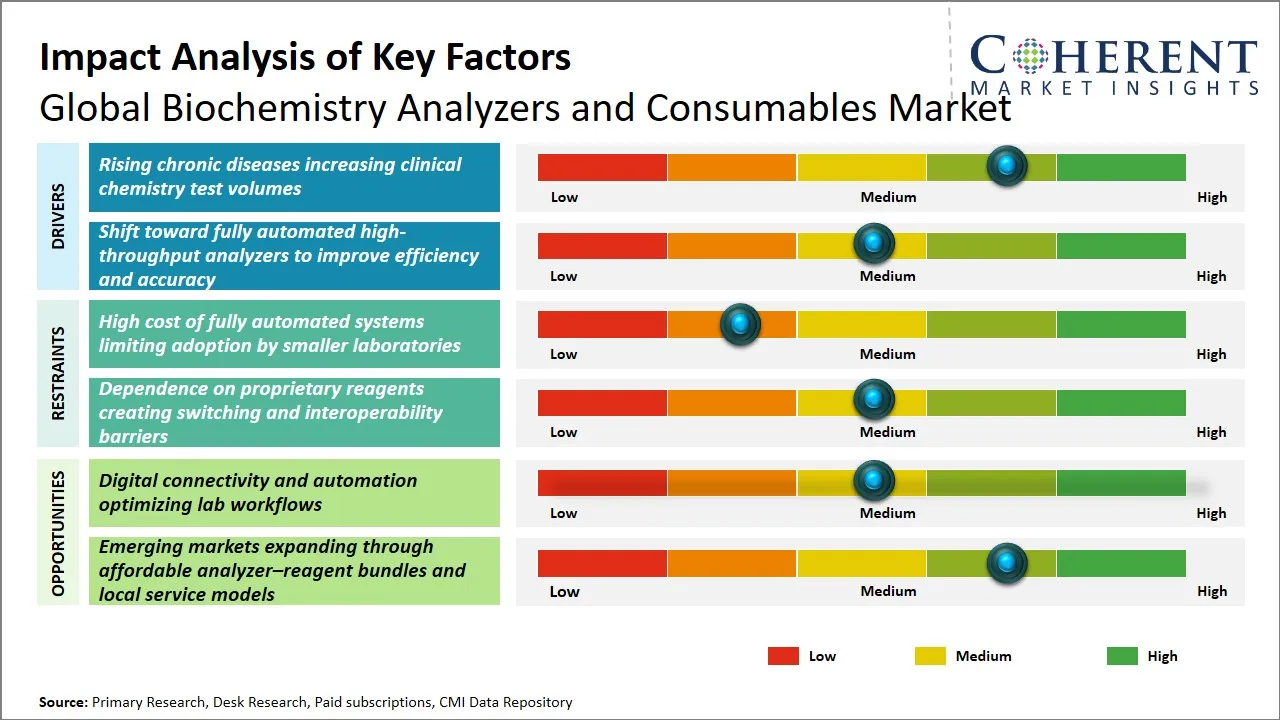
Global Biochemistry Analyzers and Consumables Market Driver - Rising chronic diseases increasing demand for clinical chemistry testing
The growing population of people with chronic illnesses including diabetes, liver problems, kidney problems, and cardiovascular diseases is largely contributing to the growing demand of biochemistry analyzers and consumables across the world. Since these diseases involve regular and accurate monitoring with regular clinical tests of chemistry, hospitals and tests proving facilities are experiencing a boom in the number of tests. Regular self-monitoring of the level of glucose, periodic laboratory diagnosis and monitoring of liver diseases, and regular monitoring of kidney diseases through constant monitoring of renal biomarkers are all conditions of diabetes management.
This trend is further aggravated by cardiovascular diseases, which have led to the need of lipid profiling and enzyme testing in diagnosing and treatment effectiveness. Such high rate of testing does not only enhance the use of modern biochemistry analyzers that can provide rapid and accurate results but also enhances the repetitive usage of such reagents and consumables.
For instance, in January 2025, the U.S. Food and Drug Administration (FDA) approved Ozempic for use in people with type 2 diabetes and kidney disease. This approval marks an important milestone in kidney disease treatment, offering a new option to help reduce complications like heart disease, dialysis, or kidney transplant.
(Source- Ozempic)
Global Biochemistry Analyzers and Consumables Market Opportunity - Digital connectivity and automation optimizing lab workflows
The global biochemistry analyzers and consumables market will grow due to rising usage of digital connectivity, remote diagnostics, and automation-based workflow optimization, especially in large reference laboratory and large hospital systems. Since the global healthcare systems focus more on the efficiency of their operations and the accuracy of data, the adoption of sophisticated digital tools into biochemistry analyzers will allow sharing data and monitoring the state of various testing locations in real-time. This connectivity enables remote diagnostics whereby specialists can read complicated biochemical results without necessarily being present thus minimizing the turnaround times and enhancing patient outcomes.
For instance, in October 2023, Roche Diagnostics introduced the cobas connection modules (CCM) Vertical, an automated conveyor system that transports laboratory samples between different floors and testing areas without reducing throughput. The system helps laboratories increase testing capacity, accelerate result turnaround time, and reduce manual errors by enabling fully connected and automated diagnostic workflows.
(Sources- Roche Diagnostics)
Analyst Opinion (Expert Opinion)
- The global biochemistry analyzers and consumables market is expanding by a steady growth because of the increase of routine diagnostic tests in diabetes, kidney disorders, liver disorders and heart related diseases. There is a shift towards automated and integrated systems which enhance speed, accuracy and efficiency in workflow and the need to replenish reagents, calibrators and controls is recurrently encouraging market growth. The greatest opportunities are in mini systems of mid-sized and decentralized labs, particularly in the emerging markets. The regulatory complexity is still the primary obstacle, especially in Europe where the compliance with IVDR is getting more expensive and demands more approval.
- The market has been maintained within the context of automation, assay growth, sustainability, and lab standardization through discussions at ADLM, IFCC WorldLab, EuroMedLab, and Medlab Middle East. Recent cases like new cobas chemistry units at Roche, introduction of DxC 500i and FDA approval of Beckman Coulter, sustainability-based lab programs at Siemens Healthineers indicate a new direction of competition, with an emphasis on platforms, more powerful consumables ecosystems, and more operational value to the laboratories.
Market Segmentation
- Product Type Insights (Revenue, USD Mn, 2021 - 2033)
- Consumable
- Cuvettes
- Tubes
- Valves
- Pump
- Others (Lamp, Electrodes,etc.)
- System/Analyzer
- Bench Top
- Floor Standing
- Consumable
- Automation Insights (Revenue, USD Mn, 2021 - 2033)
- Fully Automatic Biochemistry Analyzers
- Semi-automatic Biochemistry Analyzers
- Sample Type Insights (Revenue, USD Mn, 2021 - 2033)
- Blood
- Urine
- Serum
- Plasma
- Others (Cerebrospinal fluid, etc.)
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Diagnostic Laboratories
- Hospitals and Clinics
- Pharmaceutical and Biotechnology Companies
- Others (Ambulatory Surgical Centers, Academic Research Institutes, CRO’s, etc.)
- Regional Insights (Revenue, USD Mn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Roche Diagnostics
- Siemens Healthineers
- Abbott Laboratories
- Beckman Coulter
- Mindray
- HORIBA
- QuidelOrtho
- Randox Laboratories
- Erba Mannheim
- Agappe Diagnostics
- DiaSys Diagnostic Systems
- HUMAN Diagnostics
- Diatron
- DIRUI Industrial
- Sysmex Corporation
Sources
Primary Research Interviews
Industry Stakeholders list
- Clinical laboratory directors
- Heads of core laboratory operations
- Clinical chemistry consultants
- IVD regulatory affairs experts
- Hospital procurement managers for diagnostics
- Laboratory automation and workflow specialists
End-users list
- Hospital biochemists
- Pathologists overseeing clinical chemistry
- Reference laboratory managers
- Independent diagnostic center owners
- Laboratory quality assurance managers
- Senior medical technologists in clinical chemistry
Government and International Databases
- FDA MAUDE Database
- CMS Clinical Laboratory Fee Schedule
- CDC NHANES Laboratory Data
- WHO Global Health Observatory
- WHO Global Health Expenditure Database
- OECD Health Statistics
Trade Publications
- 360Dx
- CAP TODAY
- Clinical Lab Products
- Today’s Clinical Lab
- LabPulse
- The Pathologist
Academic Journals
- Clinical Chemistry
- Clinical Biochemistry
- Practical Laboratory Medicine
- Clinica Chimica Acta
- Annals of Clinical Biochemistry
- Journal of Applied Laboratory Medicine
Reputable Newspapers
- Financial Times
- The Wall Street Journal
- The New York Times
- The Washington Post
- The Guardian
Industry Associations
- International Federation of Clinical Chemistry and Laboratory Medicine
- Association for Diagnostics and Laboratory Medicine
- Clinical and Laboratory Standards Institute
- Association of Public Health Laboratories
- MedTech Europe
- Asia Pacific Federation for Clinical Biochemistry and Laboratory Medicine
Public Domain Resources
- ClinicalTrials.gov
- WHO Essential Diagnostics List
- NIH Bookshelf
- CDC Laboratory Data and Methods Resources
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 8 Years
Share
Share
About Author
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients