The Liquid Biopsy Market is anticipated to grow at a CAGR of 27.5% with USD 1,354.0 Mn share in 2026 and is expected to reach USD 7,340 Mn in 2033. The rising global cancer incidence (according to the data published by the American Cancer Society (ACS), at least 40% of new cancers diagnosed in U.S. Increasing demand for non-invasive diagnostics, and growing adoption of precision oncology drive market growth, supported by advancements in circulating tumor DNA (ctDNA) and RNA profiling technologies and expanding clinical applications in early cancer detection and treatment monitoring.
|
Current Event |
Description and its Impact |
|
Increasing approval of new product & Regulatory Shift |
|
|
Rising Government Initiative & Funding |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
ctNA (Circulating Tumor Nucliec Acids) is projected to account for the largest share of liquid biopsy in 2026, representing approximately 48.0% of the total volume. ctNA (Circulating Tumor Nucliec Acids) can detect tumor-specific mutations even at very low concentrations. It enables early-stage cancer detection, where other biomarkers (like CTCs) often fail. ctNA (Circulating Tumor Nucliec Acids) is extracted directly from blood samples and there is no need for complex cell isolation (unlike circulating tumor cells) and supports frequent, repeat testing. It has wide clinical applications in cancer screening, therapy selection, prognosis and recurrence monitoring and hence, broad usage increases market adoption. Growing regulatory approvals and commercial tests supports the dominance of the segment in the market. For instance, in January 2026, the U.S. Food and Drug Administration has approved Guardant360 CDx as a companion diagnostic for the Pfizer colorectal cancer treatment (BRAFTOVI + cetuximab) in those with BRAF V600E-mutant metastatic colorectal cancer. This was a big step forward for the use of ctDNA liquid biopsy in clinical practice. It allows for quick genomic profiling from a simple blood sample to find actionable mutations that help doctors decide on targeted therapy, especially when traditional tissue biopsies are hard to get or don't give enough information. The clearance also made it useful for more types of cancer than just lung and breast cancer. It was also covered by major payer plans, making it easier for doctors and patients to get.
Furthermore, precision oncology is increasing demand for rapid detection of actionable mutations to guide targeted therapy due to which the demand for the ctDNA has increased. For instance, in September 2025, according to the study carried out by the European Society of Medicine, recent clinical studies indicate that next-generation sequencing (NGS)–based liquid biopsies consistently identify actionable mutations in ctDNA among patients with advanced non-small cell lung cancer (NSCLC), even in the absence of tumor tissue samples for analysis. In a real-world observational study published in 2025–2026, ctDNA analysis detected actionable mutations in 41.6% of NSCLC patients with restricted tissue availability, including common driver changes such as EGFR, ALK, KRAS G12C, RET, and ERBB2 variations. These results allowed doctors to start matched targeted medicines (such EGFR tyrosine kinase inhibitors and ALK inhibitors) faster than they could have with tissue testing alone. This sped up the process of making precise treatment decisions in a real clinical setting.
To learn more about this report, Request Free Sample
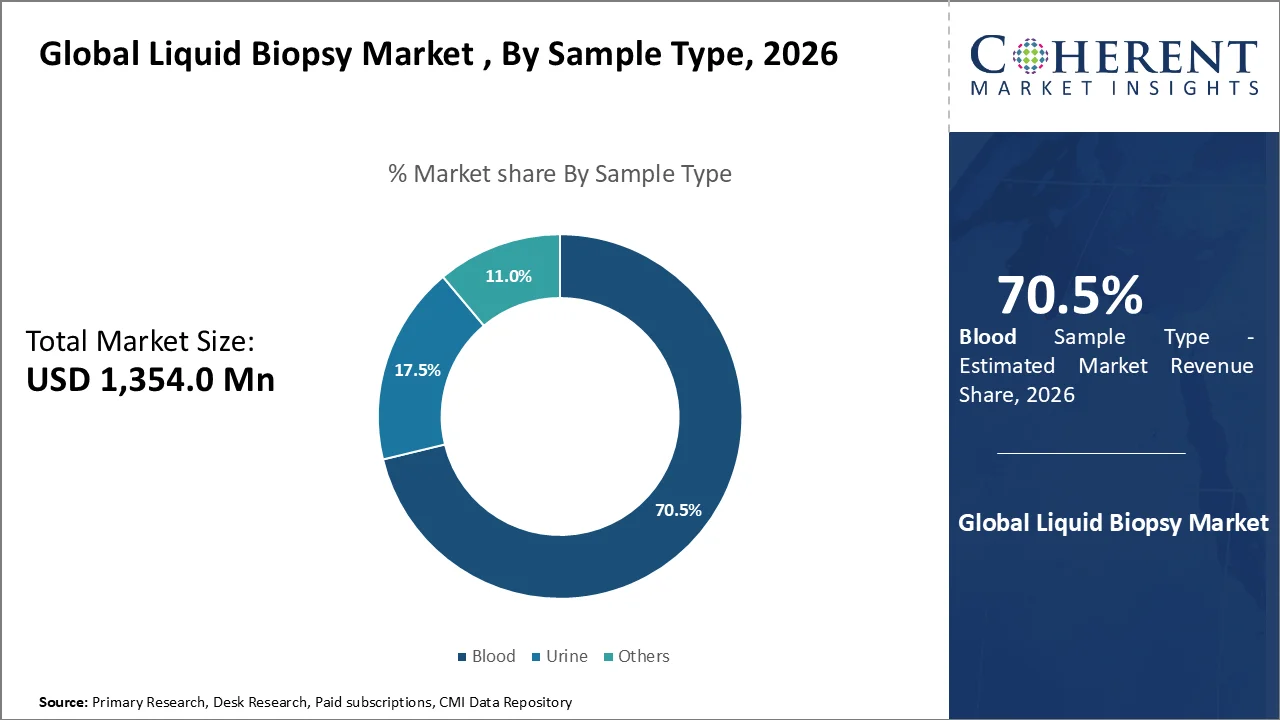
Based on sample type, blood dominate the market, accounting for a significant 70.5% share in 2026, owing to minimally invasive nature, high biomarker availability, and compatibility with advanced technologies. Rising cancer burden due to aging population globally has increased demand for early and non-invasive diagnostics, which has estimated to drive the growth of the segment in the near future. For instance, in 2026, according to the data published by the National Center for Biotechnology Information, it has been estimated that due to an aging population and lifestyle factors, the number of new cases of cancer in India is expected to increase from roughly 1.4–1.5 million in 2022–2024 to 1.57–1.87 million by 2026. Cancer cases in India may increase by 67% by 2045. This means that early detection is crucial for a disease that has the third-highest incidence in the world. By 2045, incidence is predicted to grow to over 2.45 million.
Hospitals account for the largest share of 43.5% in 2026 due to their advanced infrastructure, access to skilled physicians, and ability to handle high patient volumes. Hospitals are the first contact for cancer diagnosis and treatment which handle large patient volumes, leading to higher test utilization. Hospitals have integrated oncology infrastructure which means availability of pathology labs, oncology departments, and imaging facilities which enables end-to-end cancer managements in one place. Many hospitals have advanced molecular diagnostic labs which can perform liquid biopsy tests internally with faster turnaround time. Rising government & private investments and funding for diagnostic laboratories and cancer care centers rises the liquid biopsy market demand in hospitals. For instance, in January 2026, MedPark Hospital, unveiled Genomics Laboratory in January 2026 and stated that its NGS capabilities lets them analyze numerous genes with high resolution and a faster turnaround time to help with diagnosis, risk assessment, and treatment planning. In January 2026, 4baseCare announced the first close of a Rs 90 crore Series B round in India. In March 2026, it announced a partnership with the Government of Maharashtra to set up a Centre of Excellence for Precision Oncology that will use NGS technology and the AI-powered OncoTwin platform to make treatment decisions that are more personalized and based on data. In addition, in 2026, investments like UMMC's USD 125 million cancer center campaign, UC Davis' US$2 million early-detection grant, Illumina-Labcorp's expanded precision-oncology testing collaboration, MedPark's genomics lab launch, and 4baseCare's Rs 90 crore funding and Maharashtra precision-oncology center partnership showed how money is making the ecosystem better for finding advanced cancer and choosing the right treatment.
Increasing aging population has significantly driven the growth of the market. Aging population higher cancer risk and increases demand for diagnostics. For instance, in October 2025, according to the data published by the World Health Organization, it has been estimated that by 2050, 80% of elderly people will live in nations with low or middle incomes. One out of every six persons in the globe will be 60 years old or older by 2030. The number of people aged 60 and over will rise from 1 billion in 2020 to 1.4 billion at this time. By the year 2050, there will be 2.1 billion people in the globe who are 60 years old or older. By 2050, the number of people who are 80 years old or older is predicted to treble, reaching 426 million.
(Source: WHO)
Government campaigns, media, and healthcare initiatives educate people about cancer risks and importance of early diagnosis. More people become proactive about health check-ups. Awareness leads to more people enrolling in cancer screening camps which increases screening volume significantly. People prefer less painful and convenient tests and liquid biopsy (blood-based) becomes a preferred option over tissue biopsy. In February 2026, World Cancer Day (February 4, 2026) is still a big event that raises awareness around the world. Its topic is early identification and screening for cancer. Public health advertising, educational messages, and outreach are all part of this occasion. They all stress how important screening methods like blood testing and cancer marker tests are for finding cancer earlier in people who don't have any symptoms.
Furthermore, in February 2026, the U.S. passed the Medicare Multi-Cancer Early Detection Coverage Act, which set up a means for Medicare to pay for FDA-approved multi-cancer early detection (MCED) tests, most of which are based on liquid biopsies. This strategy helps make advanced blood-based cancer screening a regular part of healthcare for older persons. This helps with early diagnosis and makes sure that both doctors and patients know about it. Coverage support also helps people use a product and get paid for it, which can speed up adoption.
Moreover, in May 2025, NHS England became the first healthcare system to routinely use a liquid-biopsy blood test for suspected lung cancer before a tissue biopsy in about 176 hospitals. This led to the use of liquid biopsy in real-world screening and diagnostic workflows, speeding up focused therapy and cutting down on unneeded invasive procedures.
Increasing research & development and product innovation has created significant opportunity for the growth of the market in the near future. Development of new assays and multi-cancer early detection (MCED) tests drives future growth. For instance, in January 2026, GRAIL, Inc., a healthcare company whose goal is to find cancer early when it can be treated, sent the last part of its Premarket Approval (PMA) application for its Galleri multi-cancer early detection (MCED) test to the U.S. Food and Drug Administration (FDA). Furthermore, in January 2026, Caris Life Sciences, a top AI TechBio company that focuses on patients and is a leader in precision medicine, has teamed up with Everlywell, a digital health company that is leading the way in the next generation of biomarker intelligence by combining AI-powered technology with human insight to give people personalized, actionable health answers. Caris Life Sciences, biotech company and Everlywell entered into partnership to produce Caris Detect, a blood-based screening test for several cancers that will be available on Everlywell's platform. Caris Detect uses whole genome sequencing (WGS) technology to look for different types of cancer by looking at molecular signals that are in the blood.
(Source: Caris Life Sciences)
To learn more about this report, Request Free Sample
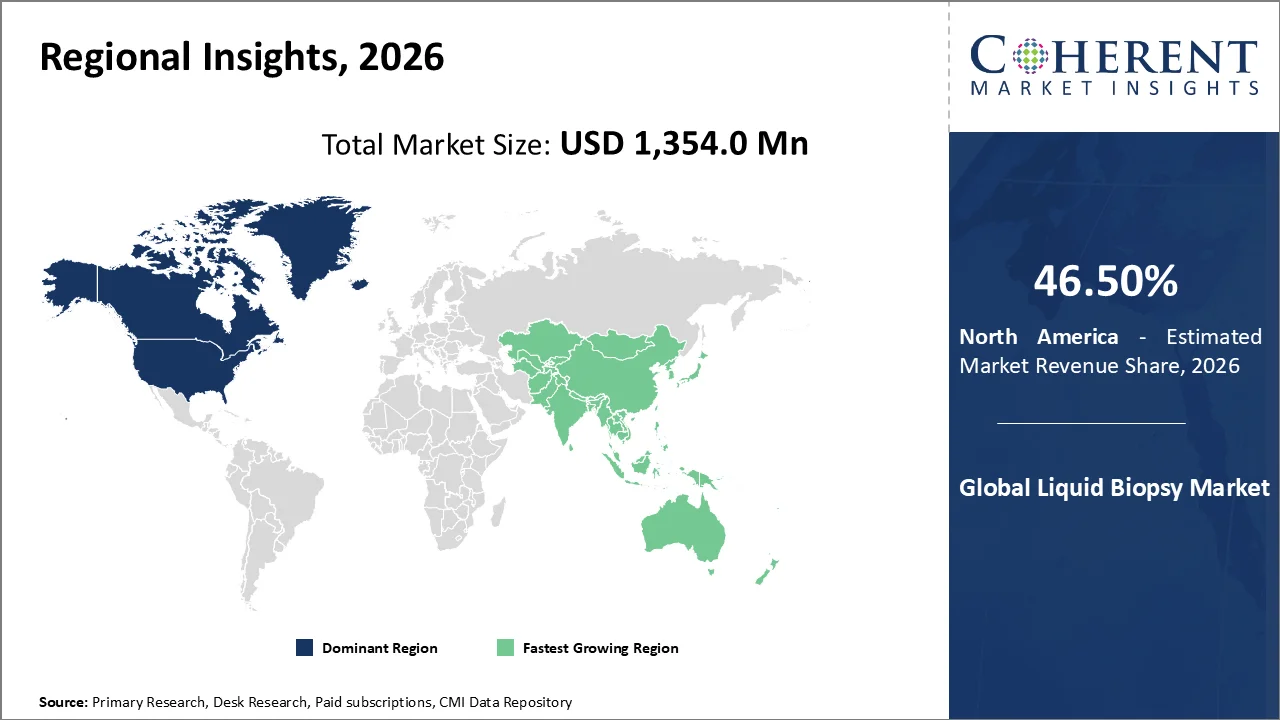
North America is expected to account for a dominant 46.50% market share, supported by early adoption innovative technologies, advanced healthcare infrastructure, and strong presence of key market players. Headquarters of major companies like Guardant Health, ILLumina, and Bio-Rad are in North America region which leads to higher product availability and commercialization. Significant spending on cancer care and advanced diagnostics enables affordability and accessibility in the region.
North America is home to a robust healthcare ecosystem, with significant investments in cancer care centers and diagnostic technologies. For instance, in April 2026, the American Cancer Society, revealed announced an USD 80.7 million investment in new cancer research grants across 128 projects at 71 U.S. institutions, aimed at advancing cancer detection, treatment, and survivorship research a major funding boost for innovative work spanning early diagnostics to precision therapies. The American Cancer Society states that the new slate covers a wide spectrum of studies in areas like cancer prevention, detection, treatment, survivorship, and more throughout the cancer control continuum. In 2026, federal funding for the National Cancer Institute (NCI) went raised to about USD 7.35 billion. This included research project awards that help pay for the infrastructure for clinical and translational cancer research at major cancer centers across the country. Furthermore, in the private sector, Precede Biosciences, a Boston-based biotechnology company raised roughly US$84 million in January 2026 to improve its liquid biopsy platform, which is meant to work with next-generation precision cancer treatments. This shows that investors have a lot of faith in new commercial diagnostic technologies.
The Asia-Pacific region is poised to be as the fastest-growing region through 2026-2033, owing to improving healthcare infrastructure, growing awareness of early diagnosis and large patient population. Asia Pacific has a huge population base which leads to high testing volumes and market potential. Rising adoption of advanced technologies for diagnosis purpose accelerates liquid biopsy market growth. Many Asia‑Pacific countries have aging populations, where cancer risk increases, further driving demand for effective diagnostic solutions. For instance, according to the data published by the UNFPA (United Nations Population Fund) it has been estimated that by 2050, the number of persons in Asia-Pacific who are 60 years old or older is predicted to rise from 2010 to 1.3 billion. This shows that the population is quickly getting older. By 2050, over one in four people in the area will be over 60 years old. This shows how quickly the population is getting older. Another estimate says that by 2029, the area might have more than 548 million people aged 65 and more, which would be a big part of the world's senior population development. Parts of the Asia-Pacific region, like Singapore, are anticipated to become "super-aged" societies by 2030, with more than 20% of the population being 65 or older. Data modeling shows that the percentage of individuals in Asia who are 65 or older could go up a lot, to 18% by 2050.
The U.S. liquid biopsy market is undergoing a period of robust expansion. The growth is fueled by a rapidly rising cancer prevalence, for instance, in 2026, according to the data published by the American Cancer Society, it has been estimated that in 2026, the United States is expected to have about 2,114,850 new cancer cases and 626,140 cancer deaths. Furthermore, support from the U.S. Food and Drug Administration (U.S. FDA) drives the liquid biopsy market growth in the U.S. country. For instance, in August 2025, the U.S. Food and Drug Administration (FDA) has given Breakthrough Device Designation to Quest Diagnostics' Haystack MRD test, which is used to find MRD-positive patients with stage II colorectal cancer who have had curative-intent surgery and may benefit from adjuvant therapy. The new designation adds to the growing body of evidence that the Haystack MRD test is useful for both medical and drug development purposes. It also fits with a lot of research that shows ctDNA-based MRD testing could find leftover or recurring malignancy from solid tumors.
The China liquid biopsy projected to grow significantly during the upcoming years. China accounts for a major share of Asia Pacific healthcare spending which leads to higher adoption of advanced diagnostics. National programs for cancer prevention and early detection accelerates liquid biopsy market growth and adoption. Moreover, presence of domestic market players in China drives the liquid biopsy market size in Asia Pacific region. Growing number of Chinese biotech and diagnostic companies enhances local production and affordability. For instance, Burning Rock Biotech Ltd, biotech company based in Guangzhou, China is still one of the top companies in China for liquid biopsy technology. In early 2026, the company received Breakthrough Device Designation from China's NMPA for its multi-cancer early detection blood test. This shows that there are more interest and regulatory support for domestic liquid biopsy solutions. Furthermore, in December 2025, Pillar Biosciences, biotechnology company, unveiled that it would be adding China to its existing laboratory access arrangement with AstraZeneca for NGS-based kitted liquid biopsy tumor profiling. The goal of this expanded partnership is to provide quick, cheap, and therapeutically useful genetic information through liquid biopsy testing. The partnership's goal is to speed up the time it takes to get a diagnosis and make precision oncology solutions more widely available by making plasma-based tumor profiling more widely available at top clinical labs in China. The Pillar Biosciences-AstraZeneca expansion in China, which was announced in late 2025 and will continue into 2026, is a prime instance of a recent cooperation that aims to bring advanced liquid biopsy testing to China. This shows that international and local companies are working together to improve China's diagnostic industry.
Some of the major key players Biocept, Inc., Qiagen N.V., Trovagene, Inc., Janssen Global Services, LLC, MDxHealth SA, Natera, Inc., F. Hoffmann-La Roche Ltd., Silicon Biosystems, Pathway Genomics Corporation, and Sysmex Corporation.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,354.0 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 27.5% | 2033 Value Projection: | USD 7,340.0 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Biocept, Inc., Qiagen N.V., Trovagene, Inc., Janssen Global Services, LLC, MDxHealth SA, Natera, Inc., F. Hoffmann-La Roche Ltd., Silicon Biosystems, Pathway Genomics Corporation, and Sysmex Corporation. |
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Abhijeet Kale is a results-driven management consultant with five years of specialized experience in the biotech and clinical diagnostics sectors. With a strong background in scientific research and business strategy, Abhijeet helps organizations identify potential revenue pockets, and in turn helping clients with market entry strategies. He assists clients in developing robust strategies for navigating FDA and EMA requirements.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients