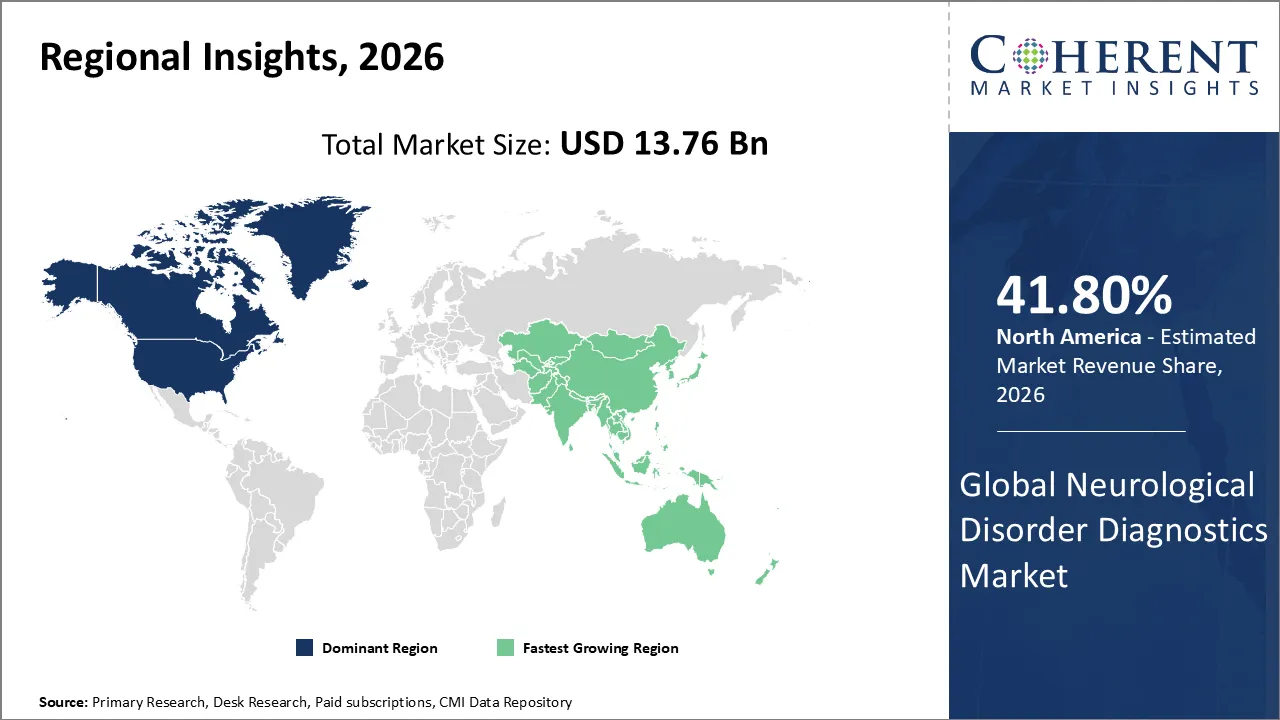
The Neurological Disorder Diagnostics Market is estimated to be valued at USD 13.76 Bn in 2026 and is expected to reach USD 15.87 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 9.1% from 2026 to 2033.
The market for neurological disorder diagnostics is growing quickly as the prevalence of neurological and neurodegenerative disorders rises and the population ages, alongside increased awareness of early detection and precision medicine. Genetic tests, advanced imaging technologies, and AI-powered diagnostic tools are making things more accurate and faster. To meet the needs of patients, hospitals, clinics, and specialized diagnostic centers are adding more services. North America and Europe are the market leaders due to they have strong healthcare systems, research funding, and new technologies. Emerging regions are progressively starting to use these solutions as well.
|
Current Events |
Description and its impact |
|
Geopolitical and Regulatory Developments |
|
|
Technological Advancements in Diagnostics |
|
|
Economic and Healthcare Infrastructure Trends |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Artificial intelligence (AI) is changing the way neurological disorders are diagnosed by making it possible to analyze complicated brain data more quickly, accurately, and objectively. AI algorithms can process an enormous amount of neuroimaging, electrophysiological, and clinical data to find small patterns that are linked to diseases like Alzheimer's, Parkinson's, epilepsy, and multiple sclerosis.
For instance, in November 2025, Royal Philips and Cortechs.ai, a leader in quantitative neuroimaging, worked together. The partnership will add Cortechs.ai's AI-powered neuroimaging analytics directly to Philips' MRI systems. This will make it possible to assess brain health faster, more objectively, and more consistently.
Imaging holds the largest market share of 53.3% in 2026. The neurological disorder diagnostics market is growing owing to improvements in MRI, CT, PET, and hybrid imaging systems that make them more accurate and allow for earlier detection. The increasing prevalence of neurological conditions and older people increases the need for clinical services. AI and advanced analytics help doctors understand results faster and make fewer mistakes. Healthcare providers focus on early diagnosis and precision medicine. They also work to improve the capabilities of hospitals and clinics and use supportive reimbursement policies to encourage more people to use these services. Ongoing research also encourages new ideas and broader use. In September 2025, GE HealthCare announced that it would buy icometrix, an AI-powered brain imaging company, to meet the growing demand for MRIs and help plan personalized treatments for neurological disorders like Alzheimer's disease.
Genetic Neurological Disorder expected to hold largest market share of 43.5% in 2026. Next-generation sequencing, whole genome, and exome analysis are making it easier to find inherited conditions, which is increasing the need for genetic tests for neurological disorders. Growing awareness of the genetic basis of diseases such as Alzheimer’s, Parkinson’s, and epilepsy drives early testing and personalized care. These tests have turned into a normal part of hospital and specialized laboratory work, and AI-powered tools help doctors understand the results. Further funding for research, more clinical trials, and the creation of gene-targeted therapies all speed up the use and development of genetic neurological diagnostics. In January 2026, Metropolis Healthcare Limited opened its Centre of Genomics in India. The center uses Illumina's NovaSeq™ X Series technology to improve precision diagnostics and genomic research.
Hospitals & Clinics acquired the prominent market share of 42.9% in 2026. Hospitals and clinics are boosting the neurological disorder diagnostics market by delivering comprehensive services, such as advanced imaging, genetic testing, and laboratory analyses within a single facility. Rising patient volumes and the urgent requirement to assess neurological conditions, including acute cases, increase demand. Multidisciplinary care teams enhance diagnostic precision and treatment planning. By adopting modern technologies, expanding clinical offerings, and catering to patient preferences for centralized care, these providers drive greater adoption. Increased awareness of early detection further strengthens market growth.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 41.80% in 2026. Widespread use of advanced imaging technologies, genetic testing, and AI-powered diagnostic tools is driving the North American neurological disorder diagnostics market. Hospitals and specialized clinics are integrating these solutions to enhance early detection and deliver personalized care. Robust healthcare infrastructure, substantial research investment, and active involvement of leading industry players are accelerating innovation and expanding market reach. Patients and healthcare providers are becoming more aware of neurological diseases, and supportive reimbursement policies are making it easier for more people to get comprehensive diagnostic services in the area. In September 2025, Beckman Coulter Diagnostics released the first fully automated Brain-derived Tau (BD-Tau) research-use-only immunoassay in the industry. It can be used on the DxI 9000 Immunoassay Analyzer and Access 2 Analyzer.
The neurological disorder diagnostics market is growing steadily due to hospitals, clinics, and diagnostic centers in Asia Pacific using advanced imaging, genetic testing, and AI-powered technologies. The growing number of neurological and neurodegenerative disorders, along with greater numbers of individuals being aware of healthcare, drives up the need for early and accurate diagnosis. To make it easier for people to get diagnostic services, governments and healthcare providers are building more infrastructure and putting funds into research and development. Emerging economies are using new technologies to improve patient care and make the region's neurological diagnostics stronger.
Healthcare providers in the US are helping the neurological disorder diagnostics market grow by using advanced imaging, genetic testing, and AI-enabled tools in their work. As neurological and neurodegenerative disorders become more prevalent, the need for accurate and early diagnosis grows. Strong healthcare systems, significant amounts of research, and active involvement from major players in the industry all help new technologies spread easily and encourage innovation. More patients and doctors are aware of comprehensive diagnostic services, and insurance and reimbursement policies that support them make them increasingly accessible across the country.
For instance, in March 2025, neuropacs™ Corp. has announced positive results from a prospective multicenter study evaluating its AI technology for diagnosing Parkinson’s disease and atypical parkinsonism.
The neurological disorder diagnostics market is growing considering hospitals, clinics, and diagnostic centers in India are using new imaging technologies, genetic testing, and AI-powered diagnostic tools. The growing number of neurological and neurodegenerative diseases, along with more people being aware of healthcare, drives up the need for early and accurate diagnosis. Access to diagnostic services is getting better through the expansion of healthcare infrastructure, government programs, and more money going into research. To improve patient care and India's neurological diagnostics capabilities, private providers and new urban centers are combining advanced technologies. In January 2026, Iswarya Hospital, a multi-specialty quaternary care facility in OMR, Chennai, opened a specialized clinic to find out exactly what causes giddiness, dizziness, vertigo, and balance problems.
The market is increasingly shaped by innovations in neuroimaging, including high-resolution MRI, PET, CT, and hybrid systems. These tools enable earlier and more accurate detection of neurological and neurodegenerative disorders. Integration of AI-assisted image analysis allows clinicians to identify subtle abnormalities, track disease progression, and improve diagnostic confidence. Hospitals and specialized clinics are upgrading equipment to meet patient demand, while research institutions leverage advanced imaging for clinical studies, driving broader adoption and expanding diagnostic capabilities.
Genetic testing and molecular diagnostics are gaining prominence in detecting hereditary and complex neurological disorders. Techniques such as whole genome sequencing, exome sequencing, and biomarker profiling enable identification of disease-causing mutations and personalized treatment planning. Clinicians and laboratories increasingly rely on these tests for early diagnosis, risk assessment, and patient stratification in clinical trials. Rising awareness of genetic contributions to conditions like Alzheimer’s, Parkinson’s, and epilepsy further boosts demand for advanced molecular diagnostic solutions.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 13.76 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.1% | 2033 Value Projection: | USD 15.87 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Siemens Healthineers, Koninklijke Philips N.V., General Electric Company, Esaote SpA, NeuroLogica Corporation, Masimo Corporation, York Instruments Ltd., Neusoft Medical Systems, and Canon Medical Systems Corporation |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Abhijeet Kale is a results-driven management consultant with five years of specialized experience in the biotech and clinical diagnostics sectors. With a strong background in scientific research and business strategy, Abhijeet helps organizations identify potential revenue pockets, and in turn helping clients with market entry strategies. He assists clients in developing robust strategies for navigating FDA and EMA requirements.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients