Autoimmune Disease Diagnostics Market Size and Forecast – 2026 – 2033
The Global Autoimmune Disease Diagnostics Market size is estimated to be valued at USD 4.8 billion in 2026 and is expected to reach USD 8.6 billion by 2033, exhibiting a compound annual growth rate (CAGR) of 8.5% from 2026 to 2033.
Global Autoimmune Disease Diagnostics Market Overview
Autoimmune disease diagnostics products include a wide range of laboratory tests and instruments designed to detect abnormal immune responses. Key products include immunoassay kits such as ELISA and chemiluminescence assays, used to identify autoantibodies linked to conditions like rheumatoid arthritis and lupus. Molecular diagnostics, including PCR-based tests, help detect genetic markers and disease susceptibility. Multiplex assays enable simultaneous testing of multiple biomarkers, improving efficiency. Laboratory instruments such as analyzers and reagents support high-throughput screening in clinical settings. Additionally, point-of-care testing devices are emerging, offering rapid results and enhancing early diagnosis and disease monitoring.
Key Takeaways
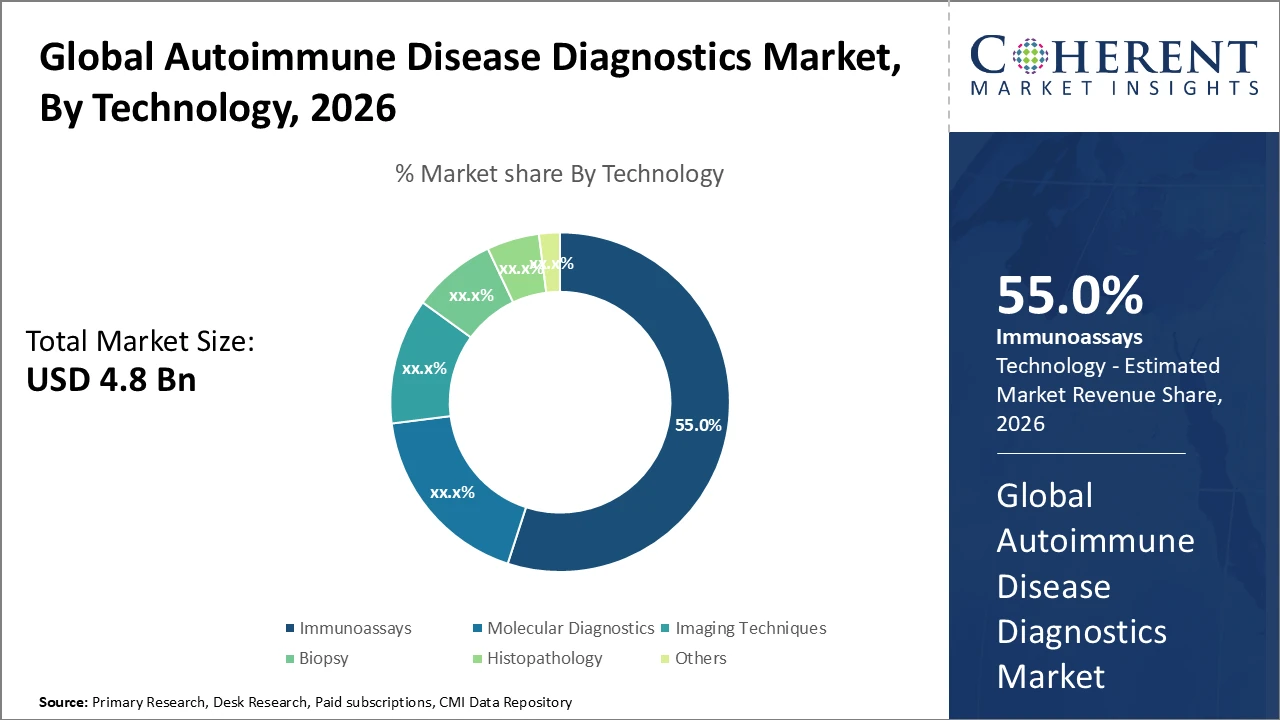
The immunoassays segment dominates the diagnostic technology market, holding about 55% share due to its high specificity and sensitivity in detecting autoimmune diseases, while molecular diagnostics is the fastest-growing subsegment driven by increased adoption of PCR and sequencing technologies
Rheumatoid arthritis represents the leading application segment because of high diagnosis rates and a growing global patient population, whereas multiple sclerosis is the fastest-growing application supported by improved detection and treatment advancements
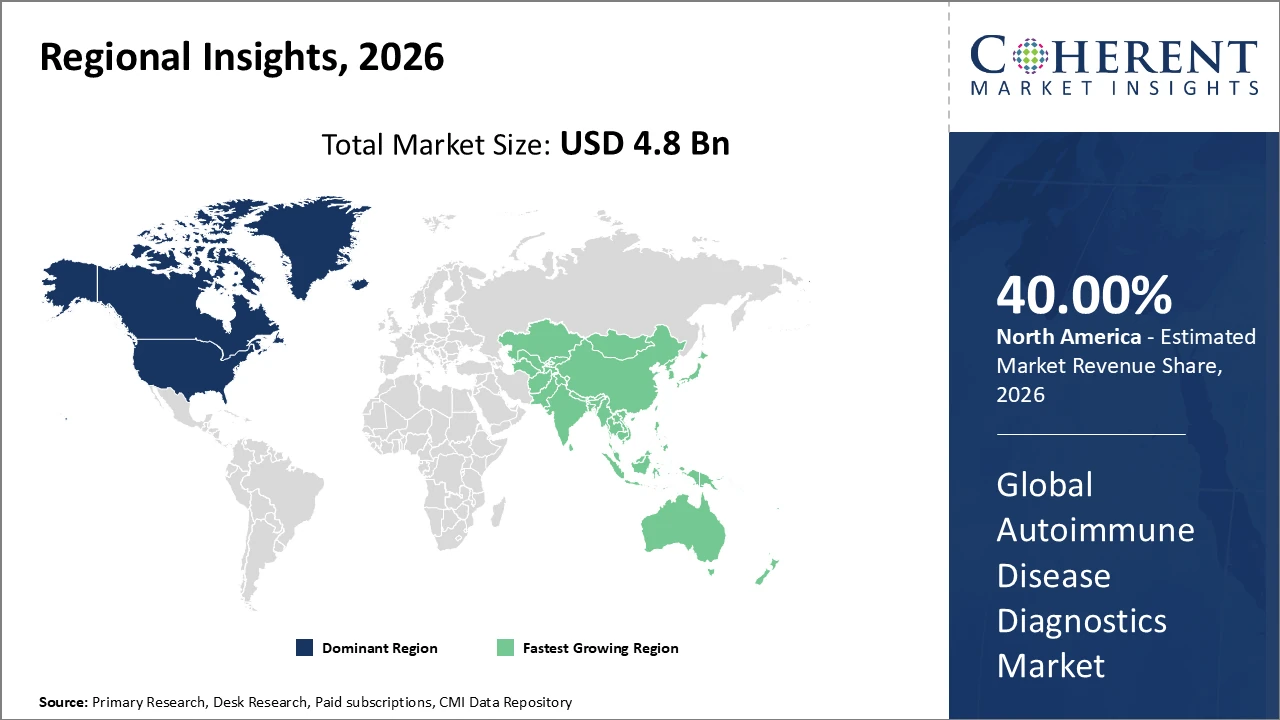
North America accounts for the largest regional share in the autoimmune disease diagnostics market, supported by strong healthcare spending and advanced diagnostic infrastructure, while Asia Pacific is the fastest-growing region with a CAGR exceeding 10%, driven by expanding healthcare access and rising disease awareness in countries like China and India
Autoimmune Disease Diagnostics Market Segmentation Analysis
To learn more about this report, Request Free Sample
Autoimmune Disease Diagnostics Market Insights, By Technology
Immunoassays dominate the market with a 55% share, driven by high sensitivity, specificity, and effectiveness in detecting autoantibodies linked to diseases such as systemic lupus erythematosus and rheumatoid arthritis. Their cost-effectiveness and standardized protocols further support widespread adoption. Molecular diagnostics is the fastest-growing segment, offering advanced genetic and proteomic insights that improve diagnostic accuracy and prognosis through techniques like PCR and gene sequencing. Imaging techniques play a supportive role in conditions like multiple sclerosis. Biopsy and histopathology remain essential for confirmation, while emerging tools such as flow cytometry and point-of-care tests are steadily gaining traction.
Autoimmune Disease Diagnostics Market Insights, By Application
Rheumatoid arthritis holds the leading market share due to its high global prevalence and the routine need for autoantibody screening. Advanced immunoassays for rheumatoid factor and anti-CCP antibodies support accurate diagnosis and monitoring. Multiple sclerosis is the fastest-growing segment, driven by rising awareness and advancements in neuroimaging and biomarker-based diagnostics that enable early detection. Systemic lupus erythematosus and type 1 diabetes show steady demand as chronic conditions requiring continuous testing. The others segment, including autoimmune thyroiditis and inflammatory bowel disease, is expanding due to increasing incidence and growing research focus on improved diagnostic approaches.
Autoimmune Disease Diagnostics Market Insights, By End User
Hospitals dominate the market due to comprehensive facilities and a high volume of patients requiring autoimmune diagnostic evaluations. Significant investments in in-house laboratories and advanced equipment enable efficient and integrated care delivery. Diagnostic laboratories are the fastest-growing segment, driven by increased outsourcing of specialized tests and adoption of high-throughput technologies, offering cost-effective and scalable services. Research institutes play a steady role in innovation and clinical trials, supporting new diagnostic developments. Point-of-care centers are expanding rapidly, improving access to quick testing in remote areas, while other segments include home-based diagnostics and emerging biotech companies developing advanced assays.
Autoimmune Disease Diagnostics Market Trends
The autoimmune disease diagnostics market is experiencing rapid adoption of AI and digital health technologies, improving early diagnosis and enabling more personalized treatment approaches
In 2025, there was a significant rise in the clinical use of AI-driven diagnostic tools in the U.S., enhancing diagnostic accuracy by more than 15%
Multiplex assays are increasingly integrated into routine screening protocols, allowing simultaneous detection of multiple autoantibodies and improving testing efficiency since 2024
North America holds over 40% of the market share, supported by advanced healthcare systems, strong R&D investments, and high disease awareness
Asia Pacific is the fastest-growing region, with a CAGR exceeding 10%, driven by expanding healthcare infrastructure, increased government support, and growing patient populations in China and India
Major companies such as Roche Diagnostics and Abbott Laboratories are expanding their regional presence, leveraging market opportunities to drive growth
Autoimmune Disease Diagnostics Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Autoimmune Disease Diagnostics Market Analysis and Trends
North America dominates the autoimmune disease diagnostics market due to advanced diagnostic infrastructure, high healthcare expenditure, and early adoption of innovative technologies. The U.S. healthcare system benefits from favorable reimbursement policies and a strong biopharmaceutical presence, enabling rapid integration of novel diagnostic solutions. Leading companies such as Thermo Fisher Scientific and Becton, Dickinson and Company drive innovation through advanced immunoassays and testing platforms. Rising prevalence of autoimmune diseases and strong clinical research collaborations further support market growth. These factors collectively reinforce North America’s leading position, allowing it to account for more than 40% of the global market share.
Asia Pacific Autoimmune Disease Diagnostics Market Analysis and Trends
Asia Pacific is the fastest-growing region in the autoimmune disease diagnostics market, with a CAGR exceeding 10%, driven by improving healthcare access and rising awareness of autoimmune conditions. Expanding diagnostic infrastructure in emerging economies such as China and India supports broader testing availability. Government initiatives focused on chronic disease management and increasing healthcare investments further accelerate growth. The region benefits from cost-efficient manufacturing and strong collaborations between local and global companies. Additionally, the expansion of point-of-care diagnostic services and rapid urbanization contribute to increased demand, strengthening the region’s overall market outlook and long-term growth potential.
Autoimmune Disease Diagnostics Market Outlook for Key Countries
USA Autoimmune Disease Diagnostics Market Analysis and Trends
The United States market leads the autoimmune disease diagnostics sector due to strong technological investments, advanced healthcare infrastructure, and robust research capabilities. In 2025, FDA approvals of innovative multiplex diagnostic platforms and companion diagnostic kits enhanced early disease detection and enabled personalized treatment approaches, boosting market growth. Major companies such as Abbott Laboratories and Roche Diagnostics maintain extensive R&D facilities and strategic collaborations, driving continuous innovation and deeper market penetration. Additionally, government support through increased funding for autoimmune disease research and expanded reimbursement policies further strengthens the market, ensuring sustained growth and leadership in the global landscape.
Germany Autoimmune Disease Diagnostics Market Analysis and Trends
Germany’s autoimmune disease diagnostics market is driven by a strong healthcare system, advanced laboratory infrastructure, and high adoption of innovative diagnostic technologies. The country benefits from well-established reimbursement policies and a strong focus on early disease detection and precision medicine. Increasing prevalence of autoimmune disorders, coupled with an aging population, supports steady demand for advanced testing solutions such as immunoassays and molecular diagnostics. Germany’s robust biomedical research ecosystem and collaborations between academic institutions and diagnostic companies further enhance innovation. Additionally, the presence of leading European diagnostics players and growing investments in digital health are shaping positive market trends and sustained growth.
Analyst Opinion
The rising prevalence of autoimmune diseases is a key demand driver, with the National Institutes of Health reporting a 12% increase in diagnosed cases in the U.S. between 2024 and 2026 due to expanded screening efforts, boosting demand for advanced diagnostics
On the supply side, production capacity for immunodiagnostic kits is expanding, with global manufacturing output of ELISA kits increasing by 15% in 2025 to meet growing laboratory demand
Global trade is increasing, with exports of autoimmune diagnostic reagents from Asia-Pacific rising by 18% in 2026, supported by cost-efficient production and improved quality standards, strengthening supply in North America and Europe
Pricing trends are improving accessibility, as multiplex immunoassays introduced in 2024 reduced per-test costs by nearly 20%, driving wider adoption in emerging markets and outpatient diagnostic settings
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 4.8 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.5% | 2033 Value Projection: | USD 8.6 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Abbott Laboratories, Roche Diagnostics, Siemens Healthineers, Thermo Fisher Scientific, Bio-Rad Laboratories, Danaher Corporation, Luminex Corporation, Hologic Inc., Cellex Inc., QIAGEN N.V. | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Autoimmune Disease Diagnostics Market Growth Factors
The rising incidence of autoimmune diseases globally is a key growth driver, with cases increasing at a compounded annual rate exceeding 5% between 2024 and 2026 due to genetic and environmental factors. Advancements in molecular and immunoassay diagnostics have enabled earlier, more accurate detection, expanding testing capabilities and improving patient outcomes. Adoption of point-of-care and home-based testing is also increasing, with a 22% rise in use across outpatient and home care settings in 2026, improving accessibility. Additionally, supportive government policies and expanded healthcare reimbursement in regions like the U.S. and Europe are enhancing patient access and driving sustained market growth.
Autoimmune Disease Diagnostics Market Development
In January 2026, 10x Genomics partnered with Brigham and Women's Hospital to study blood samples from patients with rheumatoid arthritis, systemic lupus erythematosus, and giant cell arteritis, aiming to identify immune signatures linked to disease activity, remission, and treatment response.
Key Players
Leading Companies of the Market
Abbott Laboratories
Roche Diagnostics
Siemens Healthineers
Thermo Fisher Scientific
Bio-Rad Laboratories
Luminex Corporation
QUANGEN N.V.
Hologic Inc.
Cellex Inc.
Danaher Corporation
Several leading companies are adopting strategic collaborations and product innovation to drive growth in the autoimmune disease diagnostics market. In 2025, Roche Diagnostics strengthened its position through targeted acquisitions, expanding its portfolio with advanced molecular diagnostic assays for improved disease detection. Abbott Laboratories focused on developing multiplex panels that streamline testing across multiple autoimmune conditions, reducing turnaround time and costs, contributing to a 10% revenue increase in 2026. Thermo Fisher Scientific invested in AI-based diagnostic software to enhance accuracy and support personalized treatment approaches. These strategies collectively position these companies as key innovators in the evolving diagnostics landscape.
Autoimmune Disease Diagnostics arket Future Outlook
The future outlook for the autoimmune disease diagnostics market is highly positive, driven by increasing disease prevalence, technological advancements, and growing demand for early and accurate diagnosis. Expansion of molecular diagnostics, AI-enabled testing platforms, and multiplex immunoassays will significantly enhance diagnostic precision and efficiency. Rising adoption of point-of-care and home-based testing will improve accessibility, especially in emerging economies. Continued investments in research and development, along with supportive government healthcare policies, will further accelerate market growth. Integration of personalized medicine approaches and digital health tools is expected to transform disease management, making diagnostics more predictive, preventive, and patient-centric over the coming years.
Autoimmune Disease Diagnostics Market Historical Analysis
The historical analysis of the autoimmune disease diagnostics market shows steady growth driven by rising awareness and improved diagnostic capabilities over the past decade. Early adoption of immunoassays such as ELISA and ANA testing laid the foundation for routine autoimmune screening. Between 2015 and 2020, advancements in molecular diagnostics and automation significantly improved accuracy and testing speed. From 2020 onward, increased prevalence of autoimmune diseases and expanded healthcare access further boosted demand. The introduction of multiplex assays and digital laboratory systems enhanced efficiency, while growing investments in R&D and healthcare infrastructure supported consistent market expansion across developed and emerging regions.
Sources
Primary Research Interviews:
Immunology specialists, clinical pathologists, and laboratory medicine experts involved in autoimmune disease diagnosis and biomarker discovery
Hospital procurement managers, diagnostic laboratory directors, and R&D heads in clinical diagnostics companies using immunoassay and molecular diagnostic platforms
Executives and technical experts from diagnostic companies engaged in development of autoimmune testing kits, multiplex assays, and point-of-care diagnostic solutions
Magazines:
Nature Medicine – Advances in autoimmune disease research, biomarkers, and diagnostic innovations
Clinical Laboratory News – Developments in immunoassays, laboratory automation, and diagnostic testing trends
Fierce Biotech – Industry updates on diagnostic companies, product launches, and clinical testing advancements
The Scientist – Research breakthroughs in immunology and autoimmune disease detection technologies
Journals:
Journal of Autoimmunity – Research on autoimmune mechanisms and diagnostic biomarkers
Clinical Chemistry – Advances in laboratory diagnostics and immunoassay development
Nature Reviews Immunology – Insights into immune system disorders and diagnostic innovations
Frontiers in Immunology – Studies on autoimmune disease detection and molecular diagnostics
Newspapers:
The New York Times – Healthcare innovation, disease awareness, and diagnostic advancements
Financial Times – Healthcare sector investments and diagnostic industry growth trends
Reuters Health – Updates on medical diagnostics, regulatory approvals, and healthcare technology
Bloomberg Health – Market analysis of diagnostic companies and healthcare industry developments
Associations:
American Association for Clinical Chemistry (AACC) – Standards and advancements in clinical diagnostics
World Health Organization (WHO) – Global autoimmune disease burden and diagnostic guidelines
European Federation of Clinical Chemistry and Laboratory Medicine (EFLM) – Laboratory medicine standards and diagnostic practices
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients