Cardiac Marker Testing Devices Market Size and Forecast – 2026 – 2033
The Global Cardiac Marker Testing Devices Market size is estimated to be valued at USD 3.8 billion in 2026 and is expected to reach USD 7.2 billion by 2033, exhibiting a compound annual growth rate (CAGR) of 9.3% from 2026 to 2033.
Global Cardiac Marker Testing Devices Market Overview
The Cardiac Marker Testing Devices market includes a range of diagnostic tools used for detecting and monitoring cardiac biomarkers, critical for the early diagnosis of cardiovascular diseases. Key products comprise immunoassay analyzers, point-of-care testing (POCT) devices, enzyme-linked immunosorbent assay (ELISA) kits, and automated chemiluminescence platforms, enabling rapid and accurate detection of markers such as troponin, CK-MB, and myoglobin. Consumables like test strips, reagents, and calibrators support device functionality. Innovations focus on improving sensitivity, reducing turnaround time, and enabling bedside testing.
Key Takeaways
The High-Sensitivity Cardiac Troponin Testing Devices segment dominates the market due to advanced sensitivity and rapid diagnostic capability essential for acute care.
Hospitals are the leading end-user segment, generating the highest revenue through widespread adoption in emergency cardiac care.
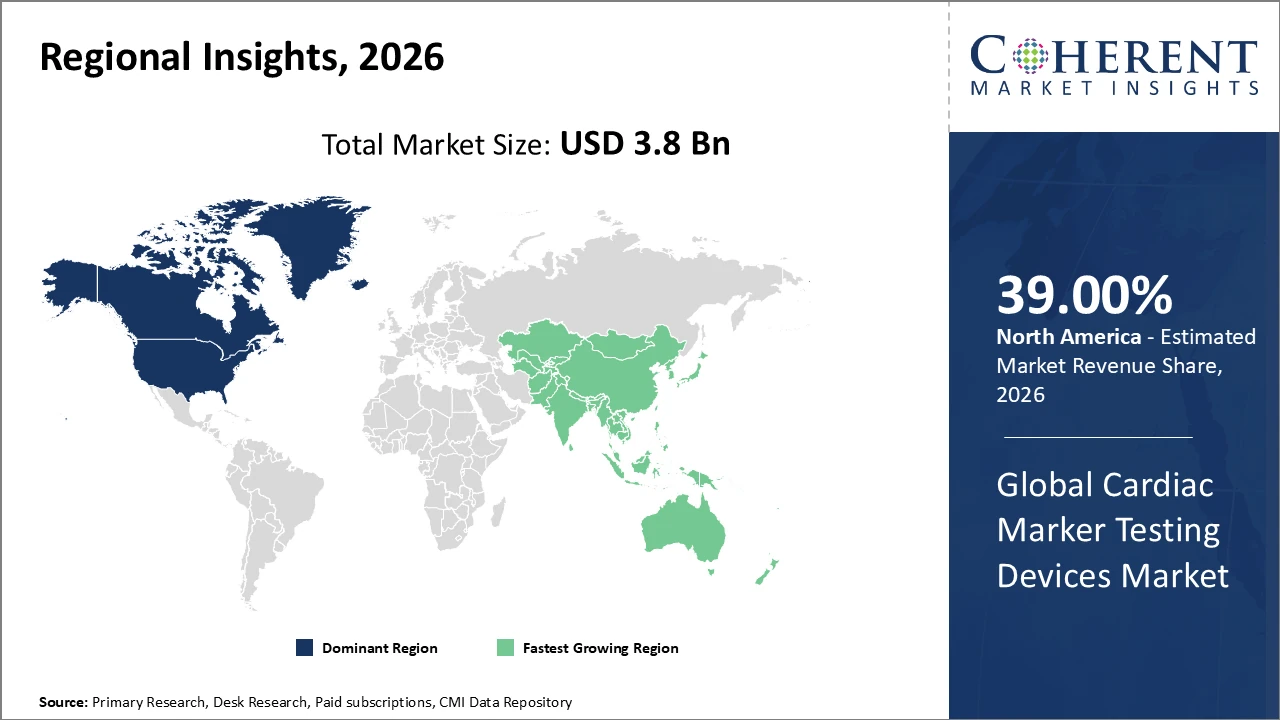
North America holds a dominant regional market share of over 37%, supported by a strong industry presence and comprehensive reimbursement infrastructure.
Asia Pacific exhibits the highest CAGR, driven by expanding healthcare ecosystems and increasing prevalence of cardiovascular diseases.
Europe is witnessing growth through the integration of point-of-care diagnostic technologies, reshaping market dynamics and creating new opportunities.
Cardiac Marker Testing Devices Market Segmentation Analysis
To learn more about this report, Request Free Sample
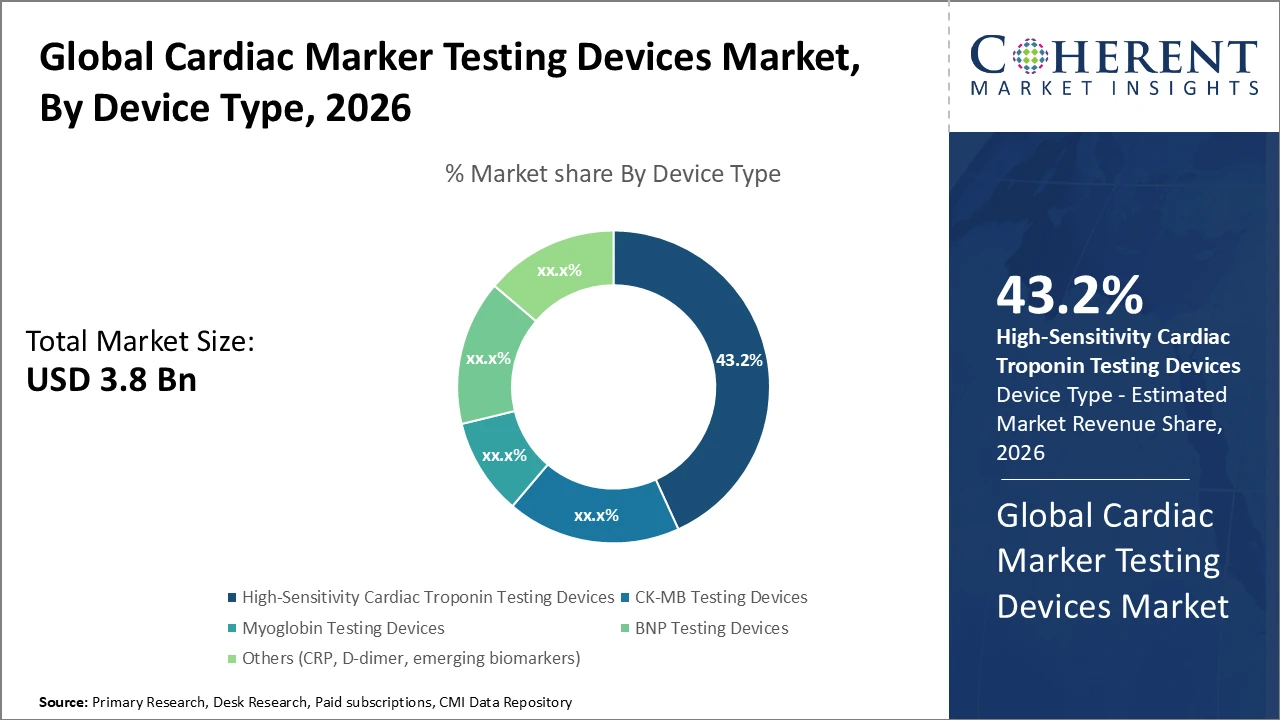
Cardiac Marker Testing Devices Market Insights, By Device Type
High-Sensitivity Cardiac Troponin Testing Devices dominate the Cardiac Marker Testing Devices market due to their superior sensitivity and specificity, allowing earlier and more accurate detection of acute coronary syndromes and meeting critical clinical needs. Integration with hospital information systems has improved workflow efficiency, boosting revenue for market players. BNP Testing Devices are the fastest-growing subsegment, driven by their increasing role in chronic heart failure diagnosis and prognosis.
Cardiac Marker Testing Devices Market Insights, By End User Industry
Hospitals hold the largest share in the Cardiac Marker Testing Devices market, benefiting from advanced infrastructure and multidisciplinary cardiology departments that emphasize rapid cardiac marker evaluation. Rising inpatient admissions for cardiovascular diseases, along with routine use of device testing in emergency departments, support sustained growth. Point-of-Care Testing facilities are the fastest-growing subsegment, driven by increasing demand for rapid bedside diagnostics in both urban and rural areas. Diagnostic laboratories, handling high-throughput testing, show steady growth, while ambulatory surgical centers and other specialized facilities account for smaller shares. Hospitals lead due to volume and clinical reliance, whereas point-of-care growth reflects decentralized diagnostic needs.
Cardiac Marker Testing Devices Market Insights, By Technology
Immunoassay-based devices dominate the Cardiac Marker Testing Devices market, driven by their proven accuracy, cost-effectiveness, and widespread clinical adoption, supported by decades of technological refinement. Biosensor-based devices are the fastest-growing subsegment, propelled by advances in nanotechnology and integration with digital monitoring systems, enabling point-of-care and wearable applications for continuous cardiac biomarker detection. Microfluidic and molecular diagnostic devices cater to specialized needs requiring precision and multiplexing but occupy smaller market shares.
Cardiac Marker Testing Devices Market Trends
The Cardiac Marker Testing Devices market is increasingly shaped by digital convergence and precision diagnostics.
Adoption of AI-enabled devices enhances diagnostic accuracy and predictive analytics, improving patient management.
In 2025, AI-integrated troponin devices showed a 15% increase in early myocardial infarction detection in clinical settings.
Decentralized testing through portable and wearable devices is expanding, enabling continuous monitoring and reducing hospital stays.
Asia Pacific is experiencing rapid market growth, driven by government healthcare investments and rising urban populations with cardiovascular risks.
China’s local manufacturers increased device output by 25% in 2025 to meet regional demand.
Cardiac Marker Testing Devices Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Cardiac Marker Testing Devices Market Analysis and Trends
In North America, the Cardiac Marker Testing Devices market is dominated by a well-established healthcare ecosystem, strong presence of key industry players, and supportive reimbursement frameworks. The United States accounts for the prominent market share, driven by widespread adoption of advanced diagnostic technologies and continuous innovation from leading companies. Government initiatives aimed at reducing cardiovascular mortality, along with substantial investments in research and development, further bolster market growth. High patient awareness, the prevalence of cardiovascular diseases, and the integration of digital and AI-enabled diagnostics contribute to North America’s leadership, ensuring robust revenue generation and sustained competitive advantage in the global market.
Asia Pacific Cardiac Marker Testing Devices Market Analysis and Trends
Asia Pacific is the fastest-growing region in the Cardiac Marker Testing Devices market, with a CAGR exceeding 11%, driven by rising prevalence of cardiovascular diseases, expanding healthcare infrastructure, and increasing disposable income. Key markets such as India and China are evolving rapidly, supported by regulatory reforms, improved healthcare access, and growing middle-class demand for advanced diagnostics. The presence of local manufacturers offering cost-competitive devices further strengthens market adoption. Government health initiatives promoting early detection and preventive care, along with investments in hospitals and diagnostic centers, continue to propel business growth, making Asia Pacific a critical region for market expansion and future opportunities.
Cardiac Marker Testing Devices Market Outlook for Key Countries
USA Cardiac Marker Testing Devices Market Analysis and Trends
The U.S. cardiac marker testing devices market leads globally, driven by a high prevalence of cardiovascular diseases and advanced healthcare infrastructure. Key companies, including Abbott and Roche, have established strategic installations in hospitals and ambulatory centers, promoting adoption of high-sensitivity troponin assays and point-of-care platforms. In 2025, U.S. hospitals reported a 30% increase in rapid cardiac marker test utilization, contributing to reduced misdiagnosis of myocardial infarction. Supportive regulatory frameworks encourage innovation, while favorable reimbursement policies incentivize early diagnostics. Combined with ongoing R&D and hospital investments, these factors sustain market growth and reinforce the U.S. position as a global leader.
Germany Cardiac Marker Testing Devices Market Analysis and Trends
The Germany Cardiac Marker Testing Devices market is marked by steady growth, driven by advanced healthcare infrastructure, high patient awareness, and strong adoption of minimally invasive cardiac diagnostics. Hospitals dominate device utilization, particularly for high-sensitivity troponin testing and point-of-care platforms, supported by well-established cardiology departments. Immunoassay-based devices remain the primary technology, while biosensor and microfluidic innovations are gradually gaining traction. Rising prevalence of cardiovascular diseases, coupled with government initiatives promoting early detection and preventive care, further fuels market expansion. Investments in hospital upgrades, digital integration, and outpatient diagnostic centers enhance procedural efficiency, positioning Germany as a key market within Europe for cardiac marker testing devices.
Analyst Opinion
Increasing adoption of high-sensitivity cardiac troponin assays is driving market growth, with a 42% rise in hospital implementation in 2025, improving diagnostic accuracy and enabling earlier detection of myocardial infarctions while reducing hospital stays and costs.
Expansion of point-of-care testing (POCT) devices is accelerating across emergency and ambulatory settings, with over 35% of cardiology clinics in Europe adopting POCT in 2024, supported by demand for faster turnaround times and efficient workflows.
Regional import-export dynamics are shaping supply chains, with North American manufacturers accounting for 48% of exports in 2026, while Asia Pacific shows strong import growth of 28% year-over-year in 2025 due to healthcare infrastructure investments.
Cardiac marker testing applications are diversifying beyond acute coronary syndrome to include perioperative monitoring and heart failure management, with a 15% increase in outpatient usage reported in 2026, expanding market opportunities.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 3.8 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.3% | 2033 Value Projection: | USD 7.2 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Abbott Laboratories, Roche Diagnostics, Siemens Healthineers, Danaher Corporation, Quidel Corporation, Siemens AG, Werfen, Genesis Diagnostics, Luminex Corporation, Ortho Clinical Diagnostics | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Cardiac Marker Testing Devices Market Growth Factors
The rising incidence of cardiovascular diseases globally is a primary driver of the Cardiac Marker Testing Devices market, with the World Health Organization reporting a 12% increase in cardiac-related hospital admissions between 2024 and 2026. Growing government healthcare expenditure, particularly in emerging economies, has accelerated the adoption of advanced diagnostic technologies aimed at reducing cardiovascular mortality. Technological advancements in assay sensitivity and portability have enabled wider deployment across hospitals, clinics, and point-of-care settings. Increasing patient awareness and demand for rapid diagnostics further boost device utilization, while favorable reimbursement policies in developed regions support sustained market growth and improved access to testing.
Cardiac Marker Testing Devices Market Development
In October 2023, Mindray launched two high-sensitivity cardiac biomarkers—hs-cTnI and NT-proBNP—strengthening its portfolio for diagnosing and managing cardiovascular diseases.
Key Players
Leading Companies of the Market
Abbott Laboratories
Roche Diagnostics
Siemens Healthineers
Danaher Corporation
Siemens AG
Werfen
Genesis Diagnostics
Luminex Corporation
Quidel Corporation
Ortho Clinical Diagnostics
Notable strategies in the Cardiac Marker Testing Devices market include Roche’s expansion into portable cardiac troponin testing devices, which led to a 20% increase in point-of-care market penetration in 2025. Additionally, Abbott’s collaboration with ambulatory care centers to integrate its i-STAT platform accelerated the adoption of home-based cardiac marker testing, resulting in a 15% rise in device adoption across North America. These strategic initiatives highlight the growing emphasis on decentralized diagnostics, improved accessibility, and technological innovation, enabling companies to strengthen their market presence and respond effectively to the increasing demand for rapid and accurate cardiac testing solutions.
Cardiac Marker Testing Devices Market Future Outlook
The Cardiac Marker Testing Devices market is expected to experience robust growth driven by rising cardiovascular disease prevalence, increasing demand for rapid diagnostics, and continuous technological advancements. High-sensitivity assays and AI-enabled diagnostic platforms will enhance accuracy and enable early disease detection. The expansion of point-of-care and wearable devices will support decentralized testing and real-time monitoring. Emerging markets in Asia Pacific and Latin America will contribute significantly due to improving healthcare infrastructure and growing awareness. Additionally, favorable reimbursement policies and government initiatives promoting early diagnosis will sustain adoption. Ongoing innovation and strategic collaborations will further strengthen market expansion and competitive positioning globally.
Cardiac Marker Testing Devices Market Historical Analysis
The historical analysis of the Cardiac Marker Testing Devices market reflects steady growth driven by rising cardiovascular disease burden and advancements in diagnostic technologies. Between 2015 and 2025, immunoassay-based systems dominated due to their reliability and widespread clinical adoption, while high-sensitivity troponin assays gained prominence for early detection of myocardial infarction. Hospitals remained the primary end users, supported by increasing emergency care needs and diagnostic integration. The emergence of point-of-care testing devices improved turnaround times and expanded accessibility. Developed regions such as North America and Europe led market growth, while gradual healthcare improvements in emerging economies contributed to expanding adoption and overall market development.
Sources
Primary Research Interviews:
Executives and operations managers at cardiac diagnostic device manufacturing companies
Cardiologists, clinical laboratory managers, and hospital procurement specialists
Technology providers for immunoassay platforms, biosensors, and point-of-care diagnostic systems
Magazines:
Medical Device and Diagnostic Industry (MD+DI) – Innovations in Cardiac Diagnostics
Clinical Laboratory News – Advances in Biomarker Testing
Healthcare Technology Today – Trends in Point-of-Care Diagnostics
Pharmaceutical Technology – Diagnostic Device Developments
Journals:
Journal of the American College of Cardiology – Cardiac Biomarker Research
Clinical Chemistry – Advances in Diagnostic Assays
European Heart Journal – Cardiovascular Diagnostics and Outcomes
Journal of Clinical Laboratory Analysis – Biomarker Testing Technologies
Newspapers:
The Wall Street Journal – Healthcare Technology and Diagnostics Market
Financial Times – Global Medical Device Industry Trends
The New York Times – Public Health and Cardiovascular Disease Updates
Chemical & Engineering News – Diagnostic Innovation and Industry Developments
Associations:
American Heart Association (AHA) – Cardiac Biomarker Guidelines and Research
American Association for Clinical Chemistry (AACC) – Diagnostic Testing Standards
European Society of Cardiology (ESC) – Cardiac Marker Testing Guidelines
World Health Organization (WHO) – Global Cardiovascular Health Initiatives
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients