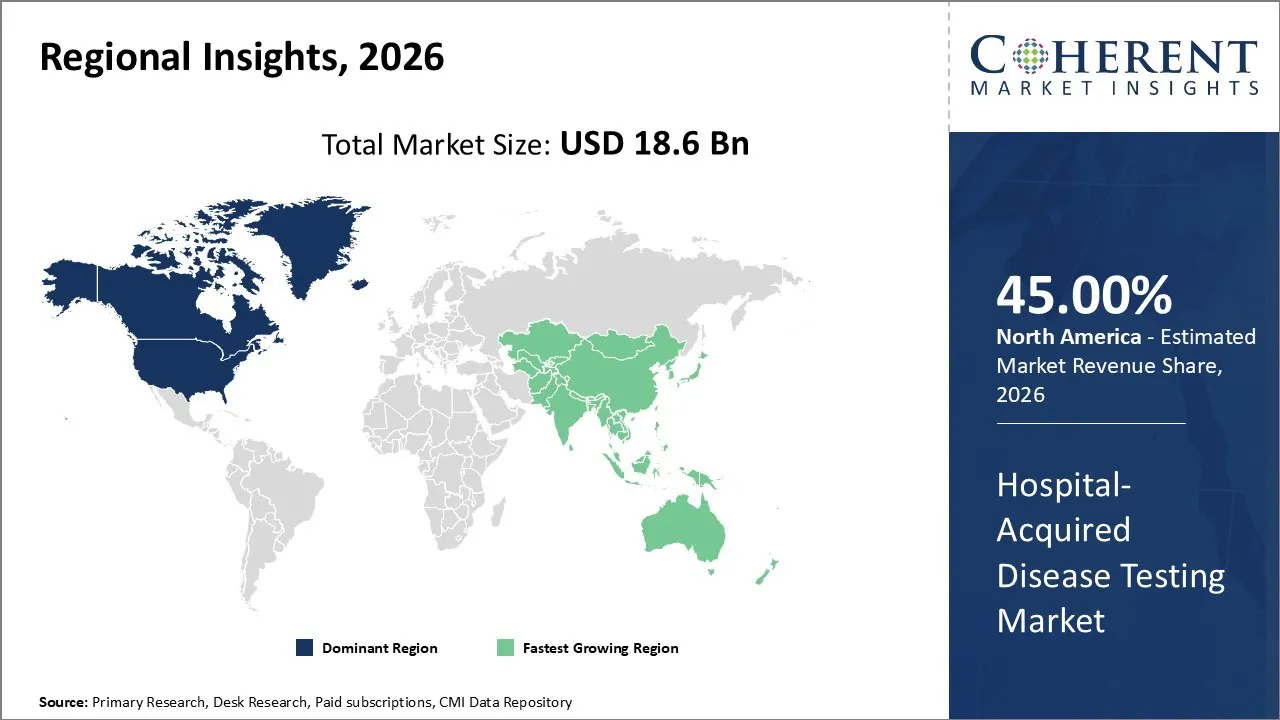
The hospital-acquired disease testing market was valued at USD 18.6 Bn in 2026 and is forecast to reach a value of USD 49.4 Bn by 2033 at a CAGR of 15.5% between 2026 and 2033.
The hospital-acquired disease (HAD) testing market is primarily driven by the rising prevalence of healthcare-associated infections (HAIs), including bloodstream infections, pneumonia, and surgical site infections, which increase the need for timely and accurate diagnostics. Growing antimicrobial resistance further accelerates demand for advanced testing solutions. Additionally, an aging population and higher incidence of chronic diseases lead to longer hospital stays, increasing infection risk. Technological advancements such as molecular diagnostics and PCR-based methods improve detection speed and accuracy, boosting adoption. Increasing healthcare expenditure, regulatory mandates for infection control, and greater awareness of patient safety and hospital hygiene also significantly contribute to market growth.
|
Current Event |
Description and its Impact |
|
Technological Innovations in Rapid Diagnostics |
|
|
Market Expansion along with Strategic Partnerships |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of test type, the urinary tract infection segment is expected to lead the market with 37% share in 2026, owing primarily to the prevalence, impact, and clinical complexity of healthcare-associated urinary tract infections, which continue to be a significant burden in hospitals worldwide. Urinary Tract Infections are among the most common hospital-acquired infections, with a major association with in dwelling catheter use, which remains widespread in clinical settings.
To learn more about this report, Request Free Sample
North America is expected to dominate the Hospital-Acquired Disease Testing market with 45% share in 2026, owing to several region-specific attributes that underscore its leadership position. A key factor driving this dominance is the well-established healthcare infrastructure combined with substantial investments in advanced diagnostic technologies. The U.S. Centers for Disease Control and Prevention (CDC) reports that hospital-associated infections affect approximately 1 in 31 hospital patients on any given day in the United States, which has escalated the urgency for comprehensive testing and monitoring programs in healthcare facilities across North America.
For instance, in June 2025, bioMerieux, a leading company in medical testing, bought Day Zero Diagnostics, a U.S. company that uses genome sequencing and AI to fight antibiotic-resistant infections. This deal will aid bioMérieux improve rapid testing and next-generation sequencing, showing its commitment to better healthcare and smarter use of antibiotics.
Asia Pacific is expected to exhibit the fastest growth, due to a combination of expanding healthcare infrastructure, increasing governmental focus on infection control, as well as rising awareness around antibiotic stewardship and rapid pathogen detection technologies. Countries such as China, India, Japan, and South Korea are experiencing high investments in hospital modernization and diagnostic lab expansions aimed at combating the growing threat of nosocomial infections exacerbated by high patient turnover and urbanization.
For instance, India’s National Centre for Disease Control (NCDC) recently reported that enhanced surveillance programs implemented in major metropolitan hospitals have resulted in increased early detection of hospital-acquired pathogens via PCR-based assays and cartridge-based nucleic acid amplification tests (CBNAAT).
The U.S. contributes the highest share in the Hospital-Acquired Disease (HAD) Testing market in the region owing to its well-established healthcare infrastructure, high adoption of advanced diagnostic technologies, as well as stringent regulatory frameworks that emphasize infection control. The U.S. focuses on preventing infections in hospitals, with help from federal agencies like the CDC and NIH, which fund research and promote the best ways to fight hospital-acquired infections.
For instance, in 2023, the CDC launched several updated guidelines on the testing and management of multidrug-resistant organisms (MDROs), which propelled hospitals and labs to adopt sophisticated molecular diagnostics and rapid testing platforms to curb the spread of HAIs.
China contributes the highest share in the Hospital-Acquired Disease (HAD) Testing Market within the region owing to several critical factors. China leads the market because it has a large healthcare system and the government is actively working to prevent infections in hospitals. Authorities have invested a lot in programs and better diagnostic tools to fight hospital-acquired infections.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 18.6 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 15.5% | 2033 Value Projection: | USD 49.4 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Luminex Corporation, Diatherix Laboratories, Life Technologies, Cantel Medical Corporation, Meridian Biosciences Inc, Nordion Inc, Qiagen GmbH, F.Hoffmann-La Roche Ltd., Biomerieux, and Cepheid |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The rising incidence of healthcare-associated infections (HAIs) is majorly driving the expansion of the hospital-acquired disease testing market, as healthcare systems globally are under high pressure to monitor, detect, and control these infections promptly. HAIs, which include infections such as bloodstream infections, surgical site infections, and pneumonia, present considerable challenges to patient safety and healthcare quality.
Stringent regulatory requirements focused on infection control and patient safety are highly steering the growth of the hospital-acquired disease (HAD) testing market. Hospitals and healthcare facilities globally face mounting pressure to adhere to rigorous standards that prevent the transmission of infections within clinical environments. Regulatory bodies such as the U.S. Centers for Disease Control and Prevention (CDC) as well as the World Health Organization (WHO) target strict infection prevention protocols, including comprehensive screening and testing procedures for pathogens responsible for hospital-acquired infections (HAIs).
The development of point-of-care (POC) testing devices for rapid infection detection represents a major opportunity within the hospital-acquired infection (HAI) testing market due to the instant need for timely diagnosis and intervention to curb the spread of infections. Hospital-acquired infections, such as bloodstream infections, surgical site infections, ventilator-associated pneumonia, etc., pose major challenges globally, usually leading to prolonged hospital stays, increased healthcare costs, and higher patient mortality.
The hospital-acquired disease (HAD) testing market is experiencing significant momentum driven by rising awareness about healthcare-associated infections and the critical need for early diagnosis and containment. Rising incidences of multidrug-resistant organisms (MDROs) and pathogens such as Clostridium difficile, MRSA, and VRE are compelling healthcare institutions to adopt advanced diagnostic solutions.
Fast and accurate tests help treat patients sooner, make them healthier, and reduce stress on hospitals. Additionally, strict regulations and guidelines by health authorities focus routine surveillance, further bolstering demand for HAD testing.
However, innovation in molecular diagnostics, point-of-care testing, as well as automation presents substantial opportunities to improve testing efficiency and accessibility. Emerging technologies like next-generation sequencing (NGS) are poised to revolutionize pathogen identification as well as antimicrobial resistance detection, thereby expanding market surge.
Geographically, North America dominates the HAD testing market owing to robust healthcare infrastructure, higher adoption of advanced diagnostics, as well as favorable reimbursement policies. Conversely, the Asia-Pacific region is the fastest-growing market segment, fueled by rising healthcare investments, improving infrastructure, as well as increasing prevalence of hospital-acquired infections.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients