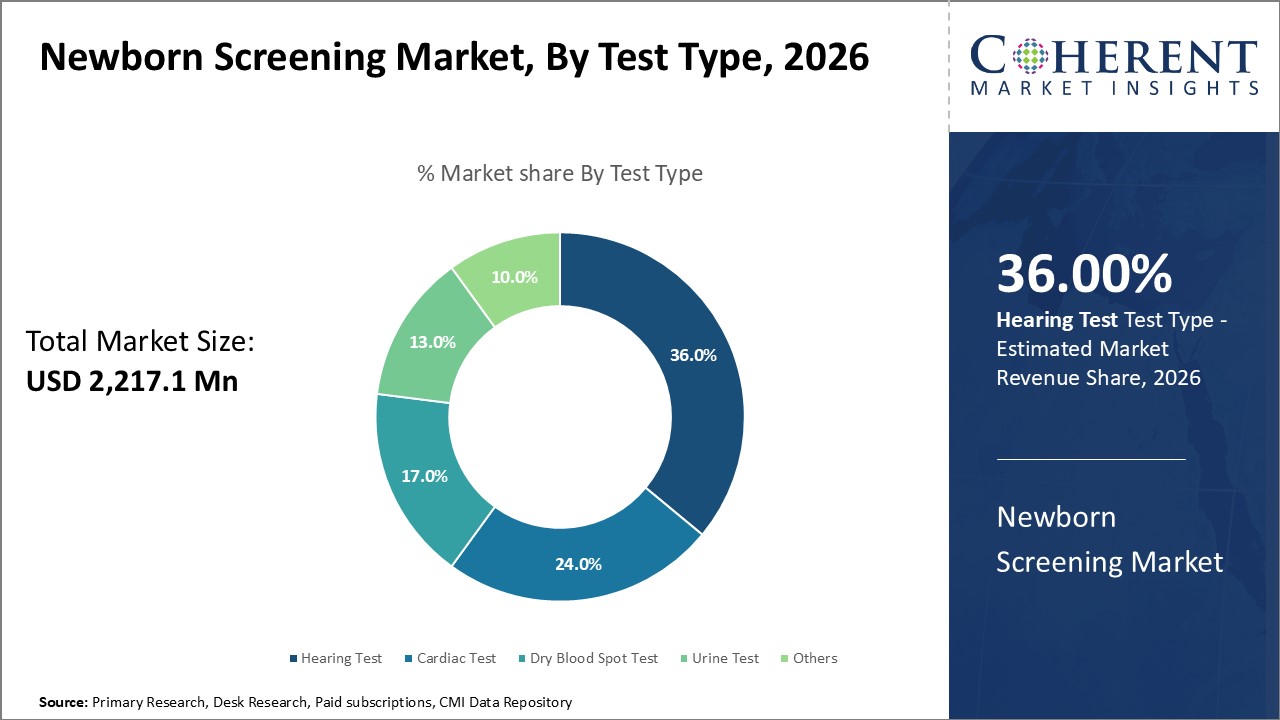
Newborn Screening Market is estimated to be valued at USD 2,217.1 Mn in 2026 and is expected to reach USD 4,603.1 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of11.0% from 2026 to 2033.
The newborn screening market demand continues to grow as more people recognize the importance of early disease detection. Governments are expanding healthcare programs, and rising global birth rates are increasing demand for these services. Technological advancements in genetic and metabolic testing now allow for more accurate and efficient diagnoses. Hospitals, diagnostic labs, and public health agencies actively lead the implementation of screening programs. Strong policy support and well-developed healthcare systems, especially in advanced regions, are driving the widespread adoption of newborn screening.
|
Current Events |
Description and its impact |
|
Regulatory and Policy Developments |
|
|
Economic and Healthcare Infrastructure Factors |
|
|
Market Consolidation and Industry Movements |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
AI plays a crucial role in modern newborn screening by enabling rapid, accurate, and scalable analysis of genomic data. It automates the interpretation of complex genetic information, allowing for early detection of rare and inherited conditions. Machine learning algorithms can predict disease risk and prioritize findings, helping clinicians make timely and informed decisions. AI also reduces the manual workload, making it feasible to screen millions of newborns efficiently. As more data is collected, AI systems continue to improve, expanding the range of detectable conditions. This early insight can lead to prompt interventions, significantly improving long-term health outcomes for affected infants.
BeginNGS leverages advanced genome sequencing and AI-driven tools to deliver affordable, scalable newborn genomic screening. By automating the interpretation of disease risk from genetic data, it can efficiently serve all 3.7 million babies born annually in the U.S.
Instruments acquired the prominent share of 45.2% in 2026. Healthcare providers are increasingly adopting instruments in the newborn screening market to ensure fast, accurate, and high-throughput detection of congenital and metabolic disorders. Technological advancements in tools like mass spectrometry, PCR, and next-generation sequencing are improving the speed and reliability of screenings. Rising birth rates and government regulations mandating early testing are pushing hospitals to expand screening programs. At the same time, growing awareness of infant health and increased investment in neonatal care continue to drive demand for advanced diagnostic instruments. For instance, PerkinElmer, Inc., a global leader in newborn screening, launched the QSight® 210 MD system to support laboratories with routine, high-throughput quantitation, reinforcing its commitment to innovating for a healthier world.
To learn more about this report, Request Free Sample
The hearing type segment in the newborn screening market share is expanding as healthcare providers respond to the increasing prevalence of congenital hearing loss and the critical need for early detection to aid language and cognitive development. Hospitals are actively implementing automated auditory brainstem response (AABR) and otoacoustic emissions (OAE) technologies to conduct efficient screenings. Governments are enforcing mandatory hearing tests for newborns, while parents and pediatricians are becoming more aware, driving wider adoption of hearing screening in neonatal care. For instance, in September 2025, Sir Lester Bird Medical Centre (SLBMC) launched its first Newborn Hearing Screening Program, ensuring all newborns are screened early for potential hearing issues to support healthy development, the hospital announced.
Healthcare providers are driving demand for hearing disability screening in the newborn screening market due to the high rates of congenital hearing impairments and the vital need for early diagnosis to enable effective intervention. They are adopting advanced technologies such as automated auditory brainstem response (AABR) and otoacoustic emissions (OAE) for fast and reliable detection. Governments are enforcing universal newborn hearing screening mandates, while parents and medical professionals are raising awareness about the advantages of early treatment, boosting the adoption of hearing disability screening in neonatal care. For instance, in September 2025, Dr. Zainuddin Ansari inaugurated the state-of-the-art OAE device at the Audiology Unit of the ENT Department and emphasized its role in testing newborns’ hearing. He stated that early hearing tests after birth are crucial for identifying and managing congenital hearing disabilities.
Hospitals play a key role in driving the newborn screening market demand by providing primary care for newborns and conducting widespread screenings immediately after birth. They use advanced infrastructure and diagnostic technologies to perform efficient and accurate tests. Governments mandate newborn screening before hospital discharge, which increases adoption. Hospitals also streamline workflows and integrate electronic health records to improve follow-up and patient management. Additionally, rising birth rates and growing awareness of early detection benefits help hospitals expand newborn screening programs. For instance, in March 2024, Niloufer Hospital, a state-run facility in Hyderabad, has launched a newborn screening programme to detect rare and serious genetic conditions in infants. The screening targets congenital hypothyroidism, congenital adrenal hyperplasia, and glucose-6-phosphate dehydrogenase deficiency.
To learn more about this report, Request Free Sample
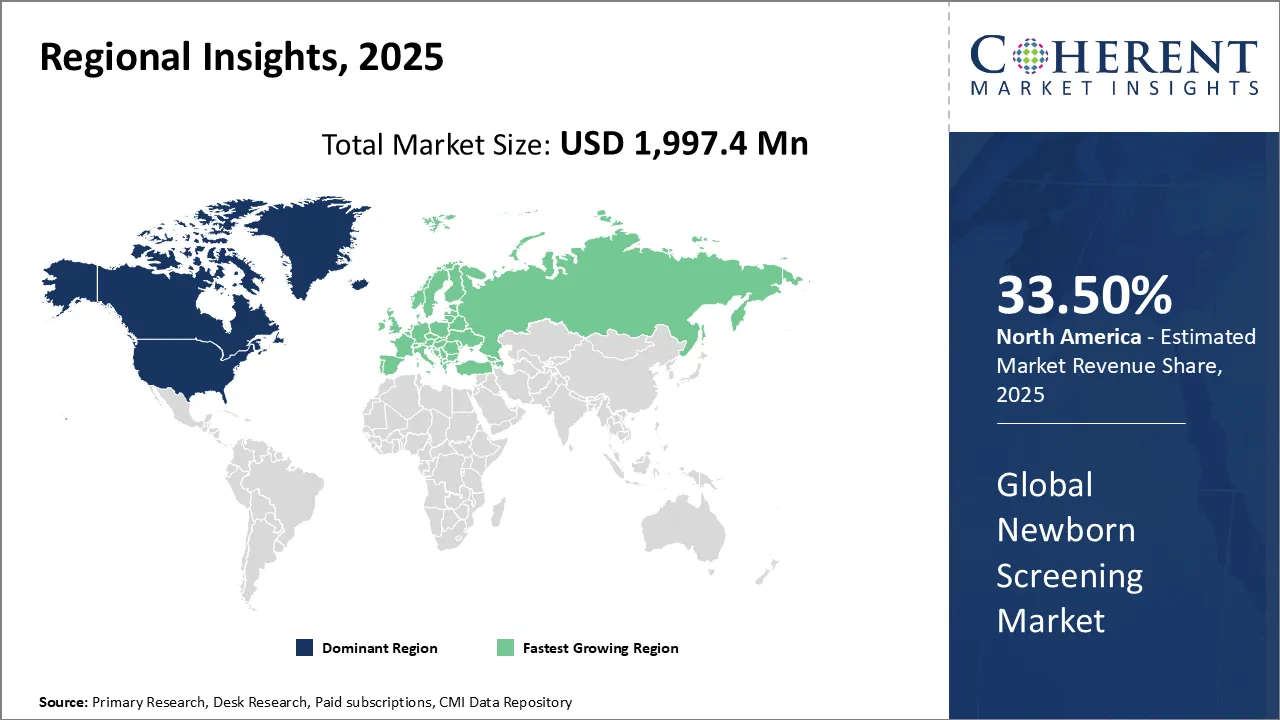
North America dominates the overall market with an estimated share of 33.5% in 2026. In North America, healthcare providers are expanding the newborn screening market by increasing the range of detectable conditions using advanced technologies like genomic sequencing and multiplex testing. Public health agencies and private organizations are collaborating to drive innovation and standardize screening processes. Governments are boosting support, while parents are becoming more aware of the benefits, encouraging widespread adoption. These combined efforts are making newborn screening a fundamental part of neonatal care throughout the region. For instance, Eight hospitals in New York have launched the U.S.’ largest pilot newborn screening program for multiple disorders, including Sanfilippo syndrome types A and B. The new test, called ScreenPlus, expands New York state's routine infant screening panel by adding 14 additional disorders.
In Europe, healthcare providers are advancing the newborn screening market by adopting personalized and precision medicine approaches that combine genetic and metabolic testing for early diagnosis. Governments are harmonizing screening protocols across countries and providing strong support through initiatives and funding. Research institutions and healthcare providers are collaborating to accelerate innovation in testing technologies. Public awareness campaigns are actively increasing acceptance and participation in screening programs. These efforts are helping Europe enhance neonatal health outcomes by expanding and improving newborn screening practices.
In the United Kingdom, healthcare providers are expanding newborn screening panels to cover more genetic and rare disorders. The National Health Service (NHS) is actively deploying advanced technologies like next-generation sequencing to improve early detection and diagnosis. The government supports nationwide screening programs through strong policies and funding. Researchers, healthcare professionals, and patient advocacy groups collaborate to drive ongoing advancements. Public education campaigns are increasing awareness and encouraging more families to participate in newborn screening across the UK. For instance, Oxford University in the U.K. launched a pilot study to conduct routine testing for SMA in newborn babies for the first time. The screening tests infants’ blood samples for genetic mutations causing SMA, enabling diagnosis within days of birth.
In the United States, healthcare providers are advancing the newborn screening market by integrating cutting-edge technologies such as whole-genome sequencing to identify a wider range of conditions early. State programs are standardizing processes and expanding screening panels to enhance neonatal outcomes. Federal initiatives provide research funding and encourage collaboration among public health agencies, hospitals, and private companies. Healthcare providers and parents are increasing awareness, which drives higher screening rates. These combined efforts improve early diagnosis and intervention throughout the country’s diverse healthcare system. For instance, US-based Revvity, Inc. launched its EONIS™ Q system, a CE-IVD declared platform that allows laboratories in CE-mark accepting countries to perform molecular testing for spinal muscular atrophy (SMA) and severe combined immunodeficiency (SCID) in newborns.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2,217.1 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 11.0% | 2033 Value Projection: | USD 4,603.1 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Agilent Technologies, Inc., Bio-rad laboratories, Inc., Medtronic Plc, Masimo Corporation, Natus Medical Inc., Perkin Elmer, Inc, ZenTec S.A., Trivitron Healthcare Pvt Ltd, Waters Technologies Corporation, Danaher Corporation, Baebies, Inc , Parseq Lab Co. Ltd , Chromesystem Instruments and Chemicals GmbH , and RECIPE Chemicals+ Instrument GmbH |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The newborn screening market is increasingly incorporating advanced genetic and genomic technologies, moving beyond traditional biochemical tests. This trend enables detection of a broader range of rare disorders with greater precision. Early identification through next-generation sequencing (NGS) and expanded panels supports personalized medicine approaches, enhancing prognosis. However, it also demands robust ethical frameworks, data interpretation skills, and counseling services to handle complex genetic information responsibly.
Leveraging detailed newborn screening data to design personalized treatment plans presents a major opportunity. Early detection of metabolic or genetic conditions allows clinicians to tailor interventions specifically to each infant’s needs, improving health outcomes. Integrating screening results with electronic health records and AI-driven analytics can support precision medicine approaches, optimizing therapy choices and monitoring. This personalized care pathway fosters better management of chronic conditions from infancy.
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients