Global preeclampsia diagnostics market is estimated to be valued at USD 2.56 Bn in 2026 and is expected to reach USD 5.16 Bn 2033, exhibiting a compound annual growth rate (CAGR) of 10.5% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
Global preeclampsia diagnostics market is expected to witness growth over the forecast period due to rising prevalence of preeclampsia among expectant. According to World Health Organization (WHO), hypertensive disorders during pregnancy affects 5-8% of all pregnancies globally and preeclampsia is a common complication responsible for significant number of maternal and fetal morbidity and mortality cases worldwide each year. Moreover, growing awareness regarding preeclampsia diagnosis and management and increasing access to advanced healthcare facilities in developing countries can present lucrative growth opportunities for players in preeclampsia diagnostics market. However, availability of alternative treatment options and lack of point-of-care diagnostic kits in low-income countries may hamper the market growth during the forecast period.
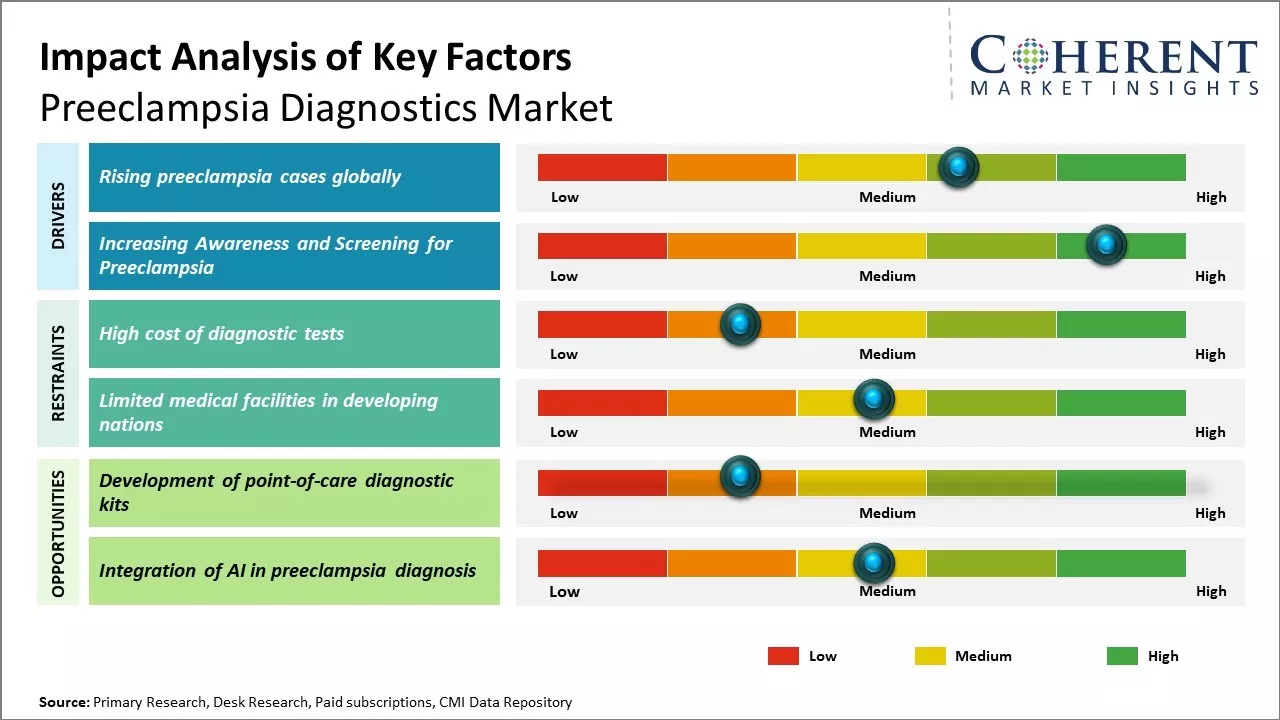
Rising preeclampsia cases globally
Rising preeclampsia cases globally can drive the global preeclampsia diagnostics market growth. Preeclampsia is a pregnancy complication characterized by high blood pressure and signs of damage to another organ system. It is a leading cause of maternal and neonatal morbidity and mortality worldwide. According to the World Health Organization (WHO), preeclampsia complicates approximately 5-8% of all pregnancies worldwide annually. According to the WHO in 2020, preeclampsia is responsible for 76,000 maternal and 500,000 fetal deaths globally every year. Increasing number of preeclampsia cases poses a huge clinical and economic burden on healthcare systems internationally. For instance, in August 2022, Mayo Clinic researchers, a nonprofit organization committed to clinical practice, found that women with a history of severe preeclampsia show more markers of brain cell damage and inflammation compared to those with uncomplicated pregnancies. These findings, presented at the Alzheimer's Association International Conference, suggest long-term effects of preeclampsia beyond pregnancy. Preeclampsia, affecting up to 15% of women, can lead to serious complications for both mother and baby if untreated. It is linked to increased risks of heart disease, stroke, and cognitive decline later in life, with no reliable early markers available. The study explored whether circulating extracellular vesicles containing brain cell membranes could be detected in women years after experiencing preeclampsia.
Get actionable strategies to beat competition: Request Free Sample
Increasing awareness and screening for preeclampsia
Rising awareness about preeclampsia and the health risks associated with it compels women to opt for regular screening and tests during pregnancy. Medical professionals and health organizations have been highlighting the importance of screening pregnant women for signs and symptoms of preeclampsia. These emphasize that early detection can help receive timely treatment and manage the condition well. Various national programs are also conducted to educate expectant mothers about underlying conditions, testing options, and managing comprehensive care.
Several new guidelines by reputed medical bodies now recommend screening protocols and tests schedules for all pregnant women based on their risk factors. This includes monitoring of blood pressure at every visit and checking for protein in urine samples at specific intervals. Detection of even mild symptoms is prompting doctors to conduct advanced tests like blood tests, ultrasounds to measure kidney function, blood platelet count, and fetal growth. Special attention is being paid to high-risk pregnancies owing to chronic conditions, multiple births, obesity, or a prior history of preeclampsia. All these efforts have certainly increased the number of tests being conducted per pregnancy.
Key Takeaways from Analyst:
Global preeclampsia diagnostics market is expected to witness significant growth over the forecast period owing to rising incidence of preeclampsia among expectant mothers. Rising health awareness regarding pregnancy complications and availability of advanced diagnostic technologies for early detection of preeclampsia can drive the market growth. However, high costs associated with diagnostic tests and lack of awareness in developing nations can hamper the market growth.
North America currently dominates the global preeclampsia diagnostics market due to increasing healthcare expenditure and growing adoption of advanced healthcare technologies in the region. Asia Pacific is anticipated to emerge as the fastest growing market due to rising healthcare investment and increasing focus of key players to tap into developing economies in the region.
Increased R&D activities for development of novel diagnostic markers and imaging modalities that help detect preeclampsia at an early stage can offer lucrative growth opportunities for the market players.
Market Challenges: High cost of diagnostic tests
The high cost of diagnostic tests can hamper the global preeclampsia diagnostics market growth. Preeclampsia is a pregnancy complication characterized by high blood pressure and signs of damage to other organ systems. It is a leading cause of maternal and fetal illness and death globally. However, due to the high cost of diagnostic tests, many expectant mothers in developing nations are unable to afford regular checkups and testing to detect preeclampsia early. Diagnostic tests play a key role in the early identification and timely management of preeclampsia. Tests such as blood pressure monitoring, complete blood count, liver and kidney function tests, urinalysis, and ultrasound are generally used to diagnose preeclampsia. However, the equipment, infrastructure and skilled professionals required to conduct these tests makes the overall diagnosis highly expensive. For example, ultrasound machines that can help detect signs of preeclampsia cost between US$ 30,000 to US$ 150,000 depending on the model. Repeat scans may sometimes be required, which further increases the overall treatment cost. This high expenditure acts as a massive barrier for expectant mothers with limited financial resources.
Market Opportunities: Development of point-of-care diagnostic kits
The development of point-of-care diagnostic kits provides a great opportunity to improve screening and diagnosis of preeclampsia. Preeclampsia diagnosis largely relies on frequent visits to healthcare facilities to monitor blood pressure and protein levels in urine samples. This conventional approach poses challenges in low resource settings with limited access to quality healthcare. Point-of-care diagnostic kits allow for simplified, affordable testing that does not require extensive clinical laboratories or trained healthcare workers. These have the potential to decentralize preeclampsia screening and enable early diagnosis in primary health centers, community clinics, and even at home. This can significantly expand access to diagnostics and allow for timely clinical management of preeclampsia cases. Point-of-care diagnostic kits employ innovative technologies to qualitatively or quantitatively detect markers of preeclampsia from whole blood, serum, plasma or urine samples collected via finger-prick or dipstick method. Some test results can be automatically generated within 15-30 minutes, compared to hours or days needed for laboratory-based tests.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
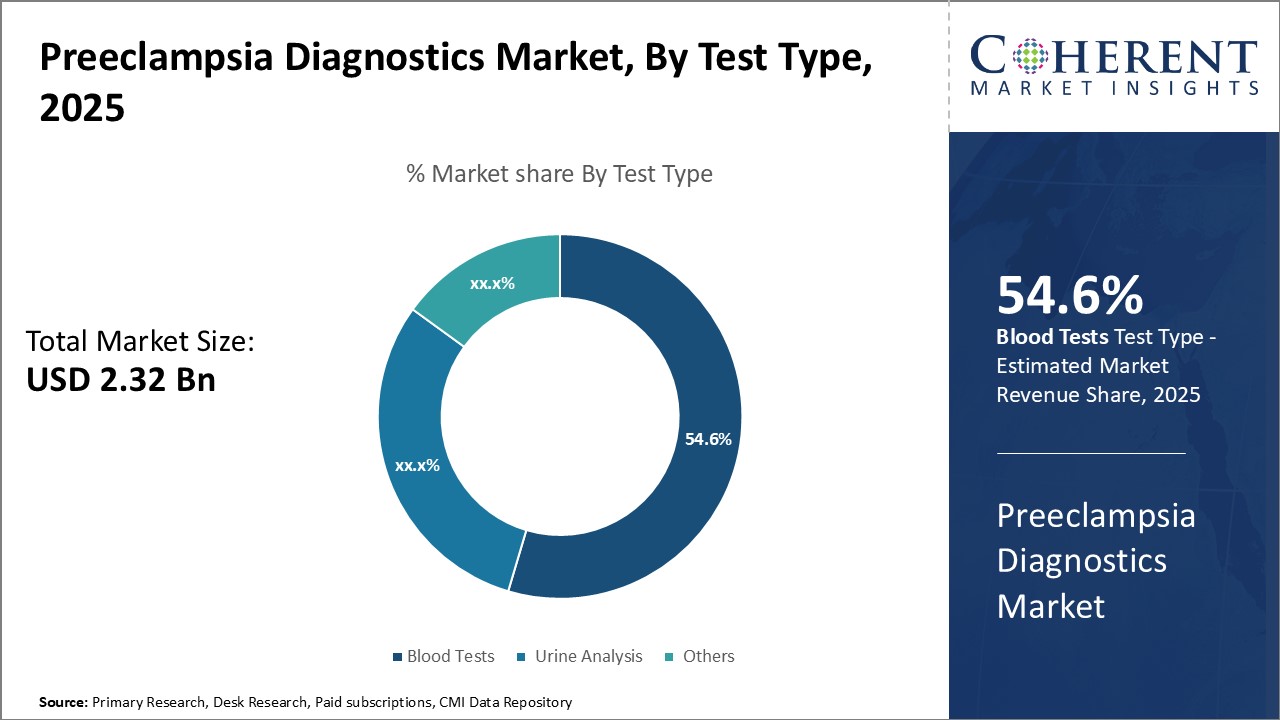
By Test Type: Fast and accurate diagnoses can drive the blood tests segment growth
In terms of test type, blood tests segment is estimated to contribute the highest market share of 55.6% in 2026, owning to its several key advantages over other diagnostic methods. Blood tests can detect pregnancy-induced high blood pressure and related conditions with a high level of accuracy through markers such as placental growth factor and soluble FMS-like tyrosine kinase-1. Receiving a definitive diagnosis in a short timeframe is critical for determining the appropriate treatment approach and ensuring the safety of both mother and baby. Blood samples can also be easily collected during routine prenatal checkups and require minimal additional visits or procedures. This convenience factor is appealing to both patients and healthcare providers. Furthermore, blood-based diagnostics allow for remote laboratory testing and centralized analysis, improving diagnostic consistency across different medical facilities. Advances in blood testing technologies continue to enhance detection capabilities while driving down costs. These benefits have made blood tests the preferred first-line diagnostic option for preeclampsia screening and monitoring.
To learn more about this report, Request Free Sample
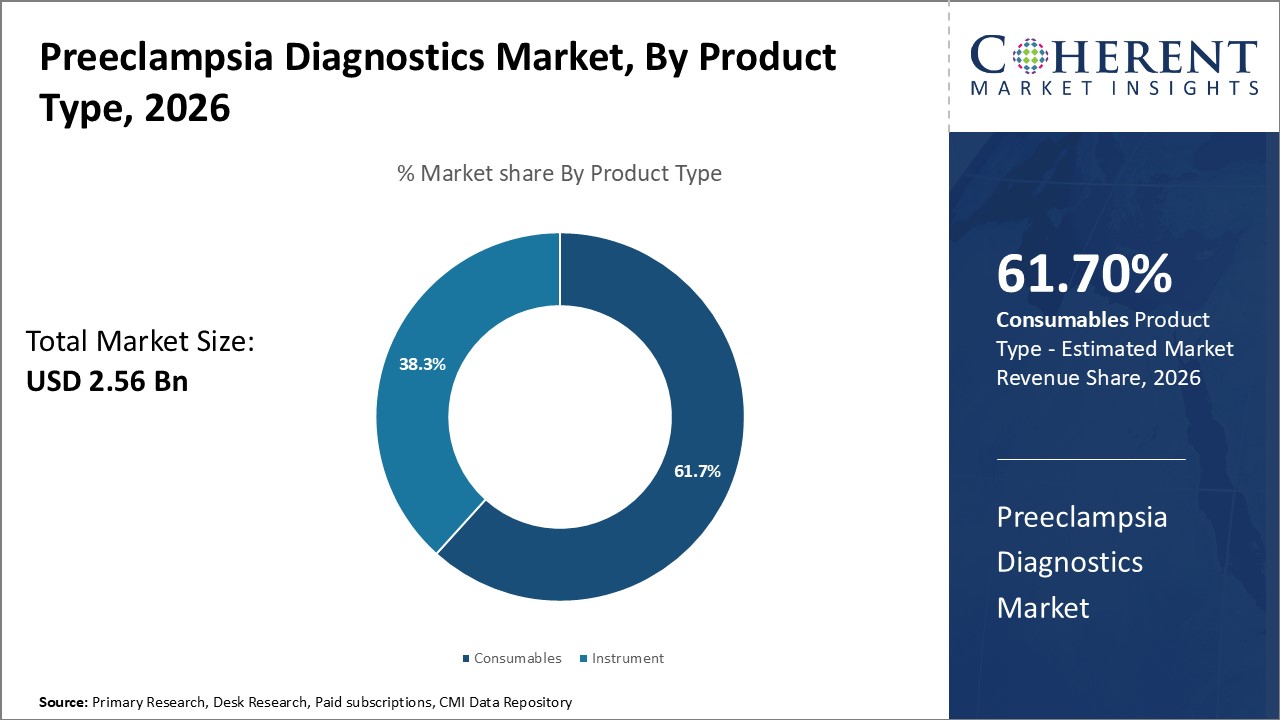
By Product Type: High consumption boosts demand for consumables
In terms of product type, consumables segment is estimated to contribute the highest market share of t 61.7% in 2026, due to the necessities of ongoing preeclampsia screening and management. Consumables such as reagents, kits, and other disposable items are essential components of virtually preeclampsia diagnostic tests. These must be regularly replenished as new patients receive screening and existing patients are monitored throughout their pregnancies. Given that preeclampsia affects approximately 5-8% of pregnancies globally, the large patient pool translates into steady, recurring demand for consumables. Furthermore, practical limitations of reusable instruments often require single-use consumables for hygienic and standardized testing. The consumable nature of these products also allows instrument manufacturers and reagent suppliers to benefit from reliable revenue streams. With diagnostic workflows becoming more streamlined and automated, consumables are engineered for simplified, high-throughput application without extensive technical skills. This enhances their utility while supporting wider diagnostic adoption.
By End User: Centralized facilities optimize resource utilization
In terms of end user, hospital segment is estimated to contribute the highest market share of 41.71% in 2026 as these are well positioned to maximize the benefits of centralized preeclampsia screening. Preeclampsia diagnostics involve sophisticated instruments, specialized medical expertise, and rigorous quality control processes. Centralizing these resources within hospital obstetric or clinical chemistry departments enables more efficient utilization across a large patient population. It allows for round-the-clock test availability and rapid delivery of results to attending physicians. This supports timely clinical management and improves health outcomes. Hospitals can also achieve important economies of scale in purchasing diagnostic products and maintaining equipment. The concentration of preeclampsia testing within hospitals better facilitates implementation of standardized diagnostic protocols and staff training. It also enables more robust data collection which aids research. These advantages have made hospitals the preferred access point for clinical adoption of advanced preeclampsia diagnostics. Their large patient volumes and logistical efficiencies continue attracting diagnostic developers.
Need a Different Region or Segment? Request Free Sample
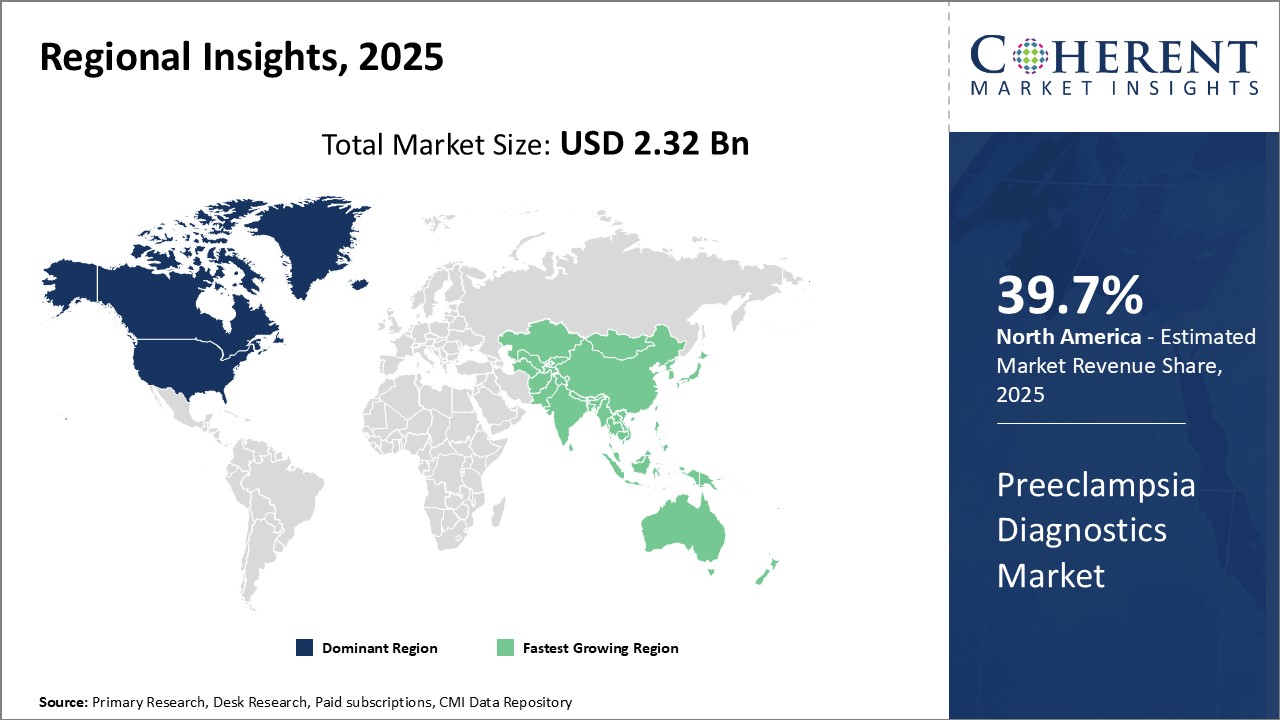
North America has established itself as the dominant region in the global preeclampsia diagnostics market with estimated market share of 39.7% in 2026. The region accounts for the largest market share due to growing prevalence of preeclampsia, strong presence of leading diagnostics players and high healthcare expenditure. The U.S., being the pioneer in medical devices and diagnostics, commands the major portion of regional market size. Streamlined regulatory approvals and favorable reimbursement policies have ensured early availability of novel diagnostic solutions.
Advanced diagnostic infrastructure, widespread adoption of precision medicine, and growing awareness about preeclampsia complications among physicians and patients can drive the regional market growth. Moreover, ongoing partnerships between regional diagnostic players and research institutes focuses on development of innovative tools with advanced detection capabilities. North American diagnostic companies have established strong global distribution networks to export their products.
Asia Pacific is recognized as the fastest growing regional market for preeclampsia diagnostics. Countries such as China, India and Japan are witnessing rapid economic expansion, leading to increased healthcare investments. Growing medical tourism coupled with expansion of private healthcare sector have promoted availability of advanced diagnostic technologies. Rising per capita incomes have augmented the ability of regional population to pay for sophisticated diagnostics.
Preeclampsia Diagnostics Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2.56 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 10.5% | 2033 Value Projection: | USD 5.16 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
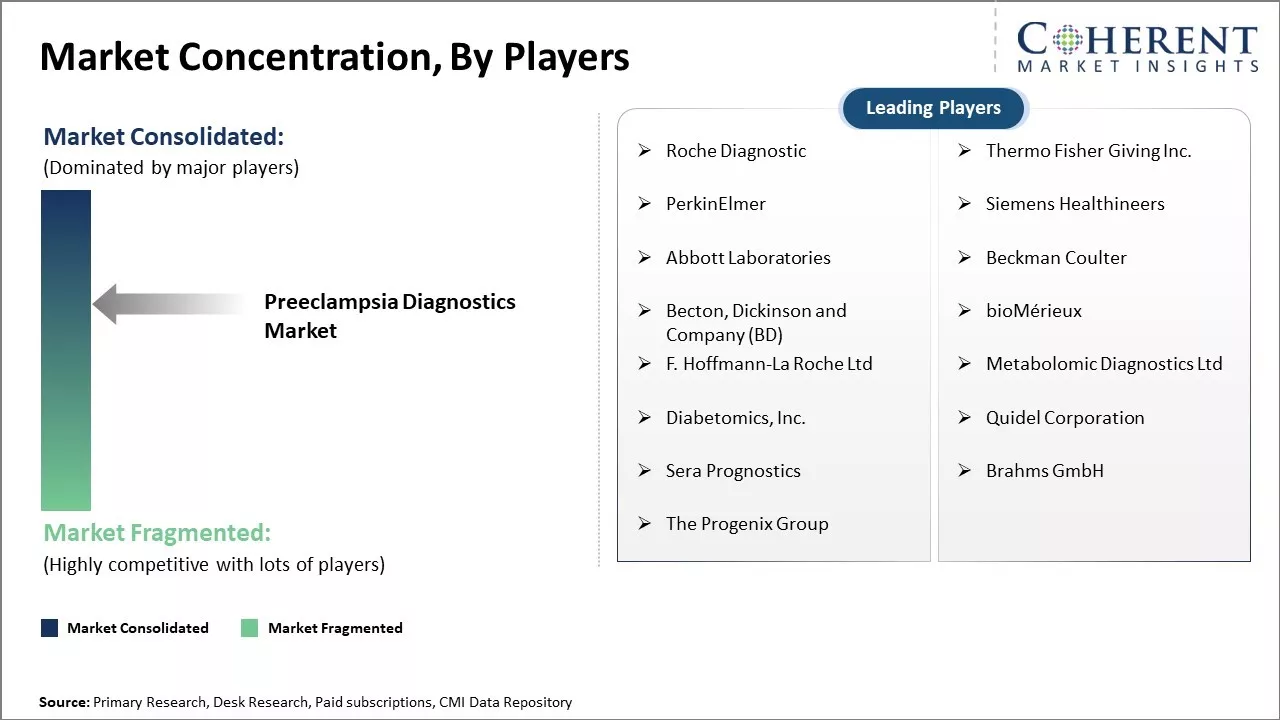
Roche Diagnostic, Thermo Fisher Giving Inc., PerkinElmer, Siemens Healthineers, Abbott Laboratories, Beckman Coulter , Becton, Dickinson and Company (BD), bioMérieux, F. Hoffmann-La Roche Ltd, Metabolomic Diagnostics Ltd, Diabetomics, Inc., Quidel Corporation, Sera Prognostics, Brahms GmbH, The Progenix Group |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: Global Preeclampsia Diagnostics Market involves diagnostic tools and techniques for detecting preeclampsia, a pregnancy complication characterized by high blood pressure and signs of damage to another organ system. Key products in this market include blood tests to detect placental proteins and measure liver and kidney function, urine tests to check for protein in the urine, ultrasounds to measure blood flow in the uterine arteries and examine fetal growth, and non-invasive tests using machine learning algorithms to analyze biophysical and biochemical markers. These non-invasive tests hold promise for early detection and monitoring of preeclampsia, potentially improving maternal and fetal outcomes.
Primary Research Interviews:
Databases:
Magazines:
Journals:
Newspapers:
Associations:
Public Domain Sources:
Share
Share
Abhijeet Kale is a results-driven management consultant with five years of specialized experience in the biotech and clinical diagnostics sectors. With a strong background in scientific research and business strategy, Abhijeet helps organizations identify potential revenue pockets, and in turn helping clients with market entry strategies. He assists clients in developing robust strategies for navigating FDA and EMA requirements.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients