The Pyrogen Testing Market is estimated to be valued at USD 1.47 Bn in 2026 and expected to reach USD 2.89 Bn by 2033, witnessing a CAGR of 10.1% over the forecast period (2026-2033), driven by rising demand for safe biologics, vaccines, and stringent regulatory requirements in pharmaceutical manufacturing.
Pyrogen testing is key to protecting patient’s safety (Drs/Docs) from toxic temp. Furthermore, testing is required for developing/approving regulated products due to the potential to cause illness or other issues. Tests employed include LAL, rFC, IPT, however, LAL is the preferred method as it has been established as the standard method across the industry due to its established reliability and acceptance with regulators (FDA/EMA). However, as there is increasing interest in eco-friendly test methods utilizing synthetic biology and bioreactors, the use of LAL is decreasing.
Application for pyrogen testing typically include injectables, vaccines, implants and CGT. Due to the growing interest in biologics/ biosimilars, pyrogen testing will also be required to comply with current standards established by agencies worldwide. Therefore, the use and demand for pyrogen testing continues to increase. However, the increase in usage/demand has increased over the last few years due to the increase in investment in the area of R&D, the significant increase in the production of biologics, and continued emphasis on patient safety and quality assurance.
|
Current Event |
Description and its Impact |
|
Expansion of biologics manufacturing |
|
|
Shift to Non-Animal Testing Options |
|
|
New FDA and EMA Regulations |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The Limulus Amebocyte Lysate (LAL) test remains the most common form of pyrogen testing due to its extreme sensitivity regarding the detection of endotoxins and for having strong regulatory approval globally. It is extensively used within the pharmaceutical industry for injectable drugs as well as for biological products. The reliability and cost-effectiveness of the LAL method make it continue to dominate this market despite the ethical concerns over animal testing.
On January 22, 2025, Lonza group AG introduced an upgraded version of its LAL assay platform with enhanced sensitivity and reduced interference from complex biologics.
Consumables are the largest portion of product type because they are repeat purchase used regularly in laboratories and for manufacturing. This category consists of reagents, assay kits and cartridges, all of which are required for each test cycle conducted. With the increasing volume of products being manufactured in the pharmaceutical industry, there is also an increasing demand for consumables making them an ongoing source of revenue.
Pharmaceutical and biotechnology companies are the major portion of the end-user segment for pyrogen testing products because they utilize this type of testing extensively throughout the development process, in clinical trials and during manufacturing. The increase in the production of biologics, vaccines and biosimilars has further contributed to the increasing need for robust pyrogen testing methodologies.
To learn more about this report, Request Free Sample
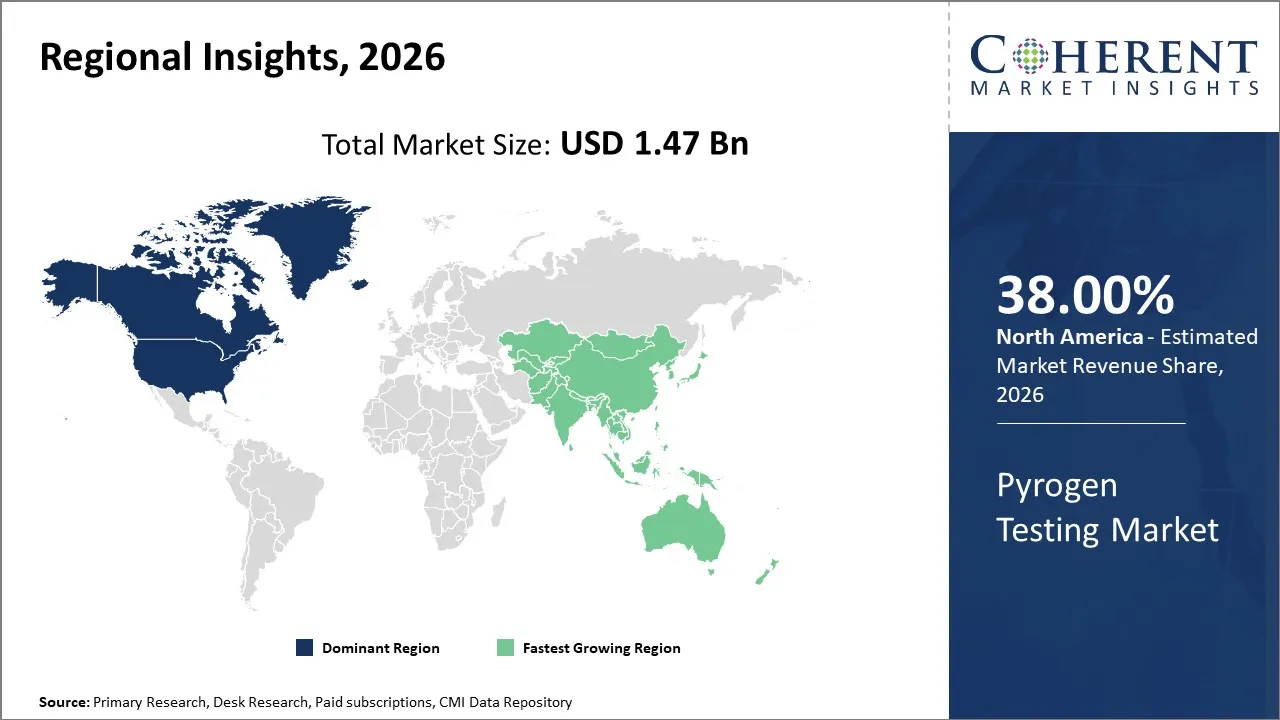
North America leads the market with 38% in 2026 and this is due to its very highly regulated environment for pharmaceuticals, advanced capabilities for biologics manufacturing, and large numbers of leading players in this space.
Asia Pacific will be the area with the highest growth in the pyrogen testing industry. Factors contributing to this growth include a boom in pharmaceutical manufacturing, an increase in drug manufacturing by third parties, and greater adoption of international quality standards.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1.47 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 10.1% | 2033 Value Projection: | USD 2.89 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Thermo Fisher Scientific, Merck KGaA, Lonza Group, Associates of Cape Cod Inc., Charles River Laboratories Inc., GenScript, WuXi PharmaTech (Cayman), Inc., bioMérieux, Wako Chemicals USA, Inc., Sanquin, Ellab A/S, and Pyrostar |
||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The rising development and commercialization of biologics and vaccines requires more rigorous safety testing to ensure products are not contaminated with pyrogens. The growing worldwide emphasis on immunizing individuals and providing advanced therapies will continue to increase the need for reliable pyrogen testing solutions.
Regulatory authorities require strict testing for endotoxins in injectable drugs and medical devices. Pharmaceutical manufacturers are therefore required to comply with these requirements by adopting newer testing methodologies, creating a steady demand for the product in the market.
Adopting an ethical and sustainable approach to laboratory testing will create immense growth opportunities in this industry through recombinant factor C testing. R-FCC is a logical substitute for the traditional Limulus Amebocyte Lysate (LAL) assays, meeting all relevant US & European regulatory and environmental requirements.
Share
Share
Abhijeet Kale is a results-driven management consultant with five years of specialized experience in the biotech and clinical diagnostics sectors. With a strong background in scientific research and business strategy, Abhijeet helps organizations identify potential revenue pockets, and in turn helping clients with market entry strategies. He assists clients in developing robust strategies for navigating FDA and EMA requirements.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients