North America and Europe Dental Sutures Market Size and Forecast – 2026-2033
The North America and Europe dental sutures market is expected to grow from USD 155.4 Mn in 2026 to USD 252.3 Mn by 2033, registering a compound annual growth rate (CAGR) of 7.2%. The market is poised for significant expansion, fueled by increasing dental care utilization.
According to the Centers for Disease Control and Prevention, in 2022, 63.7% of adults aged 65 and older in the U.S. visited a dentist within the past year. Notably, dental visits were more common among women (64.9%) compared to men (62.3%).
(Source: Centers for Disease Control and Prevention)
Additionally, according to Eurostat, individuals in the European Union recorded an average of 1.2 dental consultations per person in 2022, indicating the consistent utilization of dental care services across the region.
(Source: Eurostat)
Key Takeaways of the North America and Europe Dental Sutures Market
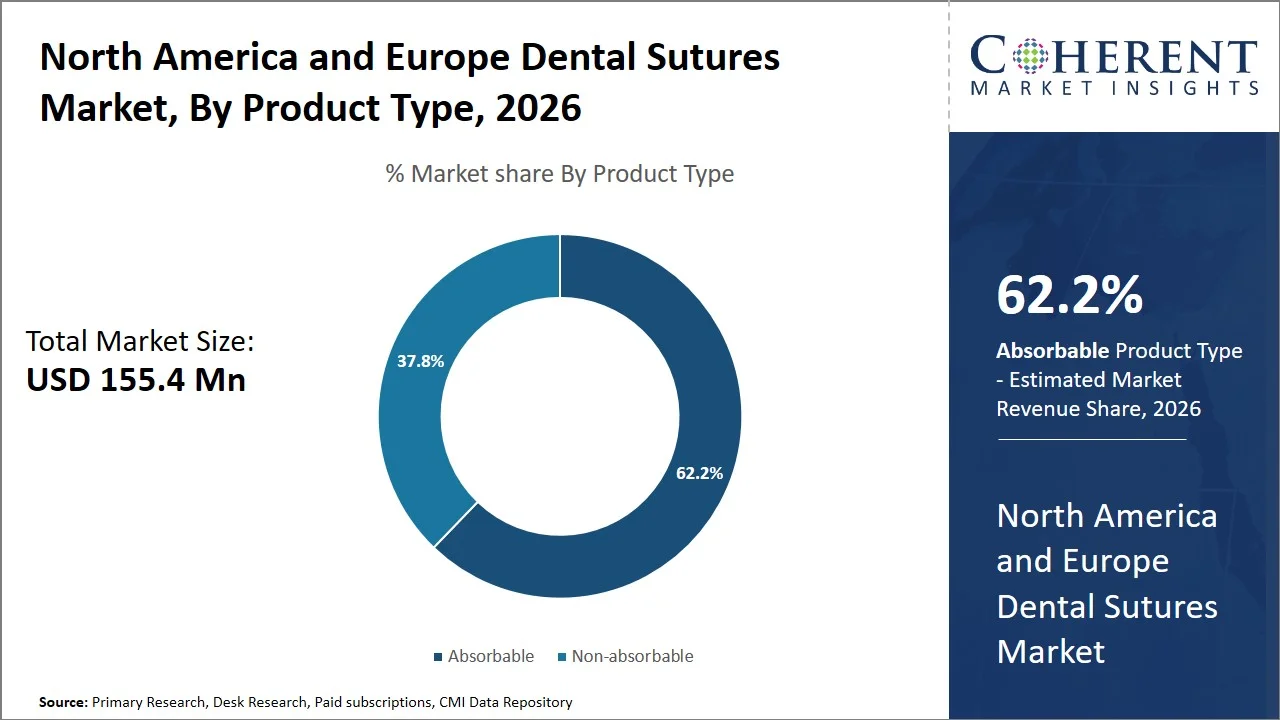
- Absorbable is projected to hold 62.2% of the North America and Europe dental sutures market share in 2026, making it the dominant product type segment, driven by strong acceptance in countries such as the U.S., Germany, and the U.K., where large numbers of dental implant and periodontal surgical procedures lead to a preference for sutures which do not require removal and improve patient compliance. For instance, according to the German Association of Implantology, approximately 1.3 million dental implants are placed annually in Germany, highlighting a high volume of dental surgical procedures that drive the preference for absorbable sutures to eliminate follow-up removal visits and improve patient compliance. (Source: National Library of Medicine)
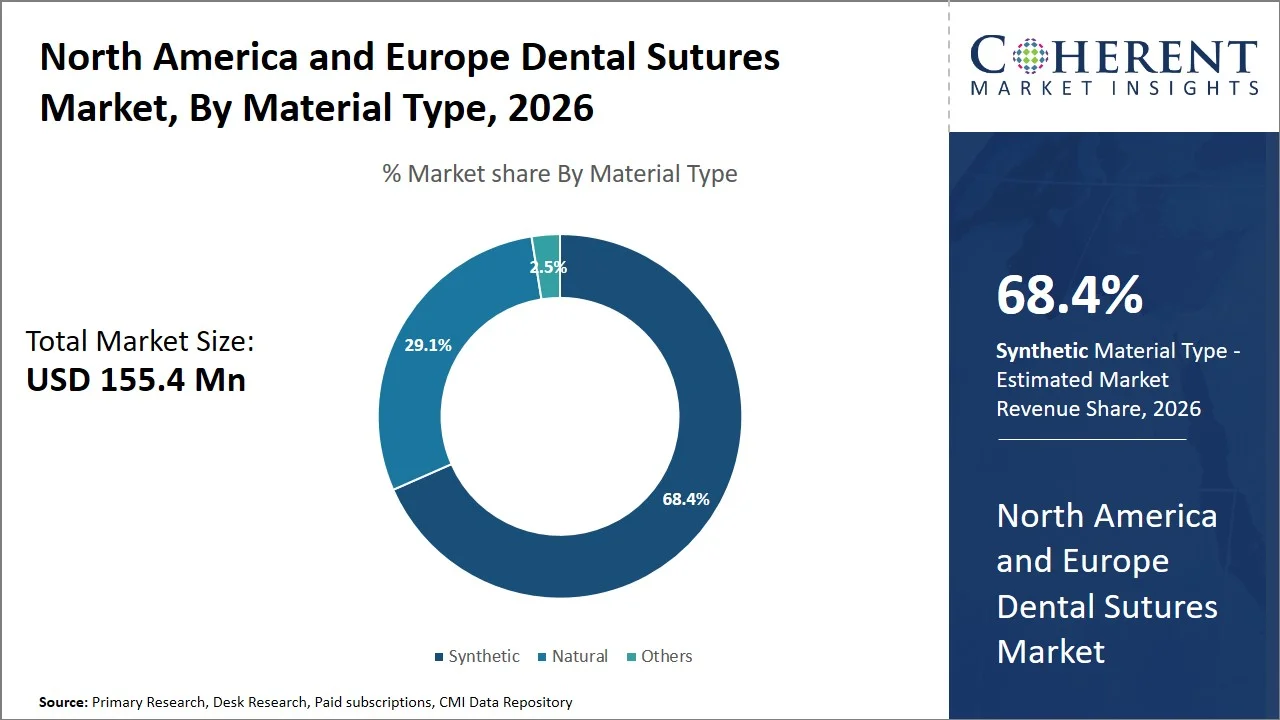
- Synthetic is projected to hold 68.4% of the North America and Europe dental sutures market share in 2026, making it the dominant material type segment, especially in the healthcare markets that are well-developed, such as the U.S. and Germany, because of their predictable absorption rates, low reactivity with tissues, and suitability for minimally-invasive or cosmetic dental surgery. For instance, according to the National Institute for Health and Care Excellence (NICE), artificial absorbable sutures, such as polyglactin 910, poliglecaprone, and polydioxanone, are manufactured such that they have controllable rates of absorption and low tissue reactivity. It means that they need not be removed since they will eventually be absorbed in the body. (Source: NICE)
- Dental Clinics segment is projected to hold 53.2% of the North America and Europe dental sutures market share in 2026, making it the dominant end user segment, led by nations like the U.S., the U.K., and France, wherein there is a high percentage of dentistry operations taking place in an outpatient environment (over 65% of surgical procedures in the U.S. are performed on an outpatient basis, including dental and minor oral surgeries). As a result of increased private clinics, reduced cycles of treatments, and increased patient visits as compared to that in hospitals, the use of dental sutures remains consistent.
- Growing Adoption of Antimicrobial-Coated Sutures: The growing emphasis on infection control within dental practices are fueling the need for antimicrobial-coated sutures. These sophisticated materials contribute to a reduction in post-surgical complications and enhance healing processes, thereby rendering them especially beneficial in implantology and periodontal interventions.
- Rising demand for aesthetic and implant dentistry: The growing popularity of dental implants and cosmetic treatments are fueling a need for sutures that offer exceptional precision, easy handling, and minimal scarring. This specialized market is leaning toward high-end absorbable sutures, which facilitate quicker healing and better cosmetic results, thereby opening doors for products with added value.
Why Do Absorbable Dominate the North America and Europe Dental Sutures Market?
Absorbable is projected to hold the market share of 62.2% in 2026, because of the efficiency of these sutures in terms of clinical efficacy and comfort as well as their compatibility with advanced methods of dental treatment. These types of sutures are naturally broken down within the body, which makes it unnecessary for patients to visit doctors' offices for sutures removal – an important aspect in a large volume healthcare system. Furthermore, the popularity of these sutures can be attributed to regulations concerning the quality of products issued by organizations such as the U.S. FDA and the EU Medical Device Regulation. For example, 510(k) clearances have been awarded by the U.S. FDA to various absorbable sutures, including those made from polyglactin 910 (Vicryl) and polydioxanone (PDS), thus proving their safety, biocompatibility, and equivalency to similar products already being used. (Source: Food and Drug Administration)
Why is Synthetic the Most Preferred Material Type in North America and Europe Dental Sutures Market ?
To learn more about this report, Request Free Sample
Synthetic is projected to hold a market share of 68.4% in 2026, due to their superior properties of reliability and consistent absorption. Synthetic sutures are produced in such a way that they become more reliable and stronger enough to be used in several types of dental operations, including dental implants. Besides, the rising uptake of synthetic sutures are partly influenced by the stringent guidelines set by the U.S. FDA in its 21 CFR Part 878-General and Plastic Surgery Devices and the EU MDR regulations, emphasizing the importance of producing high-quality materials.
Dental Clinics Segment Dominates the North America and Europe Dental Sutures Market
The dental clinics segment is projected to hold a market share of 53.2% in 2026, primarily attributable to the substantial volume of outpatient dental procedures, including extractions, implants, and periodontal surgeries, conducted within these facilities. Clinics serve as the principal healthcare providers, delivering treatments that are both economically viable and time-efficient when contrasted with hospital-based care. For example, surgical procedures like small wound stitching, stitching of the teeth, and skin surgeries are being carried out in clinics without the patient requiring admission in the hospital and greatly lowering medical expenses. Furthermore, the increasing prevalence of private dental practices and group clinics contributes to a heightened frequency of procedures and, consequently, a greater demand for sutures. In addition, the expanding utilization of sophisticated and minimally invasive dental techniques within clinics fosters a greater reliance on premium sutures.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Regulatory Shift: Strengthening Implementation of EU Medical Device Regulation (MDR 2017/745) |
|
|
Regulatory & Industry Shift: U.S. FDA Quality Management System Regulation (QMSR) Alignment with ISO 13485 (2026) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: European Commission, U.S. Food and Drug Administration)
North America and Europe Dental Sutures Market Dynamics
To learn more about this report, Request Free Sample
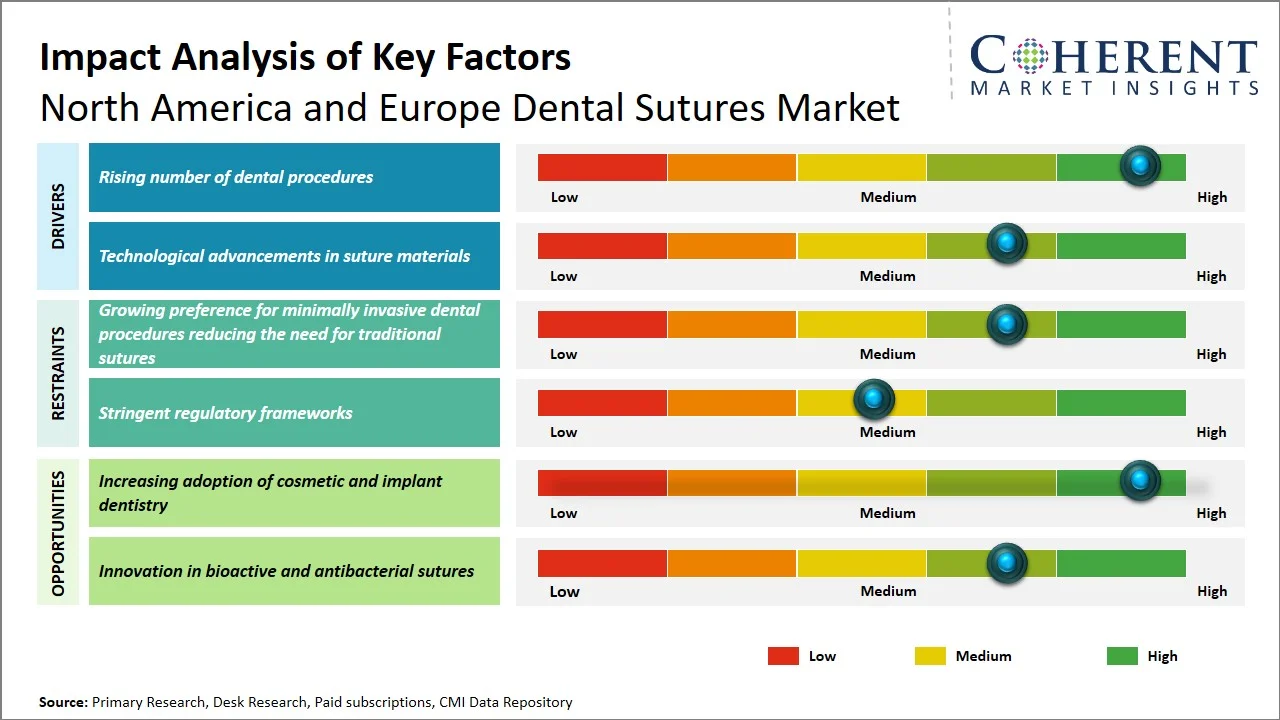
Market Drivers
- Rising number of dental procedures: The World Health Organization reports that approximately 50.1% of adults within the European region are afflicted by significant oral diseases, constituting one of the most substantial regional burdens worldwide (Source: World Health Organization). Concurrently, data from the National Institute of Dental and Craniofacial Research reveals that nearly 90% of adults aged 20–64 in the U.S. have encountered dental caries (Source: National Institute of Dental and Craniofacial Research). This considerable disease burden necessitates frequent dental interventions, including extractions, implants, and periodontal procedures, consequently stimulating the demand for dental sutures within these regions.
- Technological advancements in suture materials: Manufacturers are coming up with advanced synthetic absorbable sutures that provide better tensile strength and controlled absorption rate. Moreover, there are innovations related to antimicrobial sutures and special polymers that can help in minimizing the chances of infections, thus providing faster recovery from the surgery. For instance, Ethicon (Johnson & Johnson) has created triclosan-coated antimicrobial sutures (like Vicryl Plus) to stop bacteria from growing at the wound site. A big meta-analysis of 31 randomized clinical trials (about 17,968 patients) showed that triclosan-containing sutures lower the risk of surgical site infections (SSIs) by a lot compared to regular sutures. (Source: JAMA Network). Such developments are proving beneficial both clinically and in terms of ease of use for practitioners.
Emerging Trends
- Shift toward advanced synthetic absorbable sutures: Absorbable like polyglycolic acid and polylactic acid have become more popular because of the better control over the absorption process and enhanced tensile strength, leading to improved healing process of wounds and fewer complications following surgeries.
- Growing focus on minimally invasive and patient-centric dentistry: Developments in the field of dental surgery involving minimal invasiveness have generated a demand for suture material that is easy to handle with little tissue damage. Patients' preference for quick healing and fewer follow-up visits further drives this trend.
Regional Insights
To learn more about this report, Request Free Sample
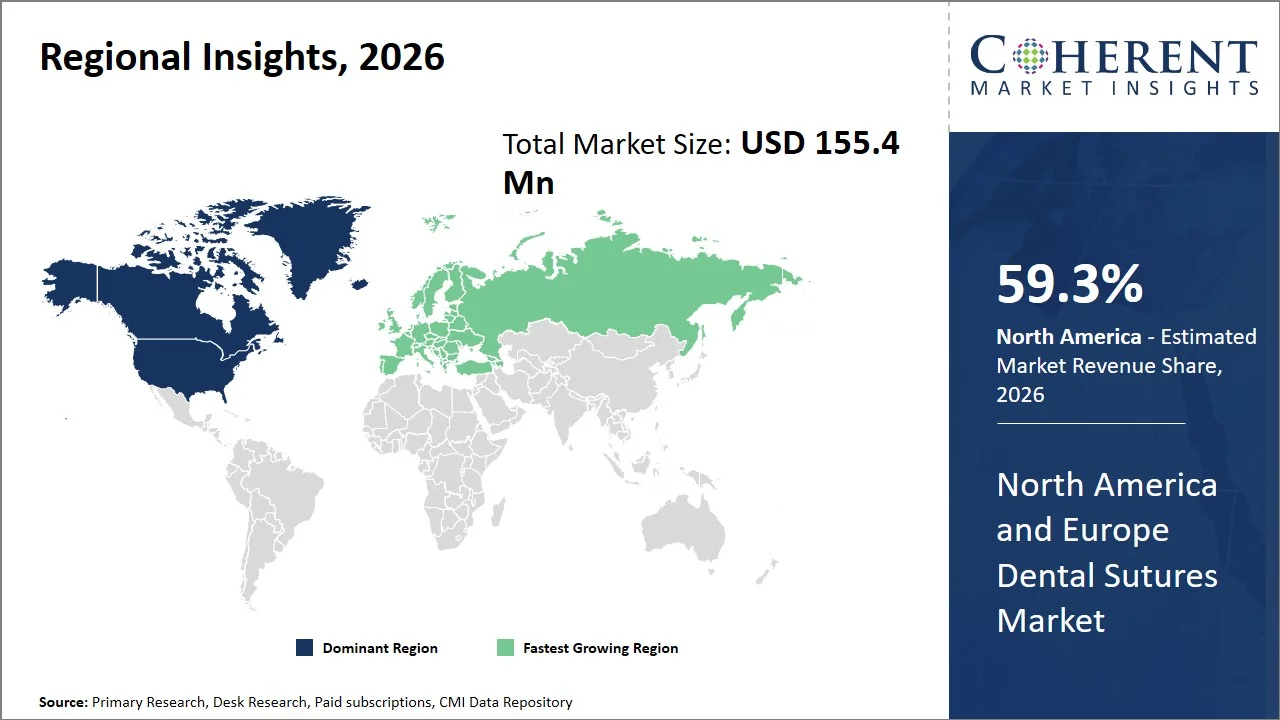
Why is North America a Strong Market for Dental Sutures?
North America leads the dental sutures market, accounting for an estimated 59.3% share in 2026, fueled by its established dental care system, the widespread use of sophisticated surgical methods, and the dominance of major industry players (such as Ethicon, Medtronic plc, B. Braun Melsungen AG, Dentsply Sirona Inc., and Patterson Companies Inc.). The region boasts a large pool of specialized dental professionals and sophisticated clinical environments, which facilitate intricate procedures like implants, periodontal surgeries, and maxillofacial operations.
Moreover, supportive reimbursement policies (e.g., Medicaid Adult Dental Benefits, Medicare Advantage dental coverage, and private dental insurance plans such as Delta Dental and Cigna Dental) and elevated healthcare expenditures (e.g., high per capita healthcare spending in the U.S. and Canada, exceeding USD 12,000 and USD 7,000 respectively) encourage the use of high-end, biocompatible suture materials. The presence of leading manufacturers and ongoing product development also bolster the market, positioning North America as a central location for advanced dental surgical solutions.
Why Does Europe Dental Sutures Market Exhibit High Growth?
The Europe dental sutures market is expected to exhibit the fastest growth with an estimated contribution of 40.7% share to the market in 2026, fueled by a robust regulatory environment, the growing popularity of sophisticated dental treatments, and heightened awareness of oral health. The region's existing healthcare infrastructure, which prioritizes product quality and safety, fosters the use of high-quality, biocompatible suture materials. Furthermore, the increasing demand for cosmetic and implant dentistry (Europe accounts for over 25% of the global dental implant market and performs millions of implant procedures annually), and a greater focus on aesthetic results, is boosting the number of procedures performed.
North America and Europe Dental Sutures Market Outlook for Key Countries
Why are the U.S. Leading Innovation and Adoption in the Dental Sutures Market?
The U.S. is at the forefront of the dental sutures market, a position bolstered by its sophisticated healthcare environment. This includes a robust clinical research network and a willingness to embrace new dental technologies. Major players like Johnson & Johnson (Ethicon), Henry Schein, and Dentsply Sirona are constantly innovating, especially in the areas of absorbable and antimicrobial sutures.
Additionally, the high frequency of dental procedures, coupled with extensive insurance coverage and favorable reimbursement policies (such as those provided by the Centers for Medicare & Medicaid Services) further fuels the adoption of these products. Furthermore, the U.S. Food and Drug Administration's stringent regulatory (21 CFR Part 878 – General and Plastic Surgery Devices) oversight allows for the swift yet safe approval of cutting-edge suture technologies. (Source: U.S. Food and Drug Administration)
Is Canada a Favorable Market for Dental Sutures?
Canada is considered as a promising market for dental sutures due to a strong regulatory environment in healthcare, high standards of clinical practice, and continuous demand for dental operations. The existence of a reliable regulatory body such as Health Canada guarantees that all dental sutures (medical devices) are subject to compliance with the Medical Devices Regulations (SOR/98-282). It implies that they need to undergo licensing, ISO 13485 certification, and a thorough safety assessment prior to entering the market. Such measures improve the quality and ensure the credibility of dental sutures (Source: Health Canada). Moreover, dental practitioners in Canada are obliged to only use certified equipment; hence guaranteeing constant demand for quality sutures.
Is U.K. Emerging as a Key Growth Hub for the Dental Sutures Market?
The U.K. seems to be making strides in becoming one of the key destinations for dental sutures owing to its highly developed healthcare system, rising dental procedure volumes (with millions of dental treatments delivered annually under the National Health Service, including a substantial number of surgical and restorative procedures), and rising use of advanced material technology. The existence of an organized public healthcare program in the form of NHS guarantees the availability of dental treatment services. The trend of moving into private dental clinics, in addition, will ensure the rise in number of complicated procedures like implants and periodontal surgeries that will have increased demand for sutures. Moreover, stringent regulation from the Medicines and Healthcare products Regulatory Agency (MHRA) according to UK Medical Devices Regulations 2002 (as amended) will help drive more investment into advanced quality and safety compliant products. (Source: The National Archives)
Why Does Germany Top the European Dental Sutures Market?
The country undertakes a considerable number of dental surgery and implantation (over 1–1.5 million dental implants placed annually), with the help of insurance facilities provided by the statutory health schemes, such as Statutory Health Insurance (GKV). Further, the presence of an advanced medtech industry (such as B. Braun Melsungen AG, Paul Hartmann AG, and Karl Storz) with consistent innovation in surgical products promotes the use of superior absorbable and antimicrobial dental sutures. Moreover, stringent regulations for the product manufacturing, according to EU Medical Device Regulation, supervised by Federal Institute for Drugs and Medical Devices (BfArM) of the country, make sure that the produced dental sutures are safe and of high quality.
Is Dental Sutures Market Developing in France?
A robust healthcare infrastructure, bolstered by the French National Health Insurance, guarantees extensive patient access to dental services, thereby maintaining procedural throughput. Regulatory supervision, as mandated by the EU Medical Device Regulation, fosters the utilization of sutures that meet stringent quality and compliance standards. The presence of prominent dental manufacturers, including Septodont, contributes to both innovation and local supply chains. Furthermore, the increasing emphasis on cosmetic and minimally invasive dental procedures is driving the accelerated adoption of sophisticated suture materials within the country.
Regulatory Landscape Governing the North America and Europe Dental Sutures Market
|
Region |
Regulatory Authority |
Regulations |
Key Requirements |
Impact on Market |
|
North America |
U.S. FDA; Health Canada |
FD&C Act; 21 CFR Part 820 (QSR); UDI; Medical Devices Regulations (SOR/98-282) |
Device classification, 510(k) clearance, GMP compliance, licensing (MDL), labeling, and traceability |
Ensures high product quality and patient safety but increases time-to-market and compliance costs, creating entry barriers for small players while favoring established manufacturers |
|
Europe |
European Commission / EMA / Notified Bodies |
MDR 2017/745; CE Certification |
CE marking, clinical evaluation, conformity assessment, quality audits, and post-market surveillance |
Strengthens product reliability and transparency; however, stricter MDR requirements have lengthened approval timelines and raised certification costs, impacting product launches and limiting market entry for new companies |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is the increasing adoption of cosmetic and implant dentistry creating new growth opportunities in the North America and Europe dental sutures market?
This surge in dental sutures are linked to the increased number of surgical dental procedures that necessitate precise wound closure. In the U.S., roughly 500,000 dental implant procedures are carried out each year, demonstrating a robust and expanding appetite for both restorative and aesthetic dental care. Concurrently, Europe, too, is a significant center for implant and cosmetic dentistry, representing a considerable portion of worldwide procedures, bolstered by growing demand for smile restoration and aesthetic enhancements. The region accounted for 40.2% of the dental implant procedures in 2023. These procedures, encompassing implants, gum surgeries, and soft tissue corrections, demand sophisticated suturing techniques to ensure optimal healing and appearance. Consequently, the need for high-performance dental sutures is rising across these regions. (Source: Aria Dental Care)
Market Players, Key Development, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In June 2025, the European Commission (DG GROW) declared that seven dental implants which were being evaluated as part of an EU-wide medical device monitoring program had passed the rigorous safety, sterility, and biocompatibility tests required by the Medical Device Regulation (MDR).
- In March 2024, Advanced Medical Solutions Group plc took a significant step by purchasing Peters Surgical. This global company specializes in surgical sutures and hemostasis products, a move designed to bolster AMS's standing in tissue repair and wound closure. The acquisition broadens the company's product range and its reach throughout Europe and North America. It also strengthens AMS's expertise in surgical and dental sutures. The expectation is that this strategic move will fuel growth, thanks to better distribution and the potential for cross-selling in dental applications.
Competitive Landscape
The dental sutures market in North America and Europe is characterized by intense competition, fueled by the presence of both established medical device firms and specialized dental product manufacturers, all responding to the increasing demand for sophisticated oral surgical techniques. This market is distinguished by ongoing advancements in suture materials, rigorous adherence to regulatory requirements, and the expansion of distribution channels. Consequently, key competitive strategies encompass product innovation in absorbable and antimicrobial sutures, the establishment of strategic alliances with dental care professionals, strict compliance with quality standards, and the augmentation of product offerings and regional reach. Key focus areas include:
- Product innovation and development of advanced absorbable and antimicrobial sutures
- Strategic collaborations and partnerships with dental clinics and healthcare providers
- Compliance with stringent regulatory frameworks and quality standards
- Expansion of product portfolios and geographic presence across North America and Europe
Market Report Scope
North America and Europe Dental Sutures Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 155.4 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.2% | 2033 Value Projection: | USD 252.3 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Ethicon, B. Braun Melsungen AG, DemeTECH Corporation, Teleflex Incorporated, Advanced Medical Solutions Group PLC, Medtronic plc, Zimmer Biomet Holdings Inc., Smith & Nephew plc, Envista Holdings Corporation, and Hu-Friedy Mfg. Co., LLC |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- The future of the North America and Europea dental sutures market would be defined by the growing trend of minimally invasive dental surgeries, coupled with a marked preference for biocompatible and absorbable sutures, which have better results and improve patients’ experiences. Innovation in products, especially synthetic absorbable sutures with high tensile strength and controlled absorption, would play a crucial role in defining the competitive scenario.
- The best opportunity will most probably be in synthetic absorbable sutures in the U.S. because of high volume procedures, developed dental facilities, and a high degree of preference towards patient-friendly products. Besides, Western European countries like Germany and the U.K. have emerged as important markets owing to increased awareness about dental care and developed reimbursement systems.
- For market players to have an edge against their competitors, they need to focus on developing sophisticated yet biocompatible materials, build networks with dental clinics and hospitals, and distinguish themselves through ease of use and fast healing. Proper cost management and regulatory compliance will be the key factors in future success.
Market Segmentation
- Product Type Insights (Revenue, USD Mn, 2021 - 2033)
- Absorbable
- Non-absorbable
- Material Type Insights (Revenue, USD Mn, 2021 - 2033)
- Synthetic
- Natural
- Others
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Hospitals
- Dental Clinics
- Others
- Regional Insights (Revenue, USD Mn, 2021 - 2033)
- North America
- Europe
- Key Players Insights
- Ethicon
- Braun Melsungen AG
- DemeTECH Corporation
- Teleflex Incorporated
- Advanced Medical Solutions Group PLC
- Medtronic plc
- Zimmer Biomet Holdings Inc.
- Smith & Nephew plc
- Envista Holdings Corporation
- Hu-Friedy Mfg. Co., LLC
Sources
Primary Research Interviews
- Dental surgeons and oral/maxillofacial specialists performing suturing procedures
- Dental clinics, hospitals, and ambulatory surgical centers utilizing dental sutures
- Manufacturers and distributors of dental sutures (absorbable and non-absorbable)
- Regulatory experts and procurement managers in dental healthcare settings
Stakeholders
- Dental suture manufacturers and suppliers
- End-use Sectors
- Hospitals
- Dental clinics and specialty centers (periodontics, implantology, oral surgery)
- Academic and dental research institutes
- Regulatory & Health Bodies: U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), American Dental Association (ADA), Council of European Dentists (CED)
Databases
- U.S. FDA – Medical device approvals and dental product database
- European Commission / EMA – Medical device and healthcare regulations database
- WHO – Global Health Observatory (oral health statistics)
- OECD Health Statistics – Healthcare expenditure and dental care utilization data
Magazines
- Dental Tribune – Dental industry trends and innovations
- Dentistry Today – Clinical advancements in dental procedures and materials
- Medical Device Network – Insights on surgical sutures and device innovations
Journals
- Journal of Dental Research – Clinical studies on dental procedures and materials
- Clinical Oral Implants Research – Implant-related surgical techniques and outcomes
- International Journal of Oral and Maxillofacial Surgery – Surgical advancements and suturing practices
- Journal of Periodontology – Periodontal surgery and wound healing studies
Newspapers
- The Wall Street Journal – Healthcare and medical device industry trends
- Financial Times – Global healthcare market insights
- The New York Times – Public health and dental care awareness trends
- The Guardian – European healthcare policies and dental sector developments
Associations
- American Dental Association (ADA)
- European Federation of Periodontology (EFP)
- International Association for Dental Research (IADR)
- FDI World Dental Federation
Public Domain Sources
- WHO – Oral health reports and global burden of dental diseases
- CDC – Dental care utilization and oral health statistics (U.S.)
- NIH – Research on oral health and dental materials
- European Commission – Healthcare and medical device regulations
Proprietary Elements
- CMI Data Analytics Tool, Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients