Global Tracheotomy Tube Market Size and Forecast: 2026-2033
The global tracheotomy tube market is expected to grow from USD 236.4 Mn in 2026 to USD 343.7 Mn by 2033, registering a compound annual growth rate (CAGR) of 5.5% from 2026 to 2033. The market for tracheotomy tubes is poised for significant expansion, fueled by the soaring burden of chronic respiratory diseases such as chronic obstructive pulmonary disease (COPD) and asthma.
According to the World Health Organization, chronic respiratory diseases (CRDs) are among the most common non-communicable diseases (NCDs), and are the sixth leading cause of death in the WHO European Region, with almost 400,000 deaths every year, and nearly 80% of these deaths are caused by chronic obstructive pulmonary disease (COPD).
(Source: World Health Organization)
Key Takeaways of the Global Tracheotomy Tube Market
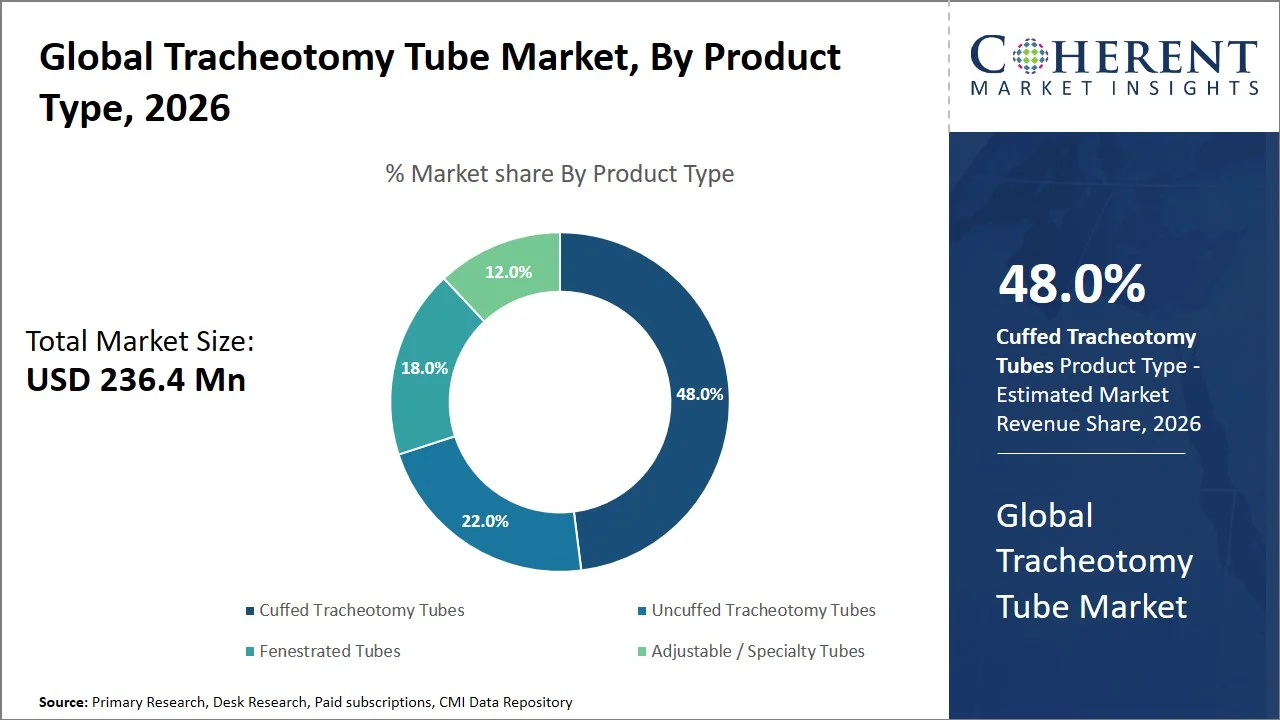
- Cuffed tracheotomy tubes are projected to hold 48.0% of the global tracheotomy tube market share in 2026, making it the dominant product type segment, with North America, especially the U.S., taking the lead on account of their rigorous ICU airway management policies (such as Centers for Disease Control and Prevention ventilator management guidance, National Institutes of Health COVID-19 Treatment Guidelines for mechanical ventilation, and American Society of Anesthesiologists difficult airway management guidelines) and extensive reliance on invasive ventilation procedures. The cuffed tubes are preferred over other product types for patients under mechanical ventilation because it guarantees proper sealing of the airway passage and prevent aspiration. For instance, Centers for Disease Control and Prevention (CDC) provides clinical guidance for managing patients suffering from acute respiratory diseases, which highlights the importance of ventilator-based management in critical care settings, where cuffed airway devices are standard practice. (Source: Centers for Disease Control and Prevention)
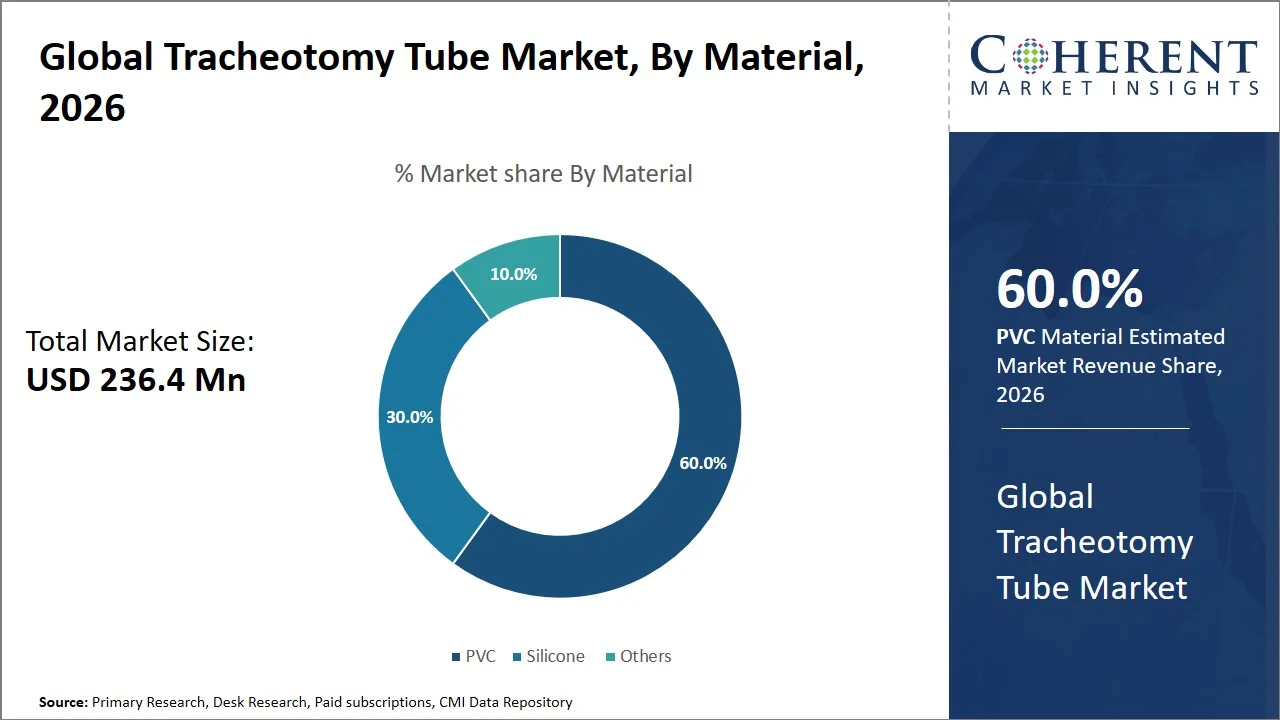
- PVC is projected to hold 60.0% of the global tracheotomy tube market share in 2026, making it the dominant material segment, with high penetration in Asia Pacific particularly India, attributed to government-led procurement and domestic manufacturing policies (such as the Ministry of Commerce and Industry (India) ‘Make in India’ initiative for medical devices, the Production Linked Incentive (PLI) Scheme for medical devices by the Department of Pharmaceuticals (India), and centralized public procurement through the Central Medical Services Society). PVC tubes are extensively used owing to their cost effectiveness and use in disposable applications in public healthcare systems. For instance, in India, the Ministry of Commerce and Industry encourages manufacturing in the country through the “Make in India” program, whereas centralized purchasing agencies like the Central Medical Services Society help bulk procurement and distribution of essential medical supplies to public hospitals, reinforcing the adoption of PVC-based devices due to cost-effectiveness. (Source: Make in India)
- Adult is projected to hold 83.0% % of the global tracheotomy tube market share in 2026, making it the dominant patient type segment, with Europe leading adoption due to entrenched clinical protocols for airway management and long-term respiratory care in hospital settings. Adult patients are the primary recipients of invasive ventilation and tracheostomy procedures within ICU pathways. For instance, the National Institute for Health and Care Excellence provides evidence-based guidance regarding critical care practices and airway management in adults, including ventilation management pathways that are used across NHS Hospitals. (Source: National Institute for Health and Care Excellence)
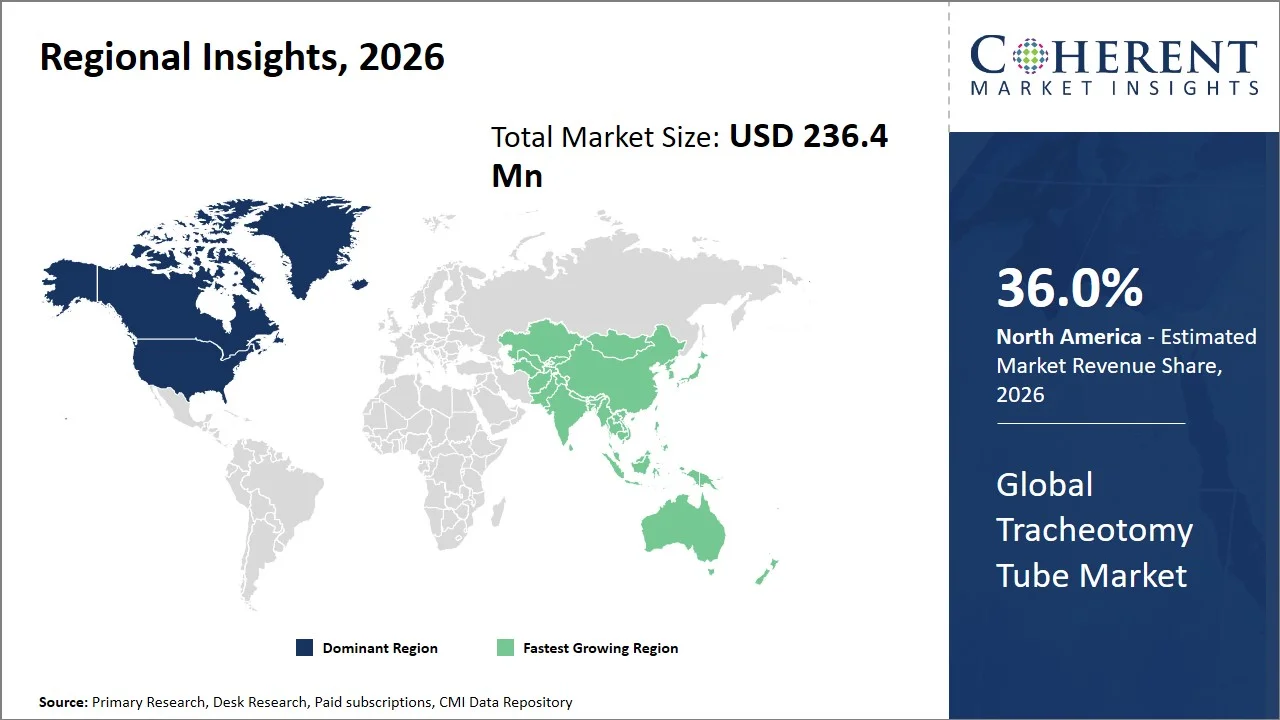
- North America maintains dominance with an estimated share of 36.0%in 2026, facilitated by the existence of structured regulatory framework (such as medical device classification and quality system regulations enforced by the U.S. Food and Drug Administration under 21 CFR Part 820, and post-market surveillance and vigilance systems under the Health Canada Medical Devices Regulations) and standardized procurement mechanisms across the U.S. For instance, tracheostomy tubes are specifically regulated by the U.S. FDA under 21 CFR § 868.5730 (Tracheostomy Tube and Tube Cuff), which specifies the classifications and performance requirements that ensure the uniformity of the approval process and accessibility across healthcare facilities. (Source: Code of Federal Regulations)
- Asia Pacific is expected to exhibit the fastest growth with an estimated contribution of 24.0% share in 2026, attributed to fast-growing public health care infrastructure and national programs aimed to increase capacity for critical care. For instance, in Australia, health infrastructure development plans supported by the government, including the Australasian Health Facility Guidelines (AusHFG) developed by the Australasian Health Infrastructure Alliance, outline the necessary design standards for ICUs as well as their expansion and modernization, thus enabling constant improvements in capacity within all public hospitals. These guidelines have been implemented by state health authorities for new hospitals and ICU upgradations, ensuring constant growth in critical care facilities. (Source: Australasian Health Infrastructure Alliance)
- Rising Demand for Specialized Pediatric and Neonatal Tracheotomy Tubes: Due to rising cases of congenital breathing problems and preterm births, there is a growing need for size-appropriate and low-pressure tracheostomy tubes that can be used on children and neonates. The market is focusing on tracheostomy tubes with improved safety features like soft construction and proper fixing systems to avoid any complications. This niche segment presents strong growth potential as pediatric critical care infrastructure expands globally.
- Growing Adoption of Single-Use and Infection-Control Focused Tubes: Greater emphasis on hospital-acquired infection (HAI) prevention will drive the transition towards sterile and single-use tracheotomy tubes and the use of more sophisticated materials, including PVC without DEHP plasticizers and silicone. There is a growing preference among healthcare professionals for devices that minimize cross-infection risks while adhering to high standards of safety. This trend is creating opportunities for manufacturers to differentiate through infection-resistant designs and regulatory-compliant materials.
Why Do Cuffed Tracheotomy Tubes Dominate the Global Tracheotomy Tube Market?
Cuffed tracheotomy tubes are projected to hold a market share of 48.0% in 2026, attributed to their vital importance in providing effective mechanical ventilation and airway protection during acute and intensive care procedures. While uncuffed tracheotomy tubes lack an important inflatable balloon required for a tight seal to be created to guarantee proper delivery of positive-pressure ventilation without leaks, cuffed tracheotomy tubes ensure that this critical feature is available. According to the literature on airway management and ventilation techniques, cuffed tracheotomy tubes should be used for patients who need mechanical ventilation as they are crucial for maintenance of proper tidal volume and oxygenation. (Source: OpenAnesthesia)
In addition, cuffed tubes protect against any possible aspiration by serving as a barrier that prevents inhalation of any stomach contents and secretions into the lungs, thus helping to reduce the risk of aspiration and its consequences. (Source: The University of Iowa) For instance, clinical reference article published by the National Library of Medicine, highlight that the cuff of a tracheostomy tube is inflated to ensure effective ventilation by preventing air leakage. In addition, these references highlight the role of cuffed airway devices in reducing aspiration risk, supporting their routine use in critical care airway management. (Source: National Library of Medicine) The combined clinical benefits efficiency in ventilation, prevention of aspiration, and routine use in ICU protocols keep pushing the global trend toward the use of cuffed tracheotomy tubes.
Why does PVC Represent the Largest Material Segment in the Global Tracheotomy Tube Market?
To learn more about this report, Download Free Sample
PVC is projected to hold a market share of 60.0% in 2026, owing to its compatibility with clinical applications, versatile design, and regulatory approval in airway management devices. PVC tubing used for medical purposes can be developed to have sufficient flexibility and stiffness to ensure atraumatic tracheal intubation, but still maintain adequate lumen patency during mechanical ventilation. In addition, it is compatible with incorporating vital attributes like inflatable cuffs, smooth exteriors, and standardized connections, which are vital for proper airway sealing and compatibility with ventilators.
For instance, regulatory submissions reviewed by the U.S. Food and Drug Administration specify that widely used tracheostomy tubes (e.g., Shiley models) are manufactured using PVC, exemplifying its consistent use in approved, hospital-grade airway devices. (Source: Food and Drug Administration) Moreover, due to its thermoplastic properties, PVC facilitates precise molding as well as mass manufacturing of disposable and sterile products aligning hospital infection control guidelines and procurement preferences.
Adult Segment Dominates the Global Tracheotomy Tube Market
The adult segment is projected to hold a market share of 83.0% in 2026, owing to its standardized use across hospital-based airway management, surgeries, and intensive care unit (ICU) management, where tracheostomy is commonly practiced on adult patients. Hospital protocols for airway management and respiratory support, including anesthesia induction and recovery airway management, mechanical ventilation for extended periods, and step-down respiratory care, mainly revolve around the management of adult patients; hence, the high usage of tracheotomy tubes among adults is ensured.
Moreover, adult care units require various types of tracheotomy tubes, which include different sizes, cuffed tracheotomy tubes, and tracheotomy tubes with inner cannulas, thus providing more applicability of the product in the tracheotomy tube market.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
FDA Strengthens Compliance Requirements for Class II Airway Devices under 510(k) Pathway (2024–2026 Updates) |
|
|
FDA Recognition Update of ISO Standards for Respiratory Devices (December 2024) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: Hogan Lovells, Food and Drug Administration)
Market Dynamics
To learn more about this report, Download Free Sample
Market Drivers
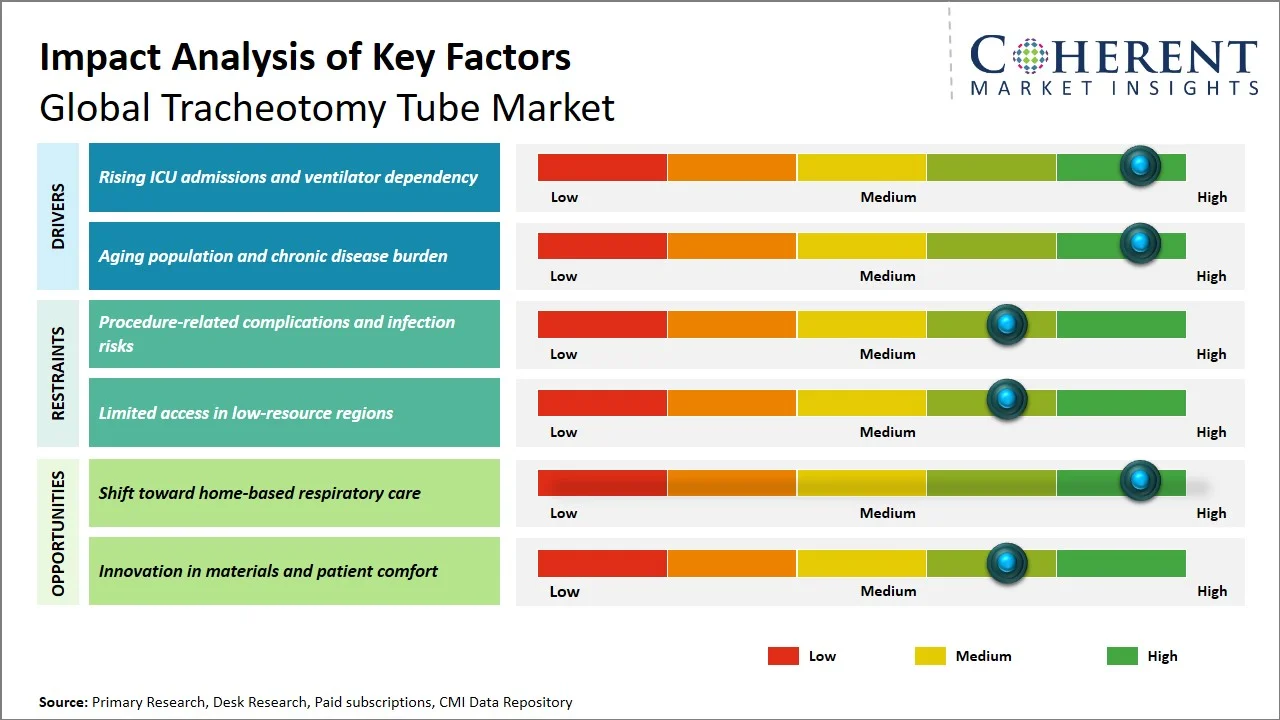
- Rising ICU admissions and ventilator dependency: Increased ICU admissions and ventilator dependence have continued to fuel the demand for tracheostomy tubes due to a significant number of critical care patients who require extended respiratory and airway management. The research published by National Library of Medicine states that at any given time, approximately two thirds of ICU beds are occupied, and approximately one third of these beds are occupied by patients requiring mechanical ventilation, emphasizing the significant impact on critical care resources and reliance on respiratory assistance. (Source: National Library of Medicine) Additionally, data published by Australian Institute of Health and Welfare, indicate that in 2023–2024, there were about 156,000 hospitalizations where patients required Level 3 ICU treatment, which translated to 13.0 million hours of critical care being delivered. In the public hospital sector, ICU services were delivered through 107,000 hospitalizations, with an average stay of over 100 hours per patient, demonstrating sustained reliance on intensive respiratory and life-support interventions. (Source: Australian Institute of Health and Welfare) With the rising number of ICU stays and critical care needs, the requirement for airway access during prolonged ventilation periods becomes crucial, resulting in an increased adoption rate of tracheostomy tubes in hospitals.
- Aging population and chronic disease burden: The aging population and burden of chronic diseases become major factors influencing the market for tracheotomy tubes since older individuals tend to need longer respiratory support and advanced airway management. According to World Health Organization, in 2019, the number of people aged 60 years and older was 1 billion. This number will increase to 1.4 billion by 2030 and 2.1 billion by 2050. (Source: World Health Organization) Additionally, the proportion of people aged 60 years and above is expected to nearly double from 12% to 22% between 2015 and 2050. (Source: World Health Organization) With the elderly individuals being more prone to diseases and health issues that may require hospitalization and ventilation, it is evident that this demographic shift will result in a greater need for tracheotomy tubes.
Emerging Trends
- Increasing Preference for Cuffless and Low-Pressure Cuffed Tubes to Minimize Airway Trauma: The use of non-cuff tracheotomy tubes and low-pressure cuff tracheotomy tubes are becoming more common among clinicians in order to minimize the risk of developing tracheal stenosis, mucosal trauma, and other complications. This is due to changes in the clinical management practices that aim for patient safety, early decannulation, and comfort of patients, particularly in long-term care settings.
- Growing Integration of Customization and Patient-Specific Tracheostomy Solutions: Tracheotomy tube customization is emerging as a growing trend especially for patients who have unique anatomical characteristics or need prolonged ventilatory support. Technological developments in medical imaging and engineering have made it possible to design tubes based on an individual’s anatomy, including tube length, flexibility, and even the type of material used to manufacture it. This growing trend has become quite popular in sophisticated health care settings where individualized airway management is a critical issue.
Regional Insights
To learn more about this report, Download Free Sample
Why is North America a Strong Market for Tracheotomy Tubes?
North America leads the global tracheotomy tube market, accounting for an estimated 36.0% share in 2026, attributed to a well-established reimbursement framework and standardized codes for airway management procedures. In the U.S., tracheostomy procedures and related care have been incorporated within the national reimbursement framework provided by the Centers for Medicare & Medicaid Services where inpatient procedures are covered under the Inpatient Prospective Payment System (IPPS) using Diagnosis-Related Groups (DRGs). This guarantees that hospitals will get structured compensation for critical care procedures requiring airway management.
For instance, Centers for Medicare & Medicaid Services (CMS) offers comprehensive coding and billing pathways to help healthcare providers with documenting and billing tracheostomy-related procedures through the official Medicare billing and coding resources. (Source: Centers for Medicare & Medicaid Services) The reimbursement-backed ecosystem eliminates financial obstacles for the hospitals, which results in the frequent use of tracheotomy tubes in ICUs and surgical settings, thereby strengthens North America’s dominance in the global tracheotomy tube market.
Why Does the Asia Pacific Tracheotomy Tube Market Exhibit High Growth?
The Asia Pacific tracheotomy tube market is expected to exhibit the fastest growth with an estimated contribution of 24.0% share to the market in 2026, attributed to the continuous development of healthcare facilities and the adoption of standardized critical care procedures by governments. Many countries in the region have been working to improve their tertiary care delivery and emergency response capacities, which significantly boosts the demand for airway management equipment.
For instance, Indian Health Facility Guidelines (HFG-India) offer elaborate guidelines on how ICU units can be effectively planned and designed by outlining specifications regarding physical infrastructure, staffing, and operational needs. These guidelines highlight the requirement for specialized ICU units equipped to handle complicated and high-acuity patients, thus promoting the increase in critical care facilities in health-care organizations. (Source: Indian Health Facility Guidelines) Such initiatives by governments, involving the strengthening of ICU infrastructure and the expansion of hospital facilities at the provincial and municipal level, have contributed to the rapid uptake of tracheotomy tubes, thus ensuring significant growth in the Asia Pacific region.
Global Tracheotomy Tube Market Outlook for Key Countries
Why is the U.S. Leading Innovation and Adoption in the Tracheotomy Tube Market?
The U.S. has the potential for leadership in innovation and adoption in the field of tracheotomy tubes due to country’s strong device innovation ecosystem and fast regulatory process that allows constant innovation and integration of new products. The U.S. Food and Drug Administration through the Center for Devices and Radiological Health ensures that advanced and high-quality medical devices are accessible quickly to allow for rapid introduction of innovative airway management technology to clinical practice.
Additionally, U.S. FDA-recognized consensus standards for respiratory and airway devices (such as ISO 5361: Anaesthetic and respiratory equipment—Tracheal tubes and connectors, and ISO 5356-1: Conical connectors for anaesthetic and respiratory equipment) provide technological innovations in airway management devices like cuff pressure control systems incorporated in tracheostomy tubes. (Source: Food and Drug Administration) The integration of regulatory backing, standardization, and product improvement gives the U.S. the edge in terms of innovation and adoption in the global tracheotomy tube market.
Is Japan a Favorable Market for Tracheotomy Tubes?
Japan emerges as a favorable market for tracheotomy tubes owing to the country’s effective regulatory structure and proper clinical standardization regarding airways management. The Pharmaceuticals and Medical Devices Agency (PMDA) guarantee the strict evaluation and approval of respiratory medical equipment. Therefore, it provides the high quality of such products and their distribution in hospitals. Besides, Japan’s health care system promotes clinical standardization and the development of hospital infrastructure; therefore, it facilitates the frequent application of tracheostomy devices in critical care and surgical settings.
Is China Emerging as a Key Growth Hub for the Tracheotomy Tube Market?
The tracheotomy tube market in China is currently experiencing significant development attributed to local production and advancements in hospital infrastructure. The National Medical Products Administration has strengthened the medical device registration process, thus permitting companies both within and outside China to increase supply. At the same time, improvements in public hospitals, along with the growth of tertiary care facilities, have increased the scope for performing airway management procedures, thus driving the adoption of tracheotomy tubes.
Why Does Germany Top the Europe Tracheotomy Tube Market?
Germany dominates the tracheotomy tube market in Europe owing to its highly organized procurement procedures within the hospitals, coupled with high levels of adherence to regulatory policies on medical devices, thus ensuring their availability and use. Both public and private hospitals have a well-established framework for their purchases, which allows for efficient procurement of critical care products such as tracheotomy tubes. Furthermore, stringent adherence to the European Commission Medical Device Regulation (EU MDR) results in high quality and extensive use of airways devices among other advantages.
Is the Tracheotomy Tube Market Developing in India?
The market for tracheotomy tubes is currently undergoing development in India owing to changes in medical device regulation as well as increased procurement of products used for airway management in public institutions. For instance, the Indian government recently brought regulation into the manufacturing of medical devices in India via the Central Drugs Standard Control Organization, in which tracheostomy tube and associated devices fall in classes requiring compliance with the Medical Device Rules, 2017. (Source: Central Drugs Standard Control Organization) Moreover, online procurement sites operated by the Government of India, such as the Government e-Marketplace (GeM), feature tracheostomy tubes among other airway devices that are eligible for procurement in large volumes.
Regulatory Landscape Governing the Global Tracheotomy Tube Market
|
Region
|
Key Regulatory Bodies |
Framework |
Impact on Market |
|
North America |
U.S. Food and Drug Administration (FDA), Health Canada |
Classified as Class II devices under FDA (21 CFR 868.5800), requiring 510(k) clearance, biocompatibility testing, and adherence to ISO 5366 airway device standards |
High compliance requirements increase approval timelines and costs, favoring established players and limiting entry of low-cost manufacturers |
|
Europe |
European Commission (under MDR), Notified Bodies |
Governed by EU Medical Device Regulation (EU MDR 2017/745) requiring CE marking, clinical evaluation, post-market surveillance, and stricter risk classification |
MDR transition (post-2024 enforcement) is causing product recertification delays and market consolidation, reducing availability of low-margin products |
|
Asia Pacific |
Central Drugs Standard Control Organization (CDSCO), National Medical Products Administration (NMPA), Pharmaceuticals and Medical Devices Agency (PMDA) |
Expanding regulation under India MDR 2017, China device registration norms, and Japan’s PMDA approval system, requiring quality certification (ISO 13485) and licensing |
Rapid regulatory tightening is driving market formalization and quality standardization, while increasing compliance costs for domestic manufacturers |
|
Latin America |
Agência Nacional de Vigilância Sanitária (ANVISA), Federal Commission for the Protection against Sanitary Risk (COFEPRIS) |
Requires device registration, GMP compliance, and technical documentation, often aligned with US FDA or EU MDR frameworks |
Gradual harmonization improves product quality and imports, but regulatory delays can slow market entry |
|
Middle East & Africa |
Saudi Food and Drug Authority (SFDA), Ministry of Health and Prevention (MOHAP), South African Health Products Regulatory Authority (SAHPRA) |
Relies on import approvals, conformity assessment, and reference country approvals (EU/US) with increasing localization of regulations |
Growing regulatory maturity is improving market access for global players, though fragmentation still creates entry complexity |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is shift toward home-based respiratory care creating new growth opportunities in the global tracheotomy tube market?
The trend towards home-based care for respiratory disorders is leading to new business prospects in the tracheotomy tube market, owing to the expansion of airway management from the hospital environment to the broader community. According to clinical guidelines and studies, a growing number of individuals with tracheostomies are being discharged from the hospital and cared for at home, facilitated by training caregivers and providing them with medical supplies like suction units and humidifiers. For instance, guidance on homecare from healthcare systems emphasizes the routine provision of tracheostomy care equipment and support training to manage such cases safely outside hospitals. (Source: Sandwell and West Birmingham NHS Trust)
Furthermore, clinical research published by National Library of Medicine, highlights the increasing relevance of self-care among patients as well as caregiver-based tracheostomy care at home. (Source: National Library of Medicine) This trend is contributing to an increased need for simpler, easy-to-manage designs of tracheotomy tubes that can be handled effectively outside clinical environments, thus boosting the market demand within the home care sector.
Market Players, Key Development, and Competitive Intelligence

To learn more about this report, Download Free Sample
Key Developments
- In June 2025, Medtronic issued a recall of certain Shiley tracheostomy tubes because of the possibility of device dislodgment arising from the separation of the flange, which would cause obstruction in the airway and endanger patients' lives. This recall was categorized as a Class I recall by the U.S. Food and Drug Administration (FDA), highlighting critical safety concerns and reinforcing the need for stringent quality and reliability standards in airway management devices.
- In November 2024, Asahi Kasei Medical Co., Ltd. launched TrachFlush, an endotracheal tube cuff inflator in Japan, targeting the management of cuff pressures in ventilated patients. This development is an indication of innovations being made in the field of airway management technologies, supporting safer and more efficient use of respiratory devices in critical care settings.
Competitive Landscape
The global tracheotomy tube market will likely stay highly competitive among manufacturers of medical devices that specialize in respiratory equipment and critical care disposables. The competitors compete based on factors such as quality of the products, regulation, and innovations in the airways management technology. Competitors are also distinguishing themselves through technological advancements in their products. For example, they are developing tubes that have cuff pressure adjustment, antimicrobial properties, and patient friendly designs. Additionally, there is a trend toward making their products suitable for home care settings and disposable ones. Key focus areas include:
- Development of ergonomic and patient-centric tracheotomy tube designs
- Expansion of homecare and long-term respiratory care product portfolios
- Strengthening regulatory compliance and quality certifications
- Partnerships with hospital procurement networks and distributors
- Focus on cost-effective, single-use and infection-control compliant devices
Market Report Scope
Tracheotomy Tube Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 236.4 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.5% | 2033 Value Projection: | USD 343.7 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Medtronic, Teleflex Incorporated, Smiths Medical, TRACOE Medical GmbH, ConvaTec Group, Fuji Systems Corporation, Sewoon Medical Co., Ltd., Well Lead Medical Co., Ltd., TuoRen Medical, and Pulmodyne Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- In the coming years, the prospects of the tracheotomy tube market will undergo a shift toward a continuum-of-care approach, whereby such devices will seamlessly integrate usage throughout ICU stay and step-down units as well as after discharge. Future market success for such devices will rely more on innovation-driven product lines which will include, for example, improved cuff pressure control technology, sub-glottic aspiration abilities, and materials conducive for patients' comfort than simple commodity devices. This trend, however, will be supported by a growing need for standardization of airway management procedures as well as stringent product specifications.
- The greatest opportunity exists in cuffed tracheotomy tubes in the U.S. as demand will arise from the established procedures in managing the critically ill patients, thus, creating a good market for expensive but innovative equipment. On the other hand, there exists a volume-oriented opportunity in China due to the expanding hospital networks, local manufacturing, and availability of affordable respiratory consumables.
- For gaining any competitive advantage in the market, companies need to move from being simply product-suppliers to becoming value-creators through the creation of devices that minimize caregiver strain and ease of use as well as integration with ventilators. In addition, companies must also ensure that they develop localized production facilities in order to remain cost-competitive in their efforts to penetrate emerging markets, and that they are capable of accelerating approval and market entry through regulatory prowess. Finally, forming partnerships and providing training programs for tracheostomies is essential.
Market Segmentation
- Product Type Insights (Revenue, USD Mn, 2021 - 2033)
- Cuffed Tracheotomy Tubes
- Uncuffed Tracheotomy Tubes
- Fenestrated Tubes
- Adjustable/Specialty Tubes
- Material Insights (Revenue, USD Mn, 2021 - 2033)
- PVC
- Silicone
- Others
- Patient Type Insights (Revenue, USD Mn, 2021 - 2033)
- Adult
- Pediatric/Neonatal
- Application Insights (Revenue, USD Mn, 2021 - 2033)
- Long-Term Ventilation
- Emergency Airway Management
- Chronic Disease Management
- Post-operative Airway Support
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Hospitals
- Ambulatory Surgical Centers
- Home Healthcare Settings
- Others
- Regional Insights (Revenue, USD Mn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Medtronic
- Teleflex Incorporated
- Smiths Medical
- TRACOE Medical GmbH
- ConvaTec Group
- Fuji Systems Corporation
- Sewoon Medical Co., Ltd.
- Well Lead Medical Co., Ltd.
- TuoRen Medical
- Pulmodyne Inc.
Sources
Primary Research Interviews
- Critical care specialists, anesthesiologists, and ENT surgeons from leading hospitals and ICU centers
- Medical device manufacturers specializing in respiratory and airway management devices
- Hospital procurement managers and biomedical engineers involved in ICU equipment sourcing
- Home healthcare providers and respiratory therapists managing long-term tracheostomy care
Stakeholders
- Tracheotomy Tube Manufacturers: (e.g., medical device companies, respiratory care solution providers, critical care equipment manufacturers)
- End-use Sectors:
- Hospitals and Intensive Care Units (ICUs)
- Ambulatory Surgical Centers
- Homecare Settings / Long-term Care Providers
- Emergency and Trauma Care Centers
- Regulatory & Health Bodies: U.S. Food and Drug Administration (FDA) – device approvals, classification, and safety regulations for tracheostomy tubes, European Commission – Medical Device Regulation (EU MDR) compliance frameworks, World Health Organization – global respiratory disease burden and critical care guidance, Central Drugs Standard Control Organization – medical device rules and classification in India
Databases
- ClinicalTrials.gov – clinical studies on airway management devices and respiratory interventions
- WHO Global Health Observatory – respiratory disease and healthcare infrastructure data
- PubMed / NCBI – clinical literature on tracheostomy procedures and outcomes
Magazines
- Nature Medicine / Translational Medicine – advancements in respiratory care and medical devices
- Science Magazine – innovations in biomaterials and airway device technologies
- NIH Research Matters – updates on respiratory health and clinical innovations
Journals
- American Journal of Respiratory and Critical Care Medicine
- Journal of Critical Care
- Intensive Care Medicine
- Respiratory Care Journal
Newspapers
- The New York Times (Health Section) – coverage on ICU care trends and respiratory therapies
- The Guardian (Health & Science) – reporting on healthcare infrastructure and critical care
- Financial Times – medical device industry developments and healthcare investments
Associations
- American Association for Respiratory Care (AARC)
- European Society of Intensive Care Medicine (ESICM)
- World Federation of Societies of Intensive and Critical Care Medicine (WFSICCM)
- Difficult Airway Society (DAS)
Public Domain Sources
- National Institutes of Health (NIH) – airway management and respiratory research
- Centers for Disease Control and Prevention (CDC) – ICU care and respiratory disease insights
- European Commission – healthcare regulatory frameworks and device policies
- National health ministries (e.g., U.S., China, Japan, India) – critical care programs and infrastructure initiatives
Proprietary Elements
- CMI Data Analytics Tool
- Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients