Intrapartum Monitoring Devices Market Size and Forecast – 2026 – 2033
The Global Intrapartum Monitoring Devices Market size is estimated to be valued at USD 945 million in 2026 and is expected to reach USD 1,528 million by 2033, exhibiting a compound annual growth rate (CAGR) of 7.2% from 2026 to 2033.
Global Intrapartum Monitoring Devices Market Overview
The intrapartum monitoring devices market comprises a range of products designed to track maternal and fetal health during labor. Key offerings include fetal heart rate monitors, cardiotocography (CTG) systems, and electronic fetal monitoring (EFM) devices that provide real-time data on fetal well-being. Additional products include maternal vital signs monitors, contraction monitors, and integrated multiparameter systems that combine fetal and maternal monitoring for comprehensive care. Non-invasive and invasive monitoring options cater to different clinical scenarios, while portable and wireless devices enhance mobility and patient comfort. These products are widely adopted in hospitals, maternity clinics, and birthing centers to improve labor outcomes and ensure safety.
Key Takeaways
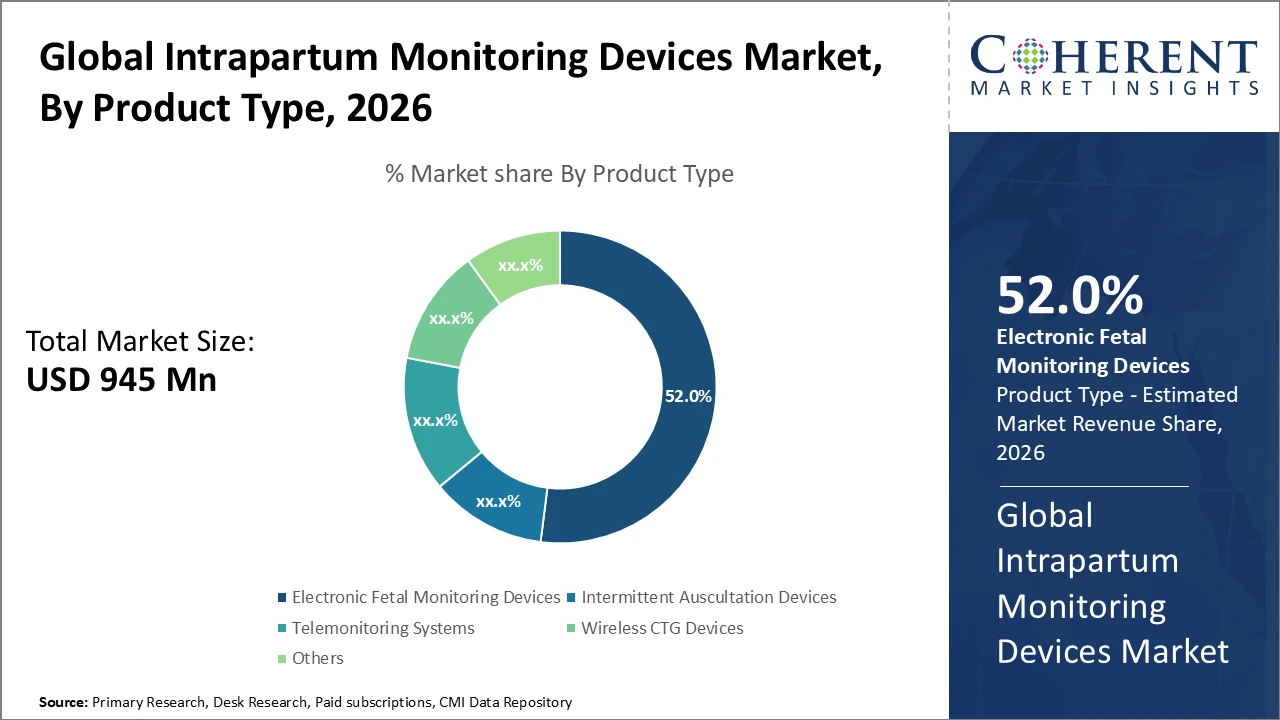
The Electronic Fetal Monitoring (EFM) segment holds over 52% of the market share, driven by its reliability and continuous monitoring capabilities, establishing it as the dominant technology.
Telemonitoring systems are expected to experience the fastest growth due to increasing healthcare digitization and remote patient management adoption.
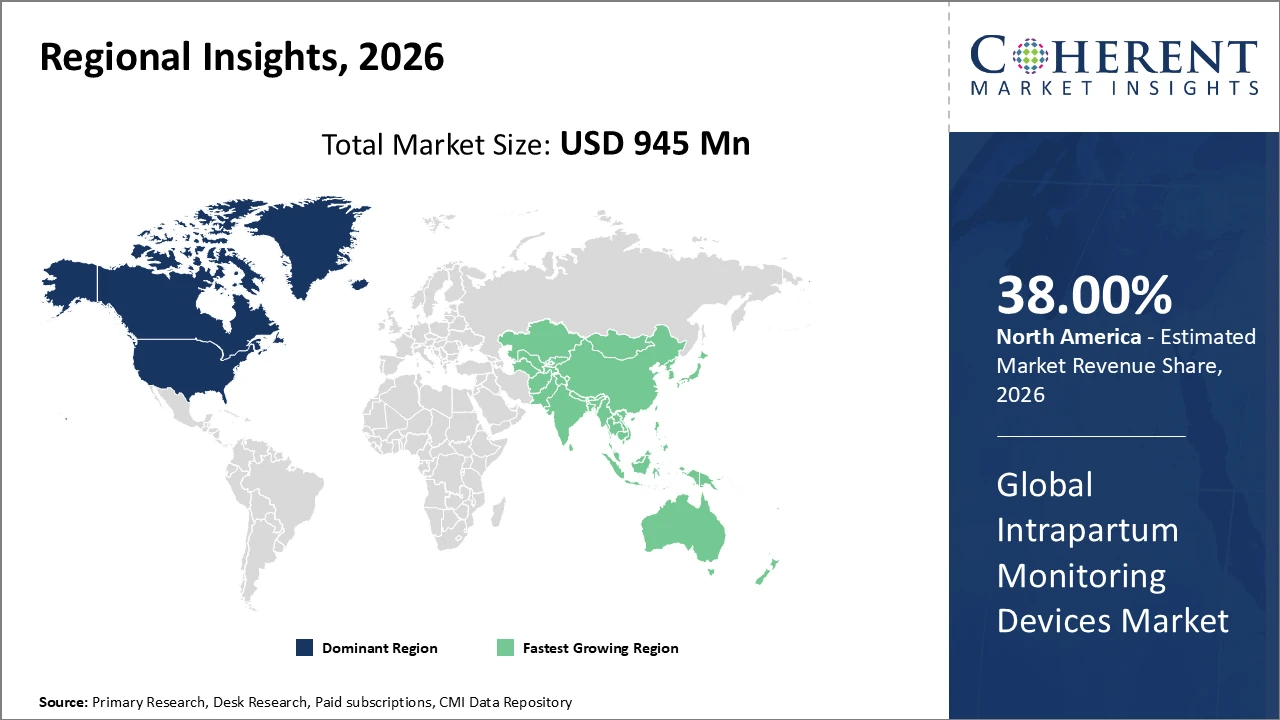
North America leads the market with approximately 38% share, supported by advanced healthcare infrastructure and strong uptake of innovative monitoring technologies.
Asia Pacific is the fastest-growing region, with a CAGR exceeding 9%, fueled by rising healthcare spending and the expansion of obstetric care facilities.
Intrapartum Monitoring Devices Market Segmentation Analysis
To learn more about this report, Request Free Sample
Intrapartum Monitoring Devices Market Insights, By Product Type
Electronic Fetal Monitoring (EFM) devices dominate the intrapartum monitoring market with a share of 52%, providing continuous fetal heart rate and uterine contraction tracking, and are widely preferred in developed regions for accurate detection of fetal distress. Integration with advanced analytics has further boosted adoption in tertiary care centers. Wireless CTG devices are the fastest-growing subsegment, offering enhanced patient comfort, mobility, and real-time data transmission to clinicians, supporting non-hospital deliveries. Intermittent auscultation remains relevant in low-resource or outpatient settings due to its cost-effectiveness.
Intrapartum Monitoring Devices Market Insights, By Application
Hospitals hold the largest share in the intrapartum monitoring devices market due to established infrastructure and high delivery volumes requiring advanced monitoring. Continuous and reliable devices, such as EFM and wireless CTGs, are essential for managing high-risk births in these settings. Ambulatory Surgical Centers and Maternity Clinics represent significant segments, increasingly adopting monitoring solutions to enhance patient outcomes in less acute environments. The Homecare Setting is the fastest-growing segment, driven by home birth trends and telemonitoring technologies that enable safe labor outside hospitals.
Intrapartum Monitoring Devices Market Insights, By End User
Obstetricians and Gynecologists comprise the largest user group in the intrapartum monitoring devices market, as they manage high-risk labor and consistently rely on electronic and wireless fetal monitoring technologies. Midwives and nurses form the fastest-growing segment, supported by portable and user-friendly monitoring devices that enable non-physician providers to deliver effective care in hospitals and home settings, reflecting the rise of midwife-led birthing centers worldwide. Healthcare institutions, including clinics and specialized maternal centers, implement comprehensive monitoring systems to ensure quality care and regulatory compliance.
Intrapartum Monitoring Devices Market Trends
Current market trends in intrapartum monitoring devices emphasize AI-powered analytics and wireless technologies.
By 2026, leading U.S. hospitals implemented AI-assisted CTG systems, improving early fetal distress detection by 15%, highlighting technology’s clinical impact.
Telemonitoring adoption has increased, driven by pandemic-induced telehealth expansion, allowing continuous remote patient monitoring.
Remote data sharing with obstetric care specialists enhances responsiveness and care quality.
Integration into digital health ecosystems improves workflow efficiency and reduces manual intervention in high-risk labor scenarios.
Intrapartum Monitoring Devices Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Intrapartum Monitoring Devices Market Analysis and Trends
In North America, the intrapartum monitoring devices market leads with around 38% market share, driven by advanced healthcare infrastructure, high expenditure, and supportive maternal health policies. Widespread reimbursement programs and the adoption of AI-powered monitoring systems in key hospitals further boost revenue. Leading companies like Philips Healthcare and GE Healthcare maintain a strong presence, deploying advanced fetal and maternal monitoring devices across major medical centers in the U.S. and Canada. This combination of robust infrastructure, favorable policies, and technological adoption solidifies North America’s dominance, supporting consistent market growth and encouraging innovation in intrapartum care solutions throughout the region.
Asia Pacific Intrapartum Monitoring Devices Market Analysis and Trends
Asia Pacific is the fastest-growing market for intrapartum monitoring devices, with a CAGR exceeding 9%, fueled by rapid healthcare infrastructure development in countries such as India and China and rising cesarean section rates. Government maternal health programs and increasing investment in digital health technologies support the adoption of advanced monitoring solutions. Leading companies, including Mindray Medical and Siemens Healthineers, have strategically expanded their presence in the region, offering cost-effective and locally tailored intrapartum monitoring devices. These initiatives address the growing demand for quality maternal care in emerging economies, positioning Asia Pacific as a key growth hub for the global intrapartum monitoring devices market.
Intrapartum Monitoring Devices Market Outlook for Key Countries
USA Intrapartum Monitoring Devices Market Analysis and Trends
The U.S. intrapartum monitoring devices market represents a significant share of global revenue, driven by rising high-risk pregnancies and technological advancements. By 2026, more than 85% of hospitals had adopted electronic fetal monitoring systems, with increasing integration of wireless and AI-assisted devices. Supportive regulatory frameworks, including FDA approvals, along with value-based healthcare models, have accelerated market adoption. Leading companies such as GE Healthcare and Philips Healthcare continue to innovate, incorporating AI technologies into fetal monitoring systems. These advancements enhance prenatal care quality, enable early detection of fetal distress, and reinforce the United States’ position as a global leader in intrapartum monitoring solutions.
Germany Intrapartum Monitoring Devices Market Analysis and Trends
In Germany, the intrapartum monitoring devices market is experiencing steady growth supported by strong healthcare infrastructure and high standards for maternal and fetal care. Hospitals and maternity clinics routinely adopt advanced monitoring systems, including electronic fetal monitors and wireless CTG devices, to improve clinical outcomes and manage high‑risk deliveries, aligning with broader European healthcare quality standards. Technological advancements such as non‑invasive sensors and AI‑enabled analytics are gradually influencing device adoption, while government initiatives aimed at reducing maternal and infant mortality support increased investment in obstetric monitoring solutions. The market is also influenced by rising awareness of maternal health and expanding digital health integration.
Analyst Opinion
Enhanced demand for wireless intrapartum monitoring solutions is reshaping market dynamics, with hospital adoption of wireless cardiotocography (CTG) devices rising approximately 25% in 2025, driven by patient mobility and comfort priorities. Wireless monitoring is projected to account for nearly 40% of total market share by 2030.
Rising utilization of telemonitoring in labor wards has expanded market scope; for example, a UK NHS trust implemented remote fetal monitoring in 2024, reducing in-person visits by 18% and promoting decentralized maternal care.
Cost optimization strategies influence growth for mid-tier devices, with adoption of intermittent fetal monitoring systems increasing 15% in emerging markets like India and Brazil in 2025.
Regulatory approvals and government programs targeting neonatal mortality have accelerated adoption of innovative biometric sensor technologies, with Southeast Asian maternal health initiatives in 2026 driving a 22% increase in electronic fetal monitoring system demand.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 945 million |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.2% | 2033 Value Projection: | USD 1,528 million |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | GE Healthcare, Philips Healthcare, Siemens Healthineers, Medtronic PLC, Ambu A/S, Cook Medical, Analogic Corporation, Noraxon USA Inc., Natus Medical Incorporated, ConvaTec Group PLC | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Intrapartum Monitoring Devices Market Growth Factors
Rising investments in maternal healthcare infrastructure have significantly influenced the intrapartum monitoring devices market, with U.S. government funding increasing by 20% between 2024 and 2026, driving demand in hospitals and specialized clinics. Growing awareness of fetal distress and related complications has also contributed, with regions adopting advanced monitoring systems reporting a 6% decline in neonatal mortality in 2025. Globally rising cesarean section rates, particularly in Europe and Asia Pacific, have further increased the need for continuous fetal monitoring. Technological innovations, including wireless and telemonitoring solutions, enhance remote patient management, improving efficiency, clinical outcomes, and overall market dynamics.
Intrapartum Monitoring Devices Market Development
In February 2026, Assam launched a statewide intrapartum central monitoring network, powered by indigenous technology from Janitri Innovations Pvt. Ltd., to enhance maternal and fetal monitoring across government healthcare facilities through improved clinical oversight.
Key Players
Leading Companies of the Market
GE Healthcare
Philips Healthcare
Siemens Healthineers
Medtronic PLC
Ambu A/S
Cook Medical
ConVaTec Group PLC
Noraxon USA Inc.
Natus Medical Incorporated
Analogic Corporation
Philips Healthcare’s strategic partnership with regional manufacturers in 2025 expanded its distribution network across Asia Pacific, resulting in a 12% increase in market share. GE Healthcare introduced AI-enabled fetal monitoring in 2026, improving diagnostic precision by 18% and strengthening its competitive positioning in developed markets. Mindray Medical focused on cost-efficient intrapartum monitoring devices for emerging economies, particularly in Latin America, enhancing accessibility and expanding its regional footprint. These initiatives reflect how major players leverage partnerships, technological innovation, and market-specific strategies to drive growth, improve patient outcomes, and maintain leadership in the global intrapartum monitoring devices market.
Intrapartum Monitoring Devices Market Future Outlook
The future outlook for the intrapartum monitoring devices market is highly promising, driven by increasing adoption of advanced technologies such as AI-assisted analytics, wireless CTG devices, and telemonitoring solutions. Rising maternal healthcare investments, growing awareness of fetal distress, and expanding hospital and homecare services in emerging markets will further fuel growth. Continuous innovation in portable, non-invasive, and cost-effective monitoring systems is expected to enhance patient comfort and clinical outcomes. Government initiatives targeting maternal and neonatal health, coupled with digital health integration, will support market expansion. Overall, the market is projected to experience robust growth, with emerging regions offering significant opportunities for stakeholders.
Intrapartum Monitoring Devices Market Historical Analysis
The intrapartum monitoring devices market has experienced steady growth over the past decade, driven by increasing focus on maternal and fetal safety and rising hospital birth volumes. Early adoption centered on electronic fetal monitoring (EFM) systems in developed regions, where continuous monitoring improved detection of fetal distress and reduced neonatal complications. Over time, technological advancements such as wireless CTG devices, telemonitoring, and AI-assisted analytics expanded market adoption, while hospitals and maternity clinics increasingly invested in reliable, cost-effective solutions. North America and Europe initially dominated, but emerging economies in Asia Pacific and Latin America gradually contributed to growth due to infrastructure development and rising healthcare awareness.
Sources
Primary Research Interviews:
Executives and operations managers at medical device manufacturing and refurbishment companies specializing in intrapartum monitoring devices
Obstetricians, gynecologists, midwives, and hospital procurement officers overseeing labor and delivery monitoring
Technology providers for electronic fetal monitoring (EFM), wireless CTG devices, telemonitoring systems, and maternal-fetal monitoring consumables
Magazines:
Medical Device News Magazine – Advances in Fetal and Maternal Monitoring Equipment
Healthcare Technology Today – Updates on Intrapartum Monitoring Solutions
Hospital Management Asia/Europe – Trends in Obstetric Care and Monitoring Devices
Clinical Innovation & Technology – Adoption of Wireless and AI-Enabled Monitoring Devices
Journals:
Journal of Clinical Engineering – Intrapartum Monitoring Devices in Hospital Settings
Biomedical Instrumentation & Technology – Innovations in Fetal and Maternal Monitoring
International Journal of Gynecology & Obstetrics – Clinical Applications of Intrapartum Monitoring
Journal of Healthcare Engineering – Workflow Optimization with Monitoring Devices
Newspapers:
The Wall Street Journal – Maternal Health Devices and Hospital Technology Markets
Financial Times – Trends in Obstetric Equipment Procurement and Adoption
Medical Device News – Intrapartum Monitoring Device Developments and Market Updates
Healthcare Innovation – Patient Safety and Advanced Maternal Monitoring
Associations:
American College of Obstetricians and Gynecologists (ACOG) – Guidelines for Fetal and Maternal Monitoring
International Federation of Gynecology and Obstetrics (FIGO) – Obstetric Monitoring Standards and Research
Society for Maternal-Fetal Medicine (SMFM) – Best Practices in Labor Monitoring
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients