Fetal Monitoring Market Size and Forecast – 2026 – 2033
The Global Fetal Monitoring Market size is estimated to be valued at USD 4.5 billion in 2026 and is expected to reach USD 7.8 billion by 2033, exhibiting a compound annual growth rate (CAGR) of 8.2% from 2026 to 2033.
Global Fetal Monitoring Market Overview
The Fetal Monitoring market includes a range of products designed to track and assess fetal health during pregnancy and labor. Key products comprise electronic fetal monitoring (EFM) systems, which provide continuous monitoring of fetal heart rate and uterine contractions, and handheld Doppler devices used for intermittent auscultation. Advanced solutions include non-invasive prenatal testing (NIPT) devices and wireless fetal monitors that enable remote tracking and real-time data transmission. Other products encompass cardiotocography (CTG) machines, fetal scalp electrodes, and maternal-fetal telemetry systems. These devices are widely adopted in hospitals, clinics, and home care settings to improve prenatal care, early detection of complications, and clinical decision-making.
Key Takeaways
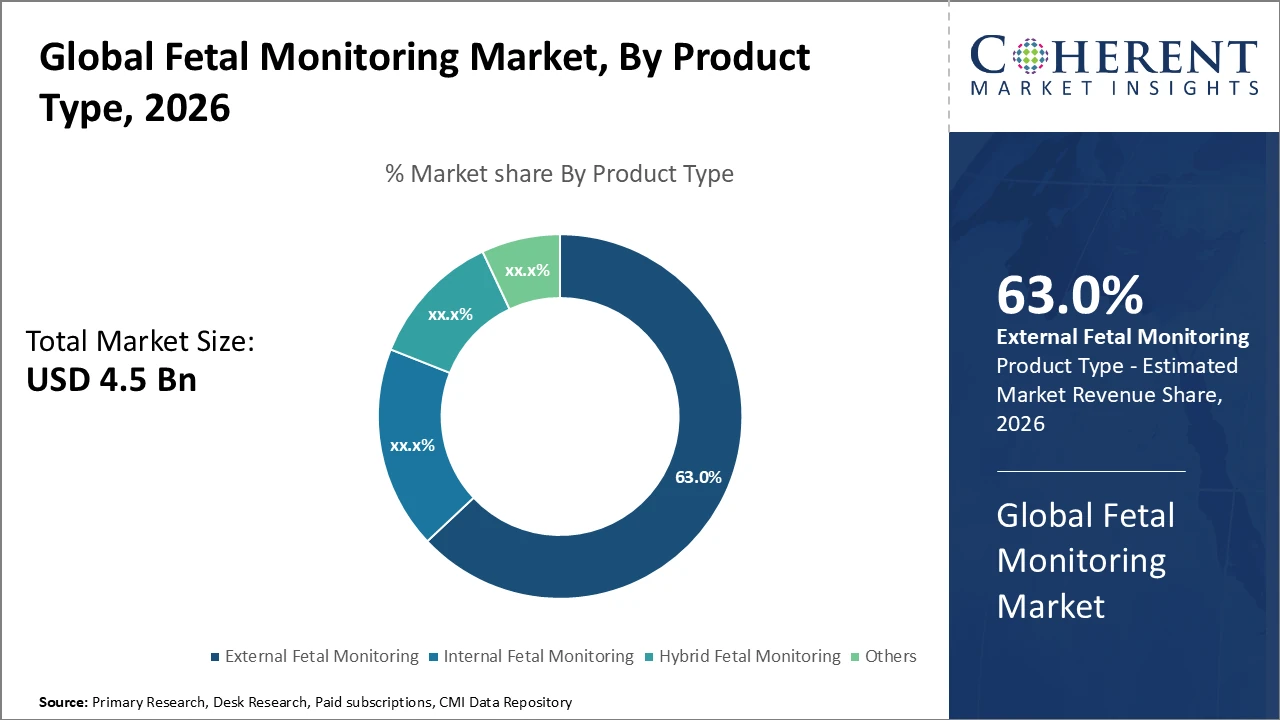
External Fetal Monitoring dominates the product segment, accounting for 63% of the market due to its non-invasive nature and wide clinical acceptance.
Ultrasound-based monitoring leads the technology segment, driven by high accuracy, real-time feedback, and integration into standard prenatal care protocols.
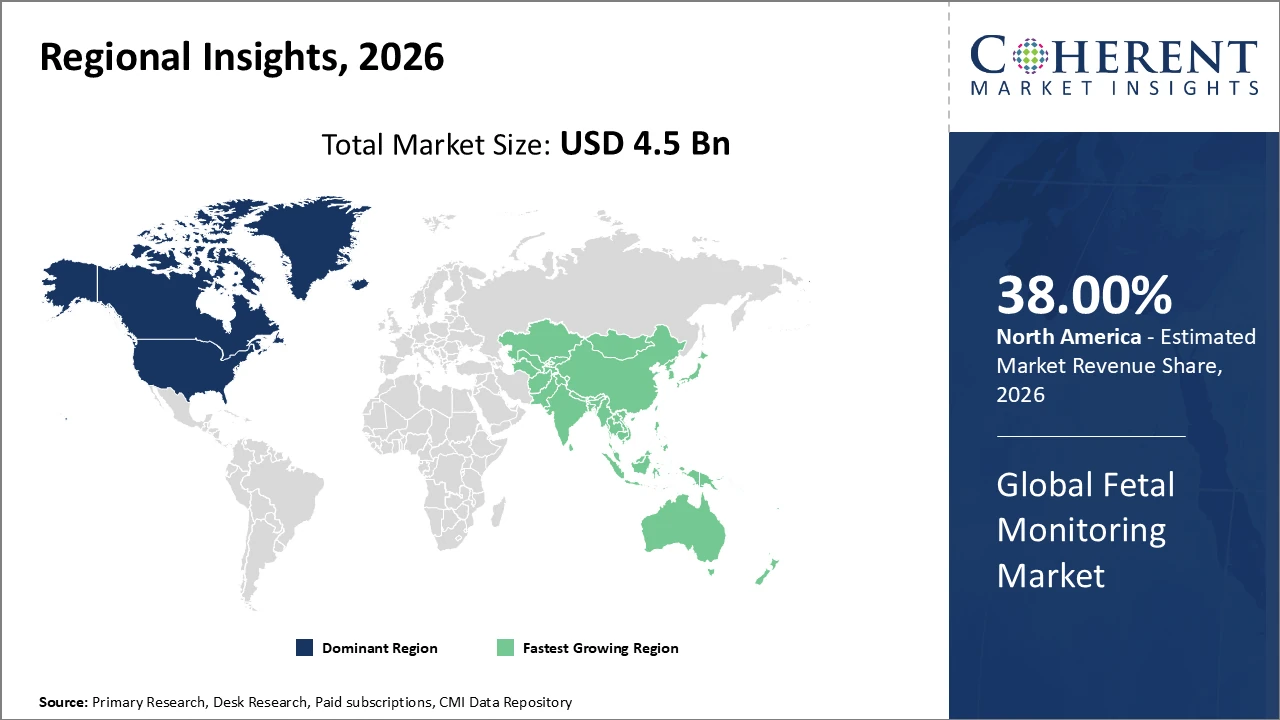
North America holds the largest regional market share, supported by strong healthcare infrastructure, well-established reimbursement frameworks, and high adoption of advanced fetal monitoring devices.
Asia Pacific is projected to exhibit the fastest CAGR, fueled by increasing healthcare investments, rising birth rates, and expanding access to maternal healthcare in countries like India and China.
Europe remains a significant revenue contributor, with growing adoption of advanced fetal surveillance technologies and regulatory emphasis on maternal-fetal health outcomes.
Fetal Monitoring Market Segmentation Analysis
To learn more about this report, Request Free Sample
Fetal Monitoring Market Insights, By Product Type
External Fetal Monitoring dominates the market due to its widespread clinical acceptance, non-invasive nature, and cost-effectiveness, making it the preferred choice in most healthcare facilities. It enables real-time monitoring of fetal heart rates and uterine contractions, supporting timely interventions during labor. Internal Fetal Monitoring is increasingly adopted in high-risk pregnancies, providing greater accuracy through direct physiological data acquisition and steadily expanding its market presence. Hybrid Fetal Monitoring combines external and internal approaches, balancing precision with patient comfort, and is particularly useful in complex cases. Other fetal monitoring devices, while a smaller segment, are emerging with innovative features for specialized clinical needs.
Fetal Monitoring Market Insights, By Technology
Ultrasound-Based technology dominates the fetal monitoring market due to its real-time, non-invasive diagnostic capabilities, making it the standard for tracking fetal heart rate and movement throughout pregnancy. Within this segment, Non-Invasive Monitoring is the fastest-growing technology, driven by advancements in wearable sensors and wireless connectivity, enabling outpatient and remote monitoring solutions. Electromechanical and Invasive Monitoring methods continue to serve specialized hospital settings where precise, comprehensive data is essential. The Others category encompasses emerging technologies, including photoplethysmography-based sensors, which show promising potential for fetal assessment but remain in early developmental and clinical evaluation stages.
Fetal Monitoring Market Insights, By End-User
Hospitals dominate the fetal monitoring market, driven by high delivery volumes and access to advanced monitoring infrastructure. Large medical centers invest in state-of-the-art fetal monitoring systems to manage complex pregnancies and ensure timely interventions. Homecare Settings are the fastest-growing segment, reflecting increased adoption of telehealth and remote monitoring solutions driven by patient preference and technological innovations. Ambulatory Surgical Centers and Maternity Clinics serve less complex cases but contribute to market growth through rising outpatient care trends. The Others category includes specialized research institutions and training facilities that use fetal monitoring technologies for educational, clinical research, and skill development purposes.
Fetal Monitoring Market Trends
AI-based fetal monitoring solutions captured an 18% share of new technology adoptions globally in 2026, enhancing fetal distress detection accuracy and lowering false positives.
Non-invasive, wireless fetal monitoring devices are increasingly preferred for patient comfort, with North America seeing a 25% adoption growth in 2025.
Telehealth expansion during the COVID-19 pandemic accelerated the development of remote fetal monitoring platforms.
Cloud-enabled fetal monitoring systems support scalable data management and enable monitoring across multiple locations.
Europe and Asia Pacific markets experienced significant growth in remote and connected fetal monitoring adoption.
Fetal Monitoring Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Fetal Monitoring Market Analysis and Trends
In North America, the fetal monitoring market is dominated by strong healthcare infrastructure, significant R&D investments, and favorable reimbursement policies, collectively driving a market share of around 38%. The U.S., in particular, emphasizes digital health integration, fostering the adoption of advanced monitoring solutions, including AI-enabled and wireless devices. Major industry players, such as Philips and GE Healthcare, play a crucial role in advancing technology, expanding product portfolios, and enhancing clinical outcomes. This combination of supportive policies, innovation focus, and established market leaders ensures sustained growth, increased revenue generation, and a competitive edge in the global fetal monitoring landscape.
Asia Pacific Fetal Monitoring Market Analysis and Trends
The Asia Pacific fetal monitoring market is experiencing the fastest growth, with a CAGR exceeding 9%, driven by rising birth rates, growing maternal health awareness, and expanding healthcare infrastructure in countries like India and China. Government initiatives focused on maternal safety, including funding for modern equipment and improved hospital capacity, are further boosting adoption of advanced monitoring solutions. Additionally, increasing investments in digital health technologies and training programs for healthcare professionals support the integration of AI-enabled and wireless fetal monitoring devices. These combined factors position the region as a rapidly expanding market, attracting both global and local players seeking growth opportunities.
Fetal Monitoring Market Outlook for Key Countries
USA Fetal Monitoring Market Analysis and Trends
The U.S. fetal monitoring market remains a global leader, driven by advanced clinical adoption and high healthcare spending. Recent FDA approvals of AI-driven fetal monitoring devices have significantly enhanced diagnostic accuracy, leading to a 15% rise in product utilization in 2026. The integration of remote fetal monitoring platforms has expanded prenatal care access, especially in rural and underserved regions, improving maternal and fetal outcomes. Strong emphasis on digital health, combined with robust infrastructure and the presence of major industry players, reinforces the country’s position as a hub for innovation, driving market revenue growth and setting benchmarks for global fetal monitoring practices.
Germany Fetal Monitoring Market Analysis and Trends
The Germany fetal monitoring market is characterized by steady growth supported by advanced healthcare infrastructure and high clinical standards, with market size estimates around €200 million and a CAGR of roughly 6–9% through the late 2020s, reflecting robust demand for both traditional and innovative monitoring systems. Continuous electronic fetal monitoring remains the dominant segment, but portable, wireless, and remote-capable devices are gaining traction, especially in outpatient and home care settings. Growing maternal health awareness, rising high‑risk pregnancies, and strong reimbursement frameworks further drive adoption, while stringent EU regulatory requirements shape product development and market entry.
Analyst Opinion
Continuous fetal monitoring technology advancements are driving market growth, with hospital acquisitions of wireless fetal monitors rising 15% in 2025, reflecting preference for non-invasive, real-time solutions and boosting market revenue.
The global increase in cesarean section rates, reported at 21% higher in 2026 than in 2023 by the World Health Organization, is intensifying demand for precise fetal distress detection and monitoring devices, expanding market share.
Nano-sensor technologies in fetal scalp electrodes have improved fetal heart rate monitoring accuracy, reducing false positives by 12% in 2024, supporting adoption in high-risk pregnancy centers.
Wearable fetal monitors with remote data transmission are alleviating hospital resource constraints, broadening market scope.
Export of fetal monitoring devices from Asia Pacific grew 18% in 2026, fueled by cost-effective manufacturing and increased regional healthcare investments, enhancing global market growth opportunities.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 4.5 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.2% | 2033 Value Projection: | USD 7.8 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Philips Healthcare, GE Healthcare, Medtronic, Hill-Rom Holdings, Edan Instruments, Becton Dickinson, Samsung Medison, Sonicaid Limited, Natus Medical Incorporated | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Fetal Monitoring Market Growth Factors
The global demand for advanced fetal monitoring tools has surged due to the rising incidence of high-risk pregnancies, aiming to reduce complications. In 2025, cases of gestational diabetes rose by 11%, highlighting the need for continuous fetal surveillance. The integration of artificial intelligence in monitoring devices has improved the prediction of fetal distress, with AI-based fetal heart rate analysis adoption increasing by 25% in 2026 across major maternity hospitals. Government initiatives supporting maternal health, including enhanced funding for hospital infrastructure in emerging economies, have further expanded market opportunities. The acceleration of telehealth adoption in the post-pandemic era led to a 22% growth in remote fetal monitoring in 2025, improving access to prenatal care. Moreover, growing global awareness of fetal health has driven market revenue, as patients increasingly prefer reliable, non-invasive monitoring solutions.
Fetal Monitoring Market Development
In October 2025, the University of Maryland Medical System launched NEST (Neonatal Outcomes Impacted by Escalation Safety Telemetry), a remote fetal monitoring center that enables real-time monitoring of labor and delivery across its hospitals.
Key Players
Leading Companies of the Market
Philips Healthcare
GE Healthcare
Hill-Roms Holdings
Edan Instruments
Becton Dickinson
Samsung Medison
Sonicaid Limited
Natus Medical Incorporated
Several leading companies have recently focused on strategic partnerships and acquisitions to enhance technological capabilities and expand their geographic reach. In 2025, Philips Healthcare acquired a telemonitoring startup, significantly advancing its remote fetal monitoring solutions and contributing to a 15% increase in its North American market share by 2026. Similarly, GE Healthcare expanded its fetal monitoring product portfolio in 2024 by integrating AI-assisted analytics, improving diagnostic accuracy and strengthening its presence across Europe and the Asia Pacific region. These strategic moves reflect the industry’s focus on innovation and market consolidation to meet growing demand for advanced, reliable fetal monitoring technologies globally.
Fetal Monitoring Market Future Outlook
The fetal monitoring market is expected to experience robust growth in the coming years, driven by rising high-risk pregnancies and increasing adoption of advanced technologies. Artificial intelligence and machine learning will continue to enhance predictive capabilities, enabling earlier detection of fetal distress. Remote and wearable monitoring solutions are likely to expand, improving prenatal care access, especially in rural and underserved regions. Government initiatives promoting maternal health and rising patient awareness of non-invasive monitoring options will further boost demand. Strategic partnerships, technological innovations, and telehealth integration are projected to shape a dynamic market landscape, with a focus on safety, accuracy, and convenience.
Fetal Monitoring Market Historical Analysis
The fetal monitoring market has witnessed steady growth over the past decade, driven by advances in technology and increasing awareness of maternal and fetal health. Traditional electronic fetal monitoring systems dominated early adoption, primarily in developed regions, focusing on hospital-based care. Over time, integration of digital solutions and AI enhanced monitoring accuracy, enabling early detection of fetal distress. Rising incidences of high-risk pregnancies, including gestational diabetes and preeclampsia, further fueled demand for continuous surveillance. Telehealth and remote monitoring began gaining traction post-2020, expanding access in emerging economies. These historical trends laid the foundation for today’s innovation-driven and patient-centric fetal monitoring landscape.
Sources
Primary Research Interviews:
Hospitals, clinics, and surgical centers
Obstetrics and gynecology departments
Research laboratories and academic institutions
Medical device consultants specializing in fetal monitoring and perinatal care
Magazines:
Medical Device & Diagnostic Industry (MD+DI) – Fetal Monitoring Section
HealthTech Magazine – Maternal and Fetal Health Technologies
OB/GYN News – Fetal Monitoring and Maternal Care Updates
Healthcare Innovation – Prenatal Care Technologies
Journals:
Journal of Perinatology
American Journal of Obstetrics & Gynecology
Prenatal Diagnosis
Journal of Maternal-Fetal & Neonatal Medicine
BMC Pregnancy and Childbirth
Newspapers:
The Wall Street Journal – Medical Devices & Maternal Health
Financial Times – Healthcare Technology
Reuters – Medical Equipment & Fetal Monitoring
Bloomberg – Medical Devices Market
Nikkei Asia – Healthcare and Technology
Associations:
International Federation of Gynecology and Obstetrics (FIGO)
American College of Obstetricians and Gynecologists (ACOG)
Society for Maternal-Fetal Medicine (SMFM)
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients