Uterine Cancer Drugs Market Size and Forecast – 2026 – 2033
The Global Uterine Cancer Drugs Market size is estimated to be valued at USD 1.90 billion in 2026 and is expected to reach USD 3.41 billion by 2033, exhibiting a compound annual growth rate (CAGR) of 8.7% from 2026 to 2033.
Global Uterine Cancer Drugs Market Overview
Uterine cancer drugs include chemotherapy agents (carboplatin, paclitaxel), hormonal therapies (progestins, aromatase inhibitors), targeted agents (lenvatinib, everolimus), and immunotherapies (pembrolizumab, dostarlimab). These products are used alone or in combination to treat endometrial and other uterine cancers. Oral, intravenous, and injectable formulations are available depending on the drug type and treatment phase. Many newer drugs are guided by molecular diagnostics, such as mismatch repair deficiency or PD-L1 expression, supporting personalized treatment regimens and improved patient outcomes.
Key Takeaways
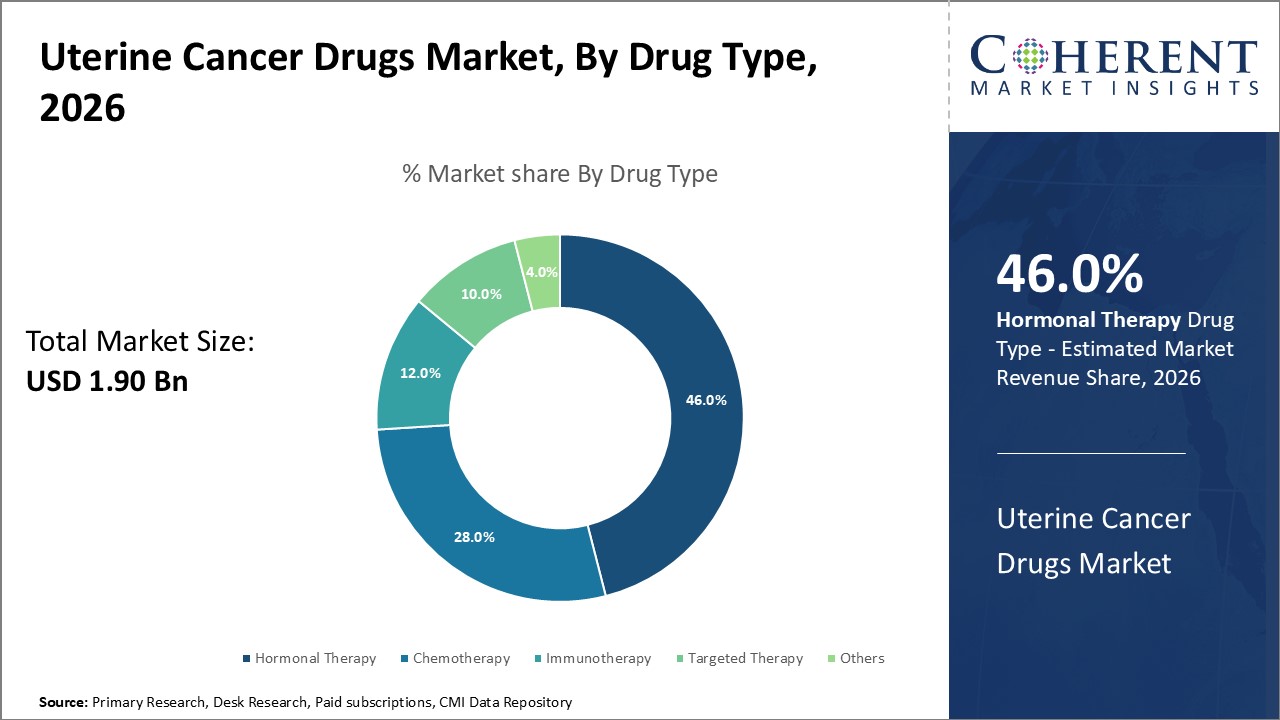
The Hormonal Therapy segment leads the uterine cancer drugs market with a 46% share, driven by its application in early and recurrent stages and steady clinical outcomes.
Immunotherapy is the fastest-growing subsegment, owing to breakthrough approvals of immune checkpoint inhibitors that are expanding treatment options and fueling market revenue growth.
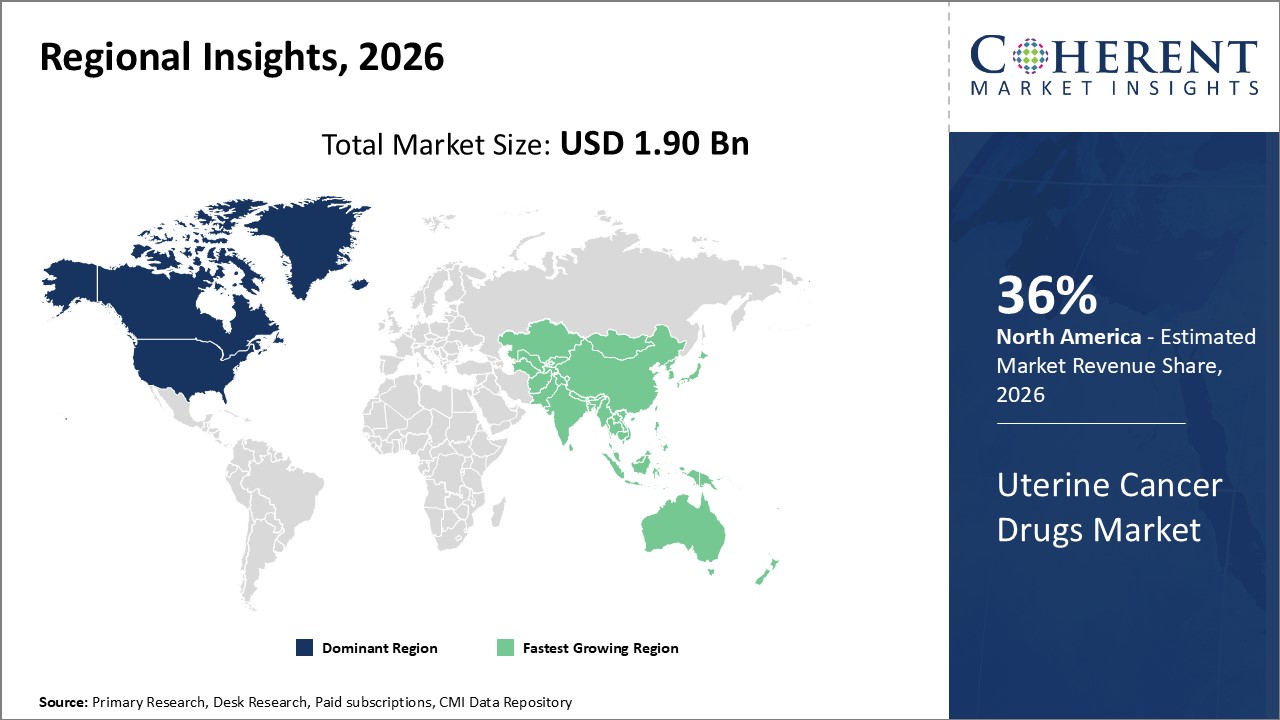
North America dominates with a commanding market share of approximately 36%, reflective of robust healthcare infrastructure and early adoption of cutting-edge uterine cancer drugs.
Asia Pacific is the fastest-growing region, registering a CAGR of around 10%, propelled by increasing disease prevalence and the expansion of oncology centers in China and India.
Uterine Cancer Drugs Market Segmentation Analysis
To learn more about this report, Request Free Sample
Uterine Cancer Drugs Market Insights, By Drug Type
Hormonal Therapy dominates the market share at 46% due to its efficacy in managing early and recurrent uterine cancers, supported by established treatment protocols and consistent clinical results. The fastest growing subsegment is Immunotherapy, driven by breakthrough approvals of immune checkpoint inhibitors and growing clinical evidence supporting its use in advanced stages. Chemotherapy continues to serve as a foundational treatment but experiences moderate growth due to emerging targeted options. Targeted Therapy is gaining momentum as more molecular pathways are identified and specific inhibitors are introduced.
Uterine Cancer Drugs Market Insights, By Stage of Cancer Treatment
Early-Stage treatments dominate owing to the increasing detection rates through screening and preventive measures, which account for the majority of therapeutic interventions. Advanced-Stage is the fastest growing treatment segment, driven by innovations in immunotherapy and combination regimens allowing better management of metastatic and refractory cancers. Recurrent and Palliative Care hold significant niche roles, optimizing quality of life and extending survival. The Others segment includes emerging clinical approaches and experimental therapies under investigation.
Uterine Cancer Drugs Market Insights, By Route Of Administration
Oral administration dominates the market due to its convenience, patient compliance, and expanding portfolio of effective oral drugs like selective estrogen receptor modulators. Intravenous (IV) administration is the fastest-growing subsegment, propelled by new monoclonal antibodies and immunotherapeutic that require hospital settings for infusion but offer superior efficacy in aggressive disease stages. Subcutaneous delivery is gaining interest as a less invasive alternative for biologics. The Others category comprises inhalational or experimental routes, still in early development phases.
Uterine Cancer Drugs Market Trends
Recent trends indicate increasing integration of personalized medicine approaches, especially with the use of biomarkers to tailor uterine cancer drugs more effectively.
The rise in approvals of novel immunotherapeutic has redefined treatment algorithms, supported by publicly available clinical trial data from 2025.
Furthermore, real-world data incorporation is providing insights into long-term efficacy and safety, influencing market dynamics globally.
Uterine Cancer Drugs Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Uterine Cancer Drugs Market Analysis and Trends
In North America, the dominance in the Uterine Cancer Drugs market is driven by advanced healthcare infrastructure, the presence of major pharmaceutical companies, and an efficient regulatory environment. The U.S. alone contributes roughly 30% to the global market revenue, supported by government initiatives like the Cancer Moonshot program focusing on gynecological cancers and the rapid adoption of immunotherapies.
Asia Pacific Uterine Cancer Drugs Market Analysis and Trends
Meanwhile, the Asia Pacific exhibits the fastest growth with a CAGR of around 10%, fueled by increasing uterine cancer incidence and expanding oncology care infrastructure. Investments from multinational pharmaceutical companies in countries like China and India have boosted local manufacturing and access to innovative treatment options.
Uterine Cancer Drugs Market Outlook for Key Countries
USA Uterine Cancer Drugs Market Analysis and Trends
The USA's market is characterized by robust product pipelines and high healthcare spending. Innovations introduced by companies such as Merck & Co. have led to an expanded availability of immunotherapy drugs, increasing market revenue substantially. Regulatory agencies like the FDA have accelerated approvals, and reimbursement frameworks favor early adoption, supporting sustained market growth.
China Uterine Cancer Drugs Market Analysis and Trends
China's market displays rapid expansion driven by increasing healthcare investments, rising cancer prevalence, and government initiatives encouraging oncology research. Collaborations between domestic manufacturers and global market players have improved drug accessibility. In 2025, China's oncology market grew over 12%, with uterine cancer drugs significantly contributing to this growth.
Analyst Opinion
Precision medicine’s growing role in uterine cancer drug development is a critical driver of market growth. Targeted therapies such as hormone receptor modulators and immune checkpoint inhibitors have expanded therapeutic pipelines, addressing patient subsets with specific genetic profiles. For instance, the approval of pembrolizumab combined with lenvatinib in 2025 increased the treatment options for advanced or metastatic uterine cancer, reflecting a notable 13% increase in patient survival rates reported in recent clinical studies.
Demand-side dynamics underline increasing treatment adoption in emerging markets due to rising incidence rates and improved healthcare infrastructure. In India and China, uterine cancer diagnosis rates have increased by approximately 9%, bolstered by government screening programs and better access to healthcare services, driving imports of novel uterine cancer drugs by 11% year-over-year.
Economic factors such as pricing models and reimbursement policies are shaping market penetration. In the United States, the introduction of value-based pricing for innovative uterine cancer therapies helped reduce patient out-of-pocket expenses by nearly 16%, thereby broadening the patient base and increasing market revenue by an estimated 8% in 2026.
Supply-side indicators, including improvements in manufacturing technologies and streamlined regulatory pathways, have accelerated product launches. For example, advanced biotechnologies reduced production cycle times for monoclonal antibodies used in uterine cancer treatment by 19%, supporting faster market availability and enhancing competitive positioning.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1.90 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.7% | 2033 Value Projection: | USD 3.41 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | AstraZeneca, Pfizer, Merck & Co., Novartis AG, Ipsen, Bristol-Myers Squibb, GlaxoSmithKline, Eli Lilly and Company, Roche Holding AG, Pfizer, Astellas Pharma Inc., and Clovis Oncology. | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Uterine Cancer Drugs Market Growth Factors
Rising incidence of uterine cancer globally continues to propel market growth, with WHO reports indicating a 6% annual rise in new cases over the past five years. Technological advances in bioinformatics and companion diagnostics are enabling more precise patient stratification, which in turn fuels demand for personalized uterine cancer drugs. Favorable regulatory landscapes, especially accelerated approval processes in North America and Europe, have reduced time-to-market for innovative therapies, contributing to increased market revenue. Additionally, growing awareness and adoption of immunotherapy and targeted therapies among oncologists and patients underscore ongoing market expansion despite challenges posed by drug pricing and reimbursement in several emerging countries.
Uterine Cancer Drugs Market Development
In August 2025, the U.S. Food and Drug Administration (FDA) granted expanded approval for GSK’s Jemperli (dostarlimab) in combination with chemotherapy for adults with primary advanced or recurrent endometrial cancer, irrespective of their mismatch repair (MMR) status. This marked a pivotal shift from its earlier indication limited to dMMR/MSI-H tumors. Jemperli, a PD-1 inhibitor, has shown significant progression-free survival benefits in clinical trials.
In June 2025, Merck & Co. received FDA approval for Keytruda (pembrolizumab) in combination with chemotherapy for the treatment of patients with primary advanced or recurrent endometrial carcinoma, covering all MMR subtypes. This broad approval builds on robust data demonstrating improved survival and response rates. At ESMO 2026, Merck presented long-term data showing a durable five-year survival benefit for advanced endometrial cancer patients treated with Keytruda plus Lenvima (lenvatinib) following prior platinum-based chemotherapy.
Key Players
Key market companies include AstraZeneca, Pfizer, Merck & Co., Novartis AG, Ipsen, Bristol-Myers Squibb, GlaxoSmithKline, Eli Lilly and Company, Roche Holding AG, Pfizer, Astellas Pharma Inc., and Clovis Oncology.
Competitive strategies involve exclusive licensing agreements to expand uterine cancer drug portfolios and strategic collaborations focusing on biomarker-driven drug development. For example, a leading global pharmaceutical company formed a strategic partnership with a biotechnology firm in 2026, resulting in the launch of a novel PD-1 inhibitor with a 16% higher efficacy over predecessors, significantly impacting market revenue and share. Market players are also investing in expanding manufacturing capacities and entering emerging markets, which has resulted in a 21% increase in penetration in the Asia Pacific regions during the past two years.
Uterine Cancer Drugs Market Future Outlook
In the future, the uterine cancer drugs market will experience robust growth driven by precision medicine and biomarker-based therapy development. Immuno-oncology agents and combination regimens will continue to redefine treatment standards, improving survival outcomes. The use of companion diagnostics and next-generation sequencing will enable more personalized therapeutic decisions. Additionally, the market will benefit from greater access to cancer care in emerging economies and the launch of biosimilars that reduce treatment costs. Collaborations between pharma companies and diagnostic firms will enhance drug efficacy and accelerate innovation in targeted uterine cancer therapeutics
Uterine Cancer Drugs Market Historical Analysis
The uterine cancer drugs market has undergone a steady transformation with advances in oncology therapeutics. Historically, chemotherapy remained the mainstay of treatment, with platinum- and taxane-based regimens dominating clinical protocols. However, the late 2010s saw a paradigm shift with the introduction of targeted therapies and immune checkpoint inhibitors. Increased understanding of genetic mutations, hormone receptor pathways, and microsatellite instability significantly influenced drug development. Growing investments in oncology R&D and rising incidence rates, especially in developed regions, expanded treatment pipelines and clinical trials.
Sources
Primary Research Interviews:
Oncologists
Gynecologic Cancer Specialists
Clinical Pharmacologists
Medical Oncologists
Pharmaceutical Executives
Databases:
ClinicalTrials.gov
GlobalData Oncology Reports
NCI Cancer Research Data
WHO Cancer Statistics
Magazines:
Oncology Times
Cancer Therapy Advisor
Pharmaceutical Technology
Medscape Oncology
Journals:
Gynecologic Oncology
The Oncologist
Journal of Clinical Oncology
Cancer Research
Newspapers:
The New York Times (Health)
The Guardian (Science)
The Economic Times (Pharma)
The Wall Street Journal (Healthcare)
Associations:
American Society of Clinical Oncology (ASCO)
National Cancer Institute (NCI)
European Society for Medical Oncology (ESMO)
American Cancer Society (ACS)
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients