U.S. Gelatin Sponge Market Size and Forecast – 2026-2033
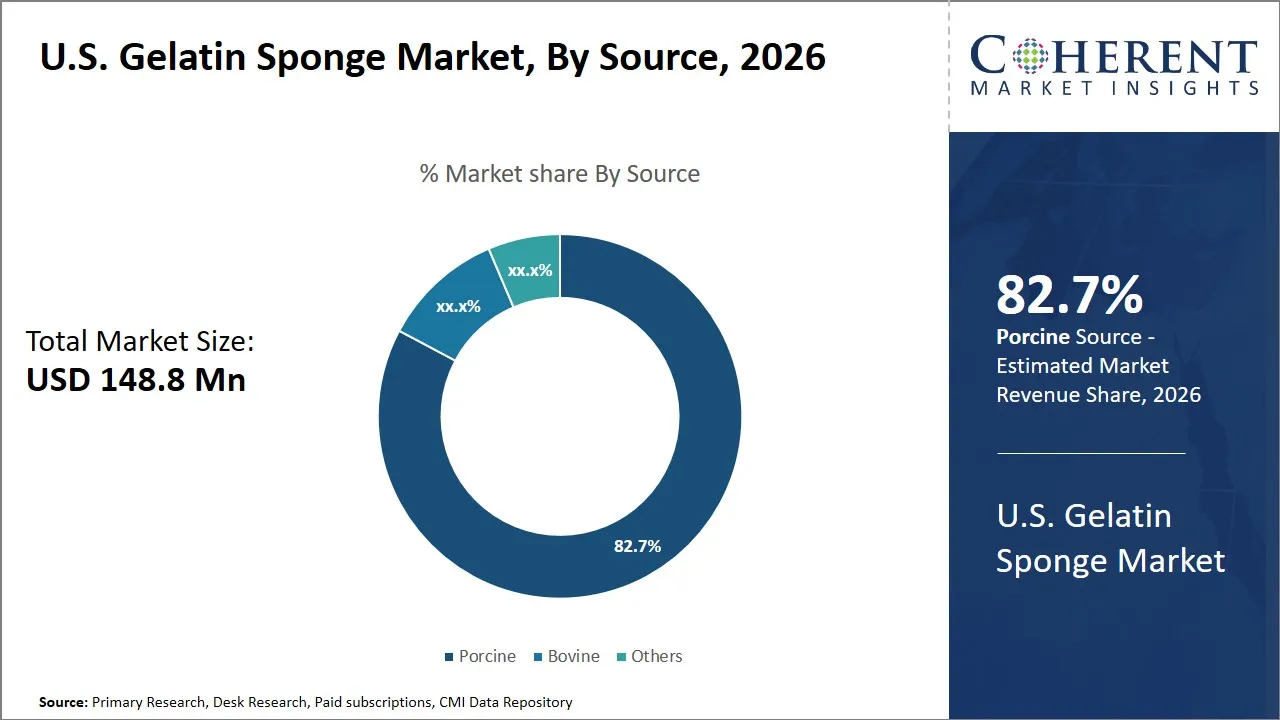
The U.S. gelatin sponge market is estimated to be valued at USD 148.8 Mn in 2026 and is expected to reach USD 220.4 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 8.1% from 2026 to 2033. This growth reflects increasing demand across various medical applications, supported by advancements in surgical procedures and a rising number of hospitalizations, driving the market's expansion over the forecast period.
Key Takeaways of the U.S. Gelatin Sponge Market
- Porcine segment is expected to lead the U.S. gelatin sponge market, capturing 82.7% share in 2026.
- Minimally invasive surgery segment is estimated to represent 58.6% of the U.S. gelatin sponge market share in 2026.
- Hospital segment is projected to dominate with 42.9% of the U.S. gelatin sponge market share in 2026.
Market Overview
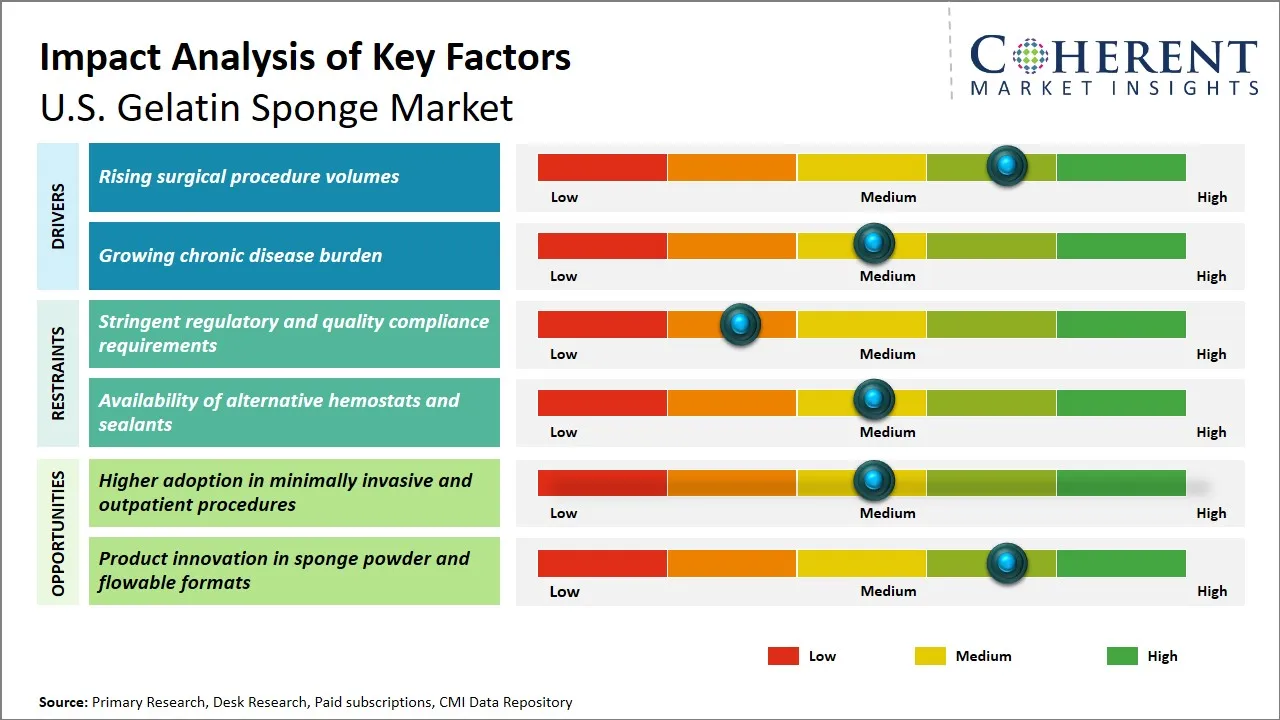
- U.S. gelatin sponge market is driven by the rising volume of surgical procedures across hospitals, ambulatory surgical centers, and specialty clinics in the U.S.
- U.S. gelatin sponge market benefits from the strong clinical preference for absorbable hemostatic materials that help control bleeding quickly and reduce procedural complexity.
- Market growth is supported by increasing demand from orthopedic, dental, ENT, gynecological, and general surgery applications where gelatin sponge remains widely used.
- Market faces competition from advanced hemostats such as flowable matrices, sealants, and thrombin-based products that offer broader performance in complex surgeries.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Rising adoption of advanced surgical hemostatic products |
|
|
Growth in outpatient and ambulatory surgical procedures |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Porcine Segment Dominate the U.S. Gelatin Sponge Market in 2026?
The porcine segment is expected to hold the largest market share of 82.7% in 2026, owing to its superior biological compatibility and availability. Porcine gelatin is also characterized by great biocompatibility, something that reduces immune responses in case it is used as a hemostatic agent in the process of conducting surgery. This is especially important since gelatin sponges are frequently left in place to facilitate clotting and tissue repair.
Furthermore, the porcine sources provide a source of a consistent supply chain in the U.S., which guarantees constant supply to both the manufacturers and the healthcare providers. An established supplier base provides the porcine gelatin with an added advantage in terms of procurement since its availability is not difficult to achieve.
The other prominent reason that makes the porcine segment more prominent is the fact that it has better physicochemical properties than that of bovine or other sources. The porcine gelatin sponges are more uniform in pores and are stronger mechanically, which makes them more effective in blood absorption and facilitating the formation of a clot. Such sponges may also be designed to break down gradually with a desired rate with trends that are in tandem with the regeneration of the tissue, which is essential in the surgical environment to achieve best recovery.
For instance, in March 2024, Aegis Lifesciences Pvt. Ltd. introduced an expanded version of its SURGISPON absorbable gelatin hemostatic sponge, developed from purified porcine gelatin for rapid bleeding control in surgical and dental procedures. The product launch focused on improved absorption capacity and multiple thickness options to support wider surgical applications such as ENT, orthopedic, and trauma procedures.
(Source- Aegis Lifesciences Pvt. Ltd.)
Why Does the Minimally Invasive Surgery Segment Dominate the U.S. Gelatin Sponge Market in 2026?
The minimally invasive surgery segment is expected to hold 58.6% of the market share in 2026, due to the rising demand for less traumatic procedural tools and enhanced patient outcomes. Gelatin sponges are also part of minimally invasive surgery (MIS) because of their application as hemostatic agents which help in the control of bleeding in the restricted anatomical areas, which are reached through small cuts or endoscopic instruments. The accuracy and quick hemostasis of gelatin sponges is ideally suited to the need of MIS procedures, which require effective control of bleeding without massive tissue damage.
The development of MIS has been supported by the growth in medical technology and superior awareness among the patients about the need to experience fewer hospitalizations and quicker recoveries. The requirement of stable easy to use hemostatic agents such as gelatin sponges is urgent because of the limited visualization and access associated with these minimally invasive procedures. The absorbent and conformable properties of gelatin enable it to form well into irregular areas and bond to the surface of tissues to enhance the capacity of the surgeons when dealing with bleeding in delicate and intricate conditions.
For instance, in February 2024, Virtual Incision Corporation announced that the U.S. Food and Drug Administration (FDA) granted marketing authorization for its MIRA Surgical System, the world’s first miniaturized robotic-assisted surgery device designed for minimally invasive procedures such as adult colectomy. The device, approved through the U.S. FDA’s De Novo pathway, allows surgeons to perform complex operations using robotic instruments inserted through a small incision.
(Source- Virtual Incision Corporation)
The Hospital Segment Dominates the U.S. Gelatin Sponge Market
The hospital segment is expected to hold the largest market share of 42.9% in 2026, due to their extensive surgical volumes and complex procedural demands. Diversity of hospital surgery i.e. emergency trauma surgery versus elective complex surgery makes a huge demand on effective hemostatic solutions like gelatin sponges. Hospitals are usually dealing with minimally invasive and general surgeries, and they need multifunctional gelatin sponge products, which could be implemented in various clinical situations. Stress on patient safety and efficiency during surgery is one of the primary reasons why gelatin sponges are implemented in hospitals. Hospitals spend a lot of money on quality surgical supplies that help minimize risks of intraoperative bleeding, complications as well as shortening operation time. Gelatin sponges are very popular since they are easy to apply, absorbent and they can be used with most surgical procedures and hence are invaluable consumables in operating rooms.
Impact of Reimbursement Policies on Surgical Consumables Demand
- The adoption of the products is highly influenced by reimbursement policies of surgical consumables in the U.S. since most items are not billed independently and are rather included as a part of the entire procedure payment within the systems of hospital outpatient and ambulatory surgery provided by Medicare. The hospitals and ASCs consider product such as gelatin sponges not only in terms of clinical value, but also in terms of whether they fit within cost goals under a fixed reimbursement system. CMS completed 2.9 percent revision of 2025 OPPS and ASC payment rates, which contribute to the procedural volumes, yet the bundled payment model continues to place a lot of pressure on routine surgical consumable prices.
- This reimbursement system, however, is more inclined towards consumables, which are easy to use, reliable, and cost effective in daily practices. To manufacturers, it has a rather self-evident consequence: to emerge victorious in this market, a product should not only be performance-wise superior, but also be able to demonstrate its worth in outpatient and hospital settings where procurement teams are increasingly bargaining on supply prices. Practically, reimbursement policy helps achieve predictable demand of vital hemostatic items, though it restricts the flexibility of pricing and reinforced competition with less expensive or more efficient options.
Market Players, Key Developments, and Competitive Intelligence
To learn more about this report, Request Free Sample
Key Developments
- In April 2025, Baxter International Inc. launched a new version of its Hemopatch sealing hemostat, a ready-to-use absorbable patch designed to control surgical bleeding and seal tissues in open and minimally invasive procedures. The launch expands Baxter’s portfolio of absorbable hemostatic products used across neurosurgery, general surgery, and trauma care.
- In November 2024, Johnson & Johnson Services, Inc. completed the acquisition of a European startup specializing in polysaccharide-based hemostat technology to strengthen its biosurgery portfolio and expand its bleeding-control product pipeline. The move supports development of next-generation hemostatic materials used in surgical procedures.
- In December 2021, Advamedica Inc., a biomaterial-focused MedTech startup, announced that the U.S. Food and Drug Administration (FDA) granted 510(k) clearance for its hemostatic product Axiostat Patch, a medical device designed to control moderate to severe bleeding in vascular procedures, surgical debridement sites, puncture sites, and other clinical settings.
Role of U.S. FDA Approvals in Advancing Hemostatic Devices and Materials
- Approval of hemostatic devices and materials by U.S. FDA is one of the primary indicators of the U.S. gelatin sponge market as it proves the safety, effectiveness, and the intended use of the device in surgery before it can be widely adopted by hospitals. Recent U.S. FDA approvals indicate that manufacturers continue to invest in next generation bleeding control products, including the ETHIZIA, approved by the U.S. FDA as PMA on December 15, 2025, and the updates of existing bleeding control products such as the SURGICEL line products offered by Ethicon Corporation and the Avitene product lines offered by Bard in 2025. These approvals are important since they increase the level of performance of all absorbable hemostats applied in surgery.
- This provides support and pressure in the case of the U.S. gelatin sponge market. On the one hand, hemostatic materials cleared and approved by U.S. FDA allow increasing the confidence of surgeons in absorbable products in general and promoting their use in more complicated operations. Competition on conventional gelatin sponge products is more difficult on the other side with tighter standards of approval and more rapid cycles of innovation particularly when newer substances provide greater indications specific data, easier handling, or wider clinical claims.
Top Strategies Followed by U.S. Gelatin Sponge Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Leading companies strengthen their position by investing heavily in research and development to improve gelatin sponge safety, absorption, and surgical usability. They also expand through partnerships with healthcare providers and distributors while widening their reach across high-growth regions with stronger surgical demand. |
Ethicon, a Johnson & Johnson MedTech company, has maintained leadership in absorbable hemostats through products such as SURGIFOAM and by leveraging its broad hospital relationships and surgical portfolio strength. |
|
Mid-Level Players |
Mid-level players in the U.S. gelatin sponge market compete through affordable, reliable products that appeal to cost-sensitive hospitals and surgical centers. They typically focus on manufacturing efficiency, selective partnerships, and broader distribution tie-ups to improve scale, pricing power, and market reach. |
Ferrosan Medical Devices is a strong example, as it focuses specifically on absorbable gelatin hemostats such as SPONGOSTAN and SURGIFOAM, using a specialized product base and partnership-led reach rather than the broad surgical dominance of larger multinationals. |
|
Small-Scale Players |
Small-scale players in the U.S. gelatin sponge market usually compete by focusing on niche applications, differentiated formulations, and flexible manufacturing rather than scale. They rely on specialized features, custom product development, and local distribution alliances to build visibility in targeted surgical and clinical segments. |
CuraMedical is a relevant example. Its CuraSpon absorbable gelatin hemostat is positioned around controlled porosity, rapid blood absorption, and availability in both sponge and powder forms, showing how smaller players use focused product differentiation instead of broad portfolio dominance. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
U.S. Gelatin Sponge Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 148.8 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.1% | 2033 Value Projection: | USD 220.4 Mn |
| Segments covered: |
|
||
| Companies covered: |
Johnson & Johnson, Pfizer Inc., Baxter International Inc., B. Braun Melsungen AG, CuraMedical B.V., GELITA MEDICAL, Curasan AG, Meril Life Sciences Pvt. Ltd., Aegis Lifesciences Pvt. Ltd., Beijing Datsing Bio-Tech Co., Ltd., Beijing Taikesiman Pharmaceutical Co., Ltd., Foryou Medical Devices Co., Ltd., Guizhou Jin Jiu Biotech Co., Ltd., Saikesaisi Holdings Group Co., Ltd., and Biotemed Co., Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
U.S. Gelatin Sponge Market Dynamics
To learn more about this report, Request Free Sample
U.S. Gelatin Sponge Market Driver - Rising Surgical Procedure Volumes Boost Demand for Gelatin Sponge
The rising cases of surgical operation carried out in the U.S. are a major factor to the demand of gelatin sponges because the products are extensively exploited as hemostatic agents in surgeries. As more sophisticated medical technology is applied, and the population is growing older, hospitals and surgical centers are experiencing an apparent steady increase of both elective and emergency surgeries. Gelatin sponges offer an effective and biocompatible method of bleeding control, encouraging clot generation, and healing wounds in most types of surgery, including orthopedic, cardiovascular, and dental surgery. Besides, the increasing use of minimally invasive surgery methods also boosts the desire to have quality hemostatic devices that reduce tissue traumas and shorten recovery time. With the widespread adoption of gelatin sponges, moves by medical professionals to implement them in the surgical procedures and the growing emphasis on patient safety and improved surgical success, they are increasingly becoming part of the standard procedure in health care.
For instance, in June 2024, AMSURG and Palomar Health announced a strategic surgery center partnership for a new facility with five operating rooms covering multiple specialties including orthopedics and ENT. Expansion of multi-specialty surgical capacity like this directly increases demand for absorbable hemostatic products such as gelatin sponges used during routine procedures.
(Sources- AMSURG)
U.S. Gelatin Sponge Market Opportunity - Higher Adoption in Minimally Invasive and Outpatient Procedures
The gelatin sponge market in the U.S. is ripe with a lot of growth due to a growing uptake of minimally invasive and outpatient procedure. Increases in surgical procedures have also resulted in more patients seeking fewer invasive surgeries, which not only decrease patient recovery time, risks of infections, and decreases the length of stay of the patient. Gelatin sponges are common hemostatic agents that are being used to manage bleeding during surgical procedures and can easily fit in this dynamic world because it is biocompatible and absorbent and easy to use. The outpatient surgeries are on the up surge due to the healthcare cost containment efforts and the preference of patients to be discharged sooner hence offers a vast application potential of the gelatin sponges. These materials also ensure that bleeding is controlled effectively in ambulatory surgical centers where timeliness of the business and promptness of patient turnover are vital.
For instance, in December 2025, Medtronic received U.S. FDA clearance for the Hugo robotic-assisted surgery system for minimally invasive urologic procedures. This supports the shift toward less invasive and outpatient surgeries, which is increasing demand for absorbable hemostatic products such as gelatin sponges used in confined surgical settings.
(Sources- Medtronic)
Analyst Opinion (Expert Opinion)
- The U.S. gelatin sponge market is growing on the back of steady surgical volumes, especially in general surgery, orthopedics, dental, and ENT procedures, where absorbable hemostats remain a practical choice. The market is also benefiting from product upgrades that improve handling and storage, as seen in Baxter, on April 10, 2025 launch of its room temperature HEMOPATCH. At the same time, competition is getting tougher as hospitals compare traditional gelatin sponge products with newer hemostatic formats that offer broader performance in complex procedures. Quality remains a hard market issue, and the U.S. FDA, on May 22, 2024 recall notice for Ethicon’s SURGIFOAM powder kit is a clear reminder that reliability and compliance directly affect purchasing confidence.
- Industry events are also helping shape the market by accelerating product visibility and clinical adoption. The ACS Clinical Congress 2024 highlighted surgical innovation through a large exhibit hall and Innovation Theater, while the AORN Global Surgical Conference & Expo 2025 continued to serve as a major platform for perioperative education and technology showcase. These events matter because they influence surgeon awareness, hospital evaluation, and adoption of newer bleeding control solutions across the market.
Market Segmentation
- Source Insights (Revenue, USD Mn, 2021 - 2033)
- Porcine
- Bovine
- Others
- Application Insights (Revenue, USD Mn, 2021 - 2033)
- Minimally Invasive Surgery
- General Surgery
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Hospital
- Clinics
- Ambulatory Surgical Centers
- Others
- Sales Channel Insights (Revenue, USD Mn, 2021 - 2033)
- Offline
- Online
- Key Players Insights
- Johnson & Johnson
- Pfizer Inc.
- Baxter International Inc.
- Braun Melsungen AG
- CuraMedical B.V.
- GELITA MEDICAL
- Curasan AG
- Meril Life Sciences Pvt. Ltd.
- Aegis Lifesciences Pvt. Ltd.
- Beijing Datsing Bio-Tech Co., Ltd.
- Beijing Taikesiman Pharmaceutical Co., Ltd.
- Foryou Medical Devices Co., Ltd.
- Guizhou Jin Jiu Biotech Co., Ltd.
- Saikesaisi Holdings Group Co., Ltd.
- Biotemed Co., Ltd.
Sources
Primary Research Interviews
Industry Stakeholders list
- Hospital procurement managers
- Operating room directors
- General surgeons
- Orthopedic surgeons
- ENT specialists
- Ambulatory surgery center administrators
End-users list
- Hospital pharmacists
- Perioperative nurses
- Dental surgeons
- Gynecologic surgeons
- Trauma care specialists
- Sterile processing department managers
Government and International Databases
- U.S. Food and Drug Administration
- Centers for Medicare & Medicaid Services
- National Center for Health Statistics
- Agency for Healthcare Research and Quality
- National Institutes of Health
- World Health Organization
Trade Publications
- MedTech Dive
- MPO Magazine
- Outpatient Surgery Magazine
- OR Manager
- Becker’s ASC Review
- Medical Device and Diagnostic Industry
Academic Journals
- Journal of Surgical Research
- Annals of Surgery
- JAMA Surgery
- The American Journal of Surgery
- Surgery
- BMC Surgery
Reputable Newspapers
- The Wall Street Journal
- The New York Times
- Financial Times
- The Washington Post
- USA Today
- Los Angeles Times
Industry Associations
- American College of Surgeons
- Association of periOperative Registered Nurses
- American Hospital Association
- Ambulatory Surgery Center Association
- American Academy of Otolaryngology Head and Neck Surgery
- American Dental Association
Public Domain Resources
- ClinicalTrials.gov
- MedlinePlus
- Federal Register
- U.S. Census Bureau
- Centers for Disease Control and Prevention
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 8 Years
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients