U.S. Medical Metal Tubing Market Size and Forecast – 2026 To 2033
The U.S. medical metal tubing market is expected to grow from USD 434.2 Mn in 2026 to USD 804.5 Mn by 2033, registering a compound annual growth rate (CAGR) of 9.2%. The U.S. medical metal tubing market is poised for significant expansion, fueled by rising volume of surgical procedures across the country.
According to Centers for Disease Control and Prevention (CDC), the nation registers more than 51 million inpatient treatments each year. Furthermore, National Library of Medicine states that over 50 million inpatient surgical procedures are performed in the U.S. every year.
(Source: Centers for Disease Control and Prevention, National Library of Medicine)
Key Takeaways of the U.S. Medical Metal Tubing Market
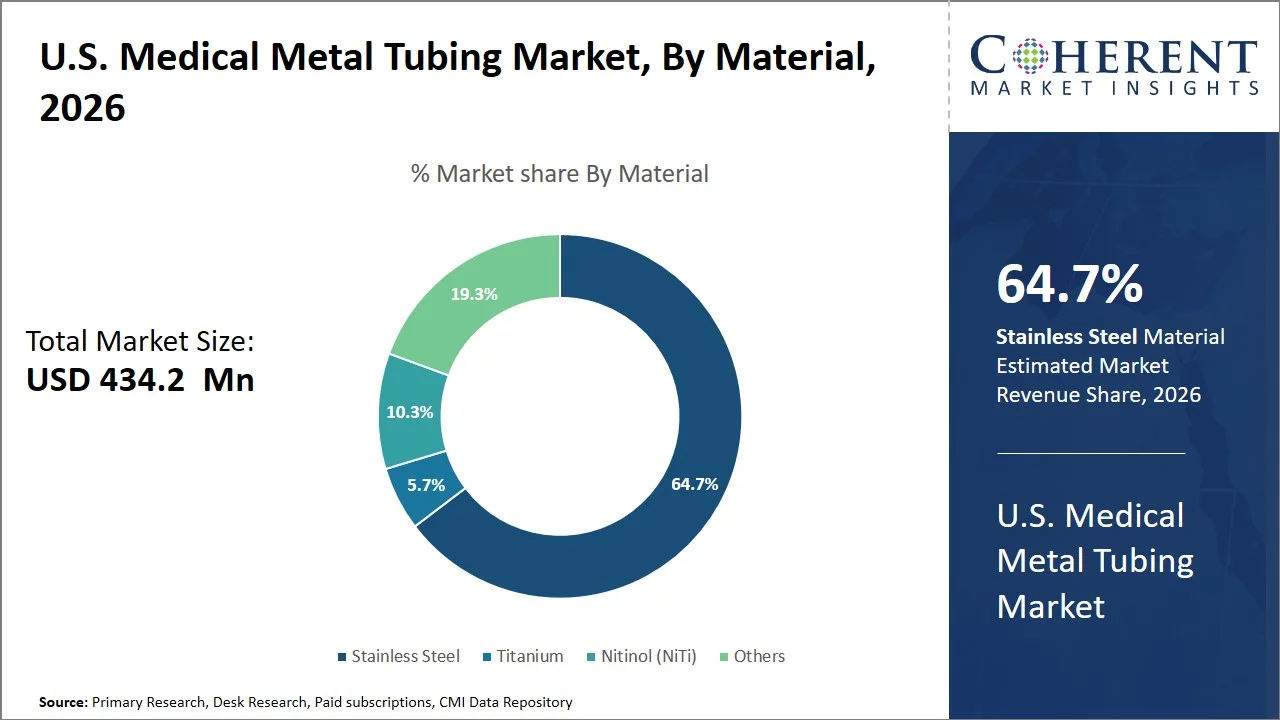
- Stainless steel is projected to hold 64.7% of the U.S. medical metal tubing market share in 2026, making it the dominant material segment, attributed to its indispensable contribution to the manufacturing process of hypotubes that find application in minimally invasive devices like guidewires, stents, and catheter tubes. Stainless steel finds wide application in the manufacturing of thin wall precision tubes used in cardiovascular and neurovascular procedures in the U.S., where precision in dimensions and torque transmission is essential. For instance, U.S. Food and Drug Administration summary of 510(k) for the Suspension Clavicle Fracture Fixation System emphasizes the importance of stainless steel in orthopedic fixation devices, demonstrating its well-documented history of being used as an implantable medical component that bears loads and illustrating its prevalence in the production of medical-grade tubing and device manufacture. (Source: U.S. Food and Drug Administration)
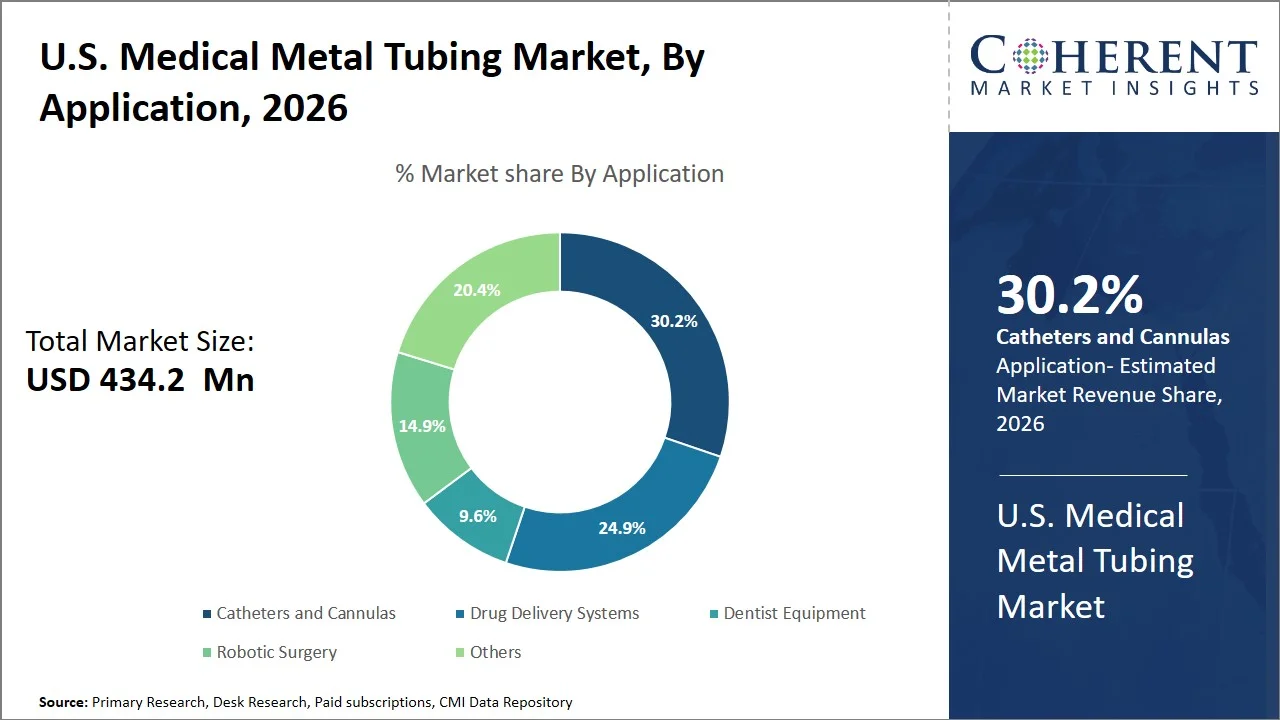
- Catheters and cannulas are projected to hold 30.2% of the U.S. medical metal tubing market share in 2026, making it the dominant application segment, owing to the large number of procedures that are carried out using catheters, including angioplasty, electrophysiology, and central venous access. In the U.S. healthcare market, there is a high adoption of interventional cardiology and imaging procedures, which require reinforced catheter tubing for ease of movement and navigation, thus stimulating demand for the catheter tubing segment. For instance, nearly 900,000 percutaneous coronary interventions (PCIs) are performed annually in the U.S., with each procedure relying on multiple catheter-based devices for vascular access and treatment delivery, directly driving demand for metal-reinforced catheter tubing. (Source: Yale Medicine)
- Hospitals are projected to hold 35.0% of the U.S. medical metal tubing market share in 2026, making it the dominant end user segment, owing to their crucial function in carrying out highly intricate procedures for both inpatients and outpatients, which involve the use of medical devices made from metal tubing. In the U.S. hospitals play a major role in performing catheterization lab procedures, surgeries, and emergency vascular access procedures, which rely on high-quality metal tubes, thus generating the highest concentration of demand at the hospital level. For instance, according to the National Committee for Quality Assurance, about 33.7 million hospital admissions occur every year in the U.S., which signifies the large number of patients passing through hospitals where surgical and intervention operations take place. This high number of inpatients makes hospitals the most common users of medical equipment with metal tubing, such as catheters and cannulas. (Source: National Committee for Quality Assurance)
- Increasing Use of High-Performance Alloys in Complex Interventions: The increasing intricacy of medical procedures, such as cardiac and neurovascular procedures, is leading to a greater need for innovative materials for metal tubes, such as nitinol and stainless steel. Such materials have great flexibility and elasticity properties; hence, they will be very suitable for navigating complex instruments.
- Customization and Miniaturization of Medical Devices: Due to the growing trend toward the use of small and highly sophisticated medical equipment, the requirement for metal tubes that are manufactured as per precise specifications has grown stronger in the U.S. medical metal tubing market. Medical device manufacturers are more inclined towards metal tubes that are produced as per exact specifications and are extremely thin.
Why Does the Stainless Steel Segment Dominate the U.S. Medical Metal Tubing Market?
Stainless steel is projected to hold a market share of 64.7% in 2026, attributed to its excellent characteristics in precision engineering, bearing loads, and blood compatibility in medicine. It facilitates the manufacture of very thin tubing with fatigue strength and corrosion resistance, which is essential for devices that operate under constant mechanical strain. Additionally, its resilience in physiological conditions makes it ideal for cardiovascular and minimally invasive applications. For instance, a peer-reviewed study indexed in the National Library of Medicine show that passivated 316L stainless steel stents used in heart surgeries have a stable oxide film and excellent resistance to corrosion in biological environments, making it highly desirable for use in critical parts of biomedical equipment such as stent tubes. (Source: National Library of Medicine)
Why are Catheters and Cannulas the Most Preferred Application in the U.S. Medical Metal Tubing Market?
To learn more about this report, Request Free Sample
Catheters and cannulas are projected to hold 30.2% of the market share in 2026, attributed to its significant role in several highly intensive and frequent surgical procedures that require accurate guidance, structural reinforcement, and efficient access to blood vessels. While the rest of the applications can function without metal tubing, catheters and cannulas cannot operate without reinforced tubing (hypotubes, braided shafts). This product allows for greater flexibility in navigating narrow anatomical spaces and maintaining stability. High utilization rates for image-guided and minimally invasive procedures are observed in the healthcare system of the country. In turn, catheter access represents the common procedure utilized in various surgeries, ensuring constant demand for metal tubing. For instance, research indexed in the National Library of Medicine indicate that the use of central venous catheters are common practice within intensive care units for various purposes including administration of medications and hemodynamic monitoring, thus making it a frequent occurrence. (Source: National Library of Medicine)
Hospitals Segment Dominates the U.S. Medical Metal Tubing Market
The hospitals segment is expected to hold 35.0% of the U.S. medical metal tubing market share in 2026, attributed to their central role in conducting numerous interventional surgeries involving metal tubing such as catheters, cannulas, and minimally invasive surgical instruments. There are special departments in U.S. hospitals that perform procedures requiring metal tubing; these include the catheterization lab, operating rooms, and ICU departments.
According to the U.S. Food and Drug Administration (FDA), medical devices are categorized into Classes I, II, and III. In the case of Class III medical devices, they require Premarket Approval (PMA), which is the most demanding process. (Source: U.S. Food and Drug Administration) This requirement ensures that hospitals will continue to be the key venues where medical devices will be implanted and evaluated, since only hospitals have all the resources needed to ensure regulatory compliance.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
U.S. FDA Quality Management System Regulation (QMSR) Alignment with ISO 13485 (2026) |
|
|
U.S. FDA Unique Device Identification (UDI) System Expansion & Enforcement (2022-Ongoing) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: U.S. Food and Drug Administration, U.S. Food and Drug Administration)
U.S. Medical Metal Tubing Market Dynamics
To learn more about this report, Request Free Sample
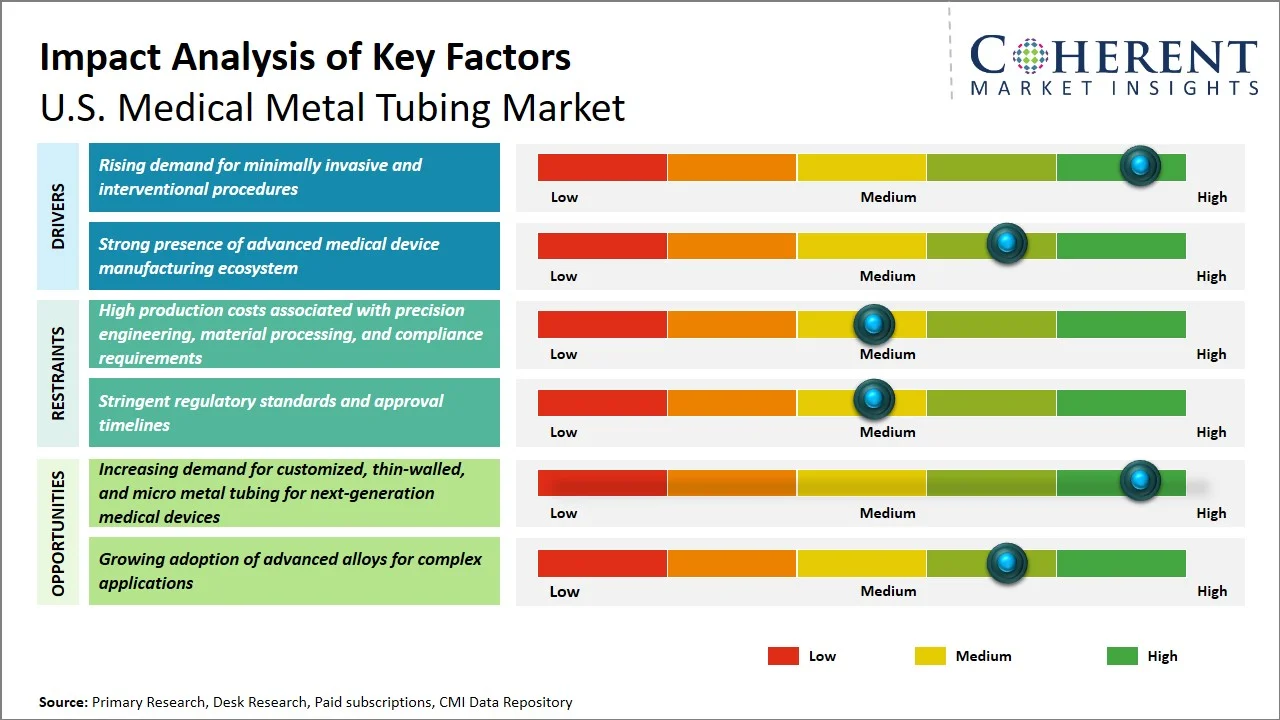
Market Drivers
- Rising demand for minimally invasive and interventional procedures: There is currently a considerable trend towards minimally invasive catheter-based treatment in the U.S. healthcare sector based on the advantages associated with low injury risk, shortened length of stay at health care facilities, and improved speed of recovery, and the increased use of devices that feature medical metal tubing. This technique is highly dependent on the use of precision hypotubes, braided shafts, and catheters reinforced with metal to allow precise guidance and placement of the devices within the complicated anatomic channels. For instance, research published in National Library of Medicine indicated that there has been a penetration of minimally invasive surgery (MIS) techniques to over 60.5% of cases among some common procedures in U.S. hospitals, while hospitals that employ state-of-the-art technologies have reached even higher numbers – about 65.8%. (Source: National Library of Medicine)
- Strong presence of advanced medical device manufacturing ecosystem: The U.S. has a highly advanced and innovation-driven medical device manufacturing ecosystem that significantly determines the need for accurate medical metal tubes in catheters, implants, and minimally invasive devices. An ecosystem that is teeming with leading OEMs, contract manufacturers, and medical device components manufacturers leads to frequent innovations and production of highly complex devices with demanding requirements like thin-walled tubing tolerances. In addition, the presence of a thriving regulatory pipeline serves to further fuel the frequency of innovations and product launches. For instance, U.S. Food and Drug Administration, states that over 3,000-3,300 medical devices are approved annually under the U.S. FDA's 510(k) process due to innovations made in catheterization and interventional devices. (Source: Intuition Labs)
Emerging Trends
- Integration of Smart and Sensor-Enabled Tubing Components: The trend towards incorporating sensing functionality into medical tubing that facilitates real-time procedure monitoring has gained momentum. Such smart features can be used for the purposes of pressure, temperature, and flow rate measurement. In critical care settings, where advanced surgery may take place, such sensors become highly valuable. The use of connected health systems is becoming more popular, and this has encouraged tubing manufacturers to incorporate digital health capabilities into their products.
- Increasing Focus on Sustainable and Recyclable Material Processing: Sustainability has become one of the most important factors in the manufacturing industry in terms of medical products, and metal tube production is not an exception. Manufacturers have started to incorporate greener production methods, minimize material wastage, and increase the recyclable rate of metal parts. In addition, there has been increasing attention towards the optimization of energy utilization in production processes such as extrusion and laser cutting.
Regulatory & Reimbursement Landscape Shaping the U.S. Medical Metal Tubing Market
|
Category |
Policy/Framework |
Description |
Impact on Market |
|
Quality & Manufacturing Compliance |
U.S. Food and Drug Administration – Quality Management System Regulation (QMSR) |
Finalized in 2024 (effective 2026), aligning U.S. device manufacturing with ISO 13485:2016, replacing 21 CFR Part 820. |
Increases requirements for process validation, supplier control, and traceability for precision tubing, favoring manufacturers with advanced quality systems and certified production capabilities. |
|
Device Traceability & Identification |
U.S. Food and Drug Administration – Unique Device Identification (UDI) System |
Expanded enforcement across Class II & III devices through 2024–2025, requiring standardized device identification linked to the GUDID database |
Drives component-level traceability, pushing tubing suppliers to adopt batch tracking, serialization, and digital documentation systems. |
|
Premarket Approval & Clearance Pathways |
U.S. Food and Drug Administration – PMA & 510(k) Framework |
Ongoing regulatory pathways requiring clinical validation (PMA) and substantial equivalence (510(k)) for device approvals. |
Sustains demand for high-specification, compliant tubing materials used in regulated devices such as catheters, stents, and implants. |
|
Procedure Reimbursement Framework |
Centers for Medicare & Medicaid Services – Medicare Coverage for Interventional Procedures |
CMS provides reimbursement for catheter-based and minimally invasive procedures, particularly in cardiovascular and oncology care. |
Encourages adoption of interventional procedures, directly increasing demand for metal tubing-based devices such as catheters and cannulas. |
|
Hospital Payment Systems |
Centers for Medicare & Medicaid Services – IPPS & OPPS Updates |
Annual updates (2024–2025) defining reimbursement rates for inpatient and outpatient procedures |
Supports sustained procedure volumes in hospitals and ambulatory centers, reinforcing continuous demand for tubing-intensive medical devices. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is the growing adoption of advanced alloys for complex applications creating new growth opportunities in the U.S. medical metal tubing market?
There have been increased developments in the use of cutting-edge alloys like Nitinol (nickel-titanium), cobalt-chromium, and titanium-based alloys that create new growth avenues in the medical metal tubing market. The unique properties of the advanced alloys like super elasticity, shape-memory characteristics, excellent fatigue-resistance, and better biocompatibility allow developing innovative next generation devices, which cannot be developed using conventional stainless-steel tubing.
According to recent researches, the application of Nitinol in vascular stents and guidewires is well recognized because of the capability to deform and regain its original shape. As a result, Nitinol can be successfully used in catheter-based procedures and helps in minimizing procedure-related complications up to 25% in comparison to conventional materials. (Source: Stanford Advanced Materials) Moreover, the usage of cobalt-chromium alloys has been popular in load-bearing orthopedic and cardiovascular implants because of better strength and wear-resistant properties, and the application of titanium alloys has increased considerably in long-term implants due to good fatigue-resistant characteristics and osseointegration properties. (Source: Runsom Precision Co., Ltd)
Market Players, Key Development, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- On April 23, 2026, Reflow Medical unveiled Cora Flex and Cora Force torque microcatheters specifically intended for use during complicated procedures in the coronary arteries with improved torque transfer capabilities. This product launch is indicative of the increasing trend in developments of catheter-based technologies, alongside rising interest in specialized metal tubing parts such as hypotubes.
- In November 2025, Freudenberg Medical acquired Fuji Seiko, adding to its abilities in the precision metal tubing and hypotube production for use in minimally invasive equipment. The move improves Freudenberg’s global logistics and metallurgical know-how, especially with regard to catheter-related products. It is indicative of an increasing trend toward vertical integration within the medical metal tubing market to ensure control of key components.
Competitive Landscape
The U.S. medical metal tubing market is extremely competitive due to specialist manufacturing organizations and Contract Development and Manufacturing Organizations (CDMOs) like TE Connectivity, Freudenberg Medical, Integer Holdings Corporation, and Nordson Corporation. Competition is based on the ability to manufacture products that have been made using sophisticated manufacturing methods as well as material knowledge and stringent adherence to regulations. Increasing attention is being paid to miniaturization, making of thin-walled hypotubes, and incorporating alloys like nitinol to develop intricate interventional devices. Key focus areas include:
- Development of ultra-thin wall, high-precision hypotubes for minimally invasive and interventional devices
- Adoption of advanced alloys such as nitinol and cobalt-chromium for enhanced flexibility and fatigue resistance
- Expansion of vertically integrated manufacturing to strengthen supply chain control and reduce lead times
- Integration of laser cutting, micro-machining, and surface finishing technologies for complex tubing geometries
- Emphasis on regulatory compliance, traceability, and quality assurance in medical-grade tubing production
Market Report Scope
U.S. Medical Metal Tubing Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 434.2 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.2% | 2033 Value Projection: | USD 804.5 Mn |
| Segments covered: |
|
||
| Companies covered: |
Saint-Gobain, Freudenberg Medical, TE Connectivity, W. L. Gore & Associates, Nordson Medical, Teleflex Incorporated, Zeus Industrial Products, American Tube Technology, Vita Needle Company, and Eagle Stainless Tube & Fabrication |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- The future of the U.S. medical metal tubing market lies in the transition of the industry towards highly sophisticated and tailored medical tubing as the interventions become increasingly sophisticated and the needs associated with medical devices grow. The trends in growth are expected to come from innovations in materials, together with advancements in manufacturing methods, with an increased focus on the use of high-end materials, such as nitinol and cobalt-chromium alloy, as well as on the development of tubing that incorporates properties like flexibility, high strength, and resistance to fatigue in one single tube design. In tandem, the industry will undergo a transition from traditional manufacturing to digitally enabled ecosystems of manufacturing, spurred on by the alignment of regulations by the U.S. Food and Drug Administration.
- Market opportunities will largely stem from applications within hypotubes found in catheters and microcatheters in the U.S. due to the increasing demand for sophisticated interventional procedures requiring superior performance and durability in bendable hypotubes. These opportunities will be mostly driven by the need for better manoeuvrability of catheters in complex anatomy, particularly microcatheters and guidewires, where there is a tendency towards replacement of conventional stainless steel with more efficient super elastic alloys and composite layers.
- In order to create an advantage for themselves, the competitors need to progress from mere manufacturing to building end-to-end capabilities such as material science knowledge, in-house processing of the hypotubes through laser cutting, grinding, and electropolishing, as well as vertical integration. Early collaboration with original equipment manufacturers (OEMs) and contract development and manufacturing organizations (CDMOs) like Freudenberg Medical and Integer Holdings Corporation in developing tubing products tailored to applications will be key. Another strategic consideration is adopting digital quality systems along with the latest monitoring systems and strict compliance frameworks in light of U.S. FDA regulations to speed up the market entry process.
Market Segmentation
- Material Insights (Revenue, USD Mn, 2021 - 2033)
- Stainless Steel
- Titanium
- Nitinol (NiTi)
- Others
- Application Insights (Revenue, USD Mn, 2021 - 2033)
- Catheters and Cannulas
- Drug Delivery Systems
- Dentist Equipment
- Robotic Surgery
- Others
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Hospitals
- Clinics
- Ambulatory Surgical Centers
- Others
- Key Players Insights
- Saint-Gobain
- Freudenberg Medical
- TE Connectivity
- L. Gore & Associates
- Nordson Medical
- Teleflex Incorporated
- Zeus Industrial Products
- American Tube Technology
- Vita Needle Company
- Eagle Stainless Tube & Fabrication
Sources
Primary Research Interviews
- Chief of Cardiology / Interventional Cardiologists – catheter-based procedure trends (U.S. hospitals)
- Directors – Cardiac Catheterization Labs / Interventional Radiology Units
- Clinical Specialists – catheter systems, guidewires, and implantable device manufacturers
- Ambulatory Surgical Center (ASC) Administrators – outpatient interventional procedure adoption
- Biomedical Engineers – tubing performance, material selection, and device integration
Stakeholders
- Medical Metal Tubing Manufacturers & CDMOs
- End-use Sectors
- Hospitals (tertiary care & interventional centers)
- Ambulatory Surgical Centers (ASCs)
- Specialty cardiovascular & interventional clinics
- Regulatory & Health Bodies: U.S. Food and Drug Administration – approvals, safety monitoring, and post-market surveillance; Centers for Medicare & Medicaid Services – reimbursement and coverage frameworks
Databases
- Centers for Medicare & Medicaid Services – procedure volumes, reimbursement, and coverage decisions
- Healthcare Cost and Utilization Project – hospitalization and procedure-level data
- National Center for Health Statistics – disease prevalence and mortality trends
- ClinicalTrials.gov – ongoing trials involving catheter-based and implantable devices
- U.S. Food and Drug Administration MAUDE Database – adverse event and device performance reports used for post-market surveillance
Magazines
- Medical Design & Outsourcing – trends in medical device engineering, precision tubing, and catheter technologies
- MD+DI (Medical Device and Diagnostic Industry) – innovations in biomaterials, manufacturing processes, and regulatory updates
- MedTech Dive – industry developments, product launches, and strategic movements in medical devices
- DeviceTalks – expert insights on interventional devices, minimally invasive technologies, and OEM–CDMO collaboration trends
Journals
- Journal of Biomedical Materials Research – material performance (stainless steel, nitinol, cobalt-chromium) in medical applications
- Acta Biomaterialia – advanced alloy behavior, fatigue resistance, and biocompatibility
- Journal of Medical Devices – device engineering, microfabrication, and tubing applications
- Cardiovascular Engineering and Technology – catheter-based device innovation and interventional technologies
Newspapers
- The Wall Street Journal – Covers medtech investments, M&A activity, and healthcare manufacturing trends
- The New York Times – Reports on healthcare advancements, medical devices, and regulatory developments
- USA Today – General healthcare coverage including medical innovations and patient trends
- Los Angeles Times – Covers biotech, medical device innovation, and regional manufacturing activity
- The Washington Post – Focuses on healthcare policy, FDA regulations, and public health developments
Associations
- American College of Cardiology
- Heart Rhythm Society
- American Heart Association
- Medical Device Innovation Consortium
Public Domain Sources
- U.S. Food and Drug Administration – regulatory pathways, approvals, and post-market surveillance databases such as MAUDE (adverse event reporting system)
- Centers for Medicare & Medicaid Services – reimbursement frameworks and procedure coverage policies
- Centers for Disease Control and Prevention – disease burden and procedural trends influencing device demand
- National Institutes of Health – funding and innovation in biomaterials and interventional devices
- Agency for Healthcare Research and Quality – patient safety, outcomes, and device utilization insights
Proprietary Elements
- CMI Data Analytics Tool, Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients