Global Veterinary Vaccine Adjuvants Market Size and Forecast: 2026-2033
The global veterinary vaccine adjuvants market is expected to grow from USD 410 Mn in 2026 to USD 640 Mn by 2033, registering a compound annual growth rate (CAGR) of 6.6%. The global veterinary vaccine adjuvants market is poised for significant expansion, fueled by the rising prevalence of zoonotic diseases.
According to the Centers for Disease Control and Prevention, zoonotic diseases are very common, both in the U.S. and around the world, with more than 6 out of every 10 known infectious diseases in people can be spread from animals, and 3 out of every 4 new or emerging infectious diseases in people come from animals. (Source: Centers for Disease Control and Prevention)
Additionally, according to the World Health Organization, over 30 new human pathogens have been detected in the last three decades, 75% of which have originated in animals. Furthermore, in last two decades, emerging zoonotic diseases have been reported from 18 out of 22 countries in the region, often, with explosive outbreaks and high fatalities never seen in any other WHO region. (Source: World Health Organization)
Key Takeaways of the Global Veterinary Vaccine Adjuvants Market
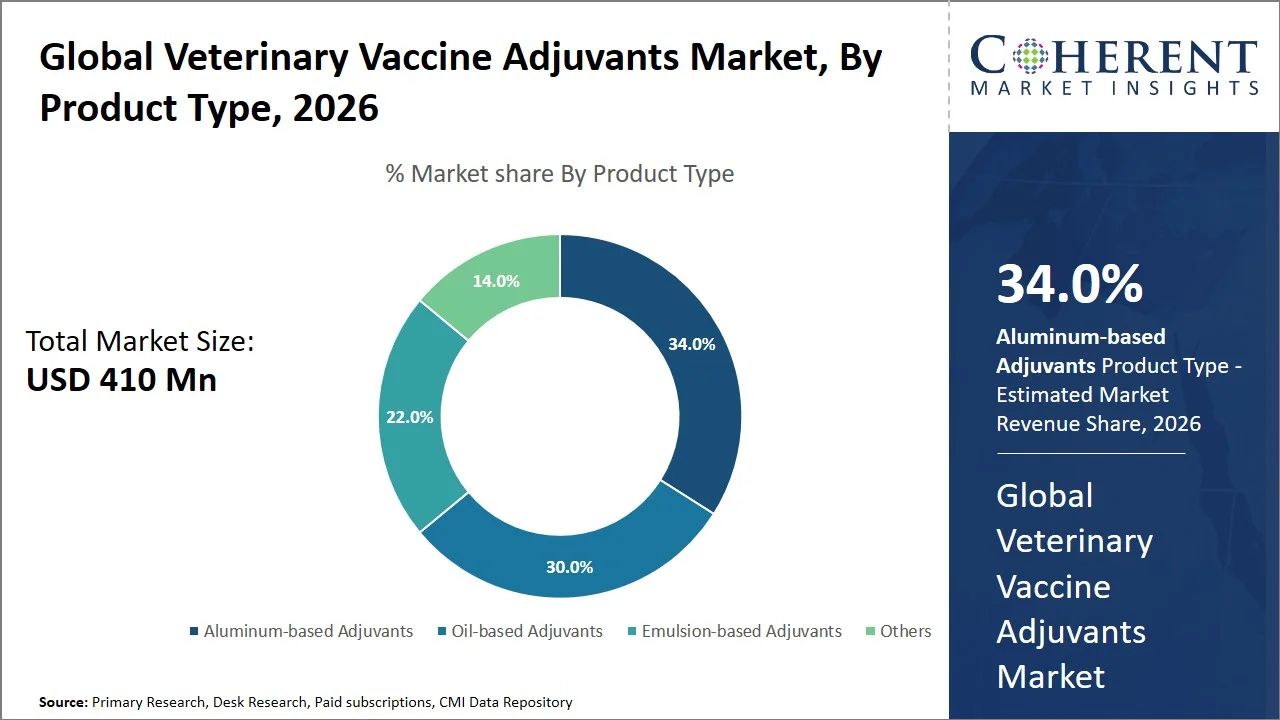
- Aluminum-based adjuvants are projected to hold 34.0% of the global veterinary vaccine adjuvants market share in 2026, making it the dominant product type segment, with North America leading due to vigorous regulatory frameworks and standardized vaccine approval systems. For instance, the U.S. has stringent requirements for veterinary biologics (including purity, safety, potency, efficacy, and serial release testing of veterinary biologics) that are under the jurisdiction of U.S. Department of Agriculture (USDA) Animal and Plant Health Inspection Service (APHIS), thereby ensuring high safety standards, quality, and efficacy, and hence allowing the utilization of proven adjuvants such as aluminum salts. (Source: U.S. Department of Agriculture)
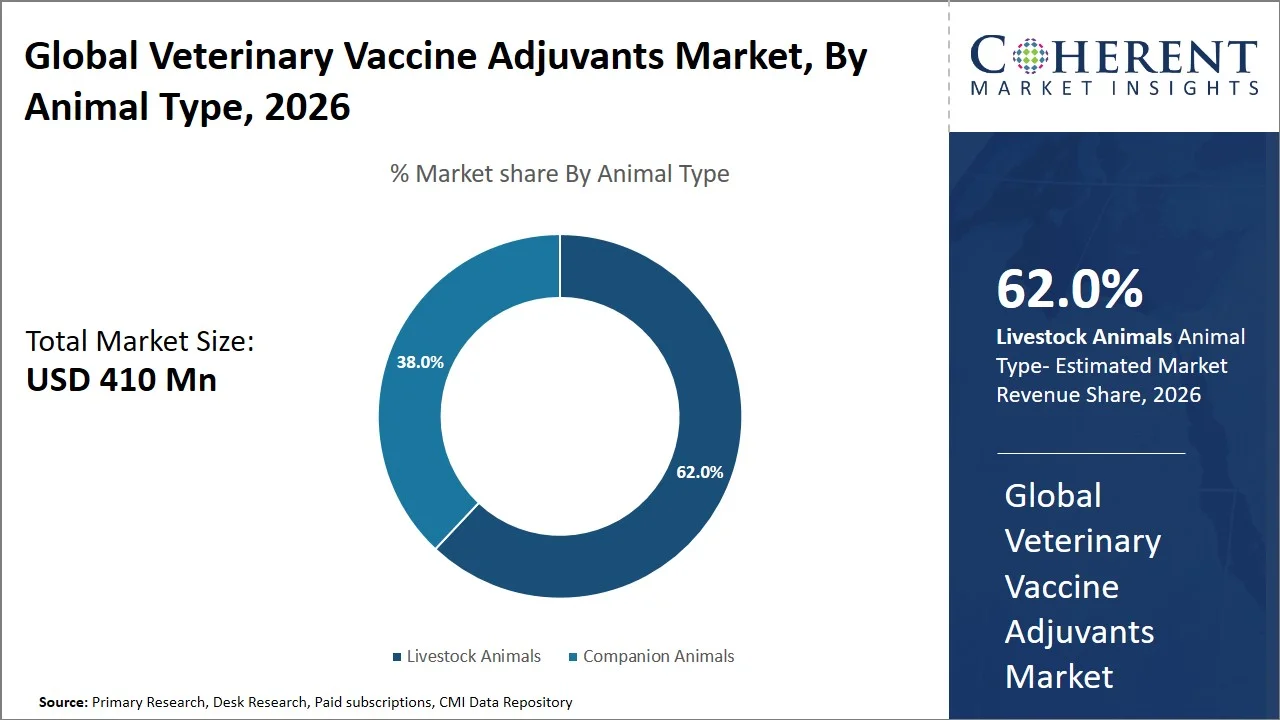
- Livestock animals are projected to hold 62.0% of the global veterinary vaccine adjuvants market share in 2026, making it the dominant animal type segment, with dominance especially in Asia Pacific attributed to the large-scale vaccination programs conducted by governments. For instance, the Department of Animal Husbandry and Dairying in India has started the National Animal Disease Control Programme (NADCP) that aims at vaccinating 100% cattle and buffaloes against Foot-and-Mouth Disease (FMD), significantly driving demand for adjuvanted vaccines. (Source: Department of Animal Husbandry and Dairying)
- Injectable is projected to hold 72.0% of the global veterinary vaccine adjuvants market share in 2026, making it the dominant route of administration segment, with wide acceptance in Europe and Asia Pacific owing to its enhanced immunological efficacy and accuracy of dosage. Injectable vaccines guarantee control over antigen administration and a better immune reaction, thus being the best option in organized vaccination programs. For instance, Vaccine protocols in the EU fall under the purview of the European Medicines Agency through the Veterinary Medicinal Products Regulation (EU) 2019/6, focusing on the need for validated methods of delivery, standardized dosage, and proof of efficacy, with injectable vaccines preferred for most licensed veterinary biologics (Source: European Medicines Agency). Additionally, extensive livestock immunization efforts, conducted in countries such as India, involve uniform injectable protocols that are aided by cold chain infrastructure and traceability mechanisms to ensure efficient immunity and effective coverage among animal communities through national campaigns organized by the Department of Animal Husbandry and Dairying. (Source: Department of Animal Husbandry and Dairying)
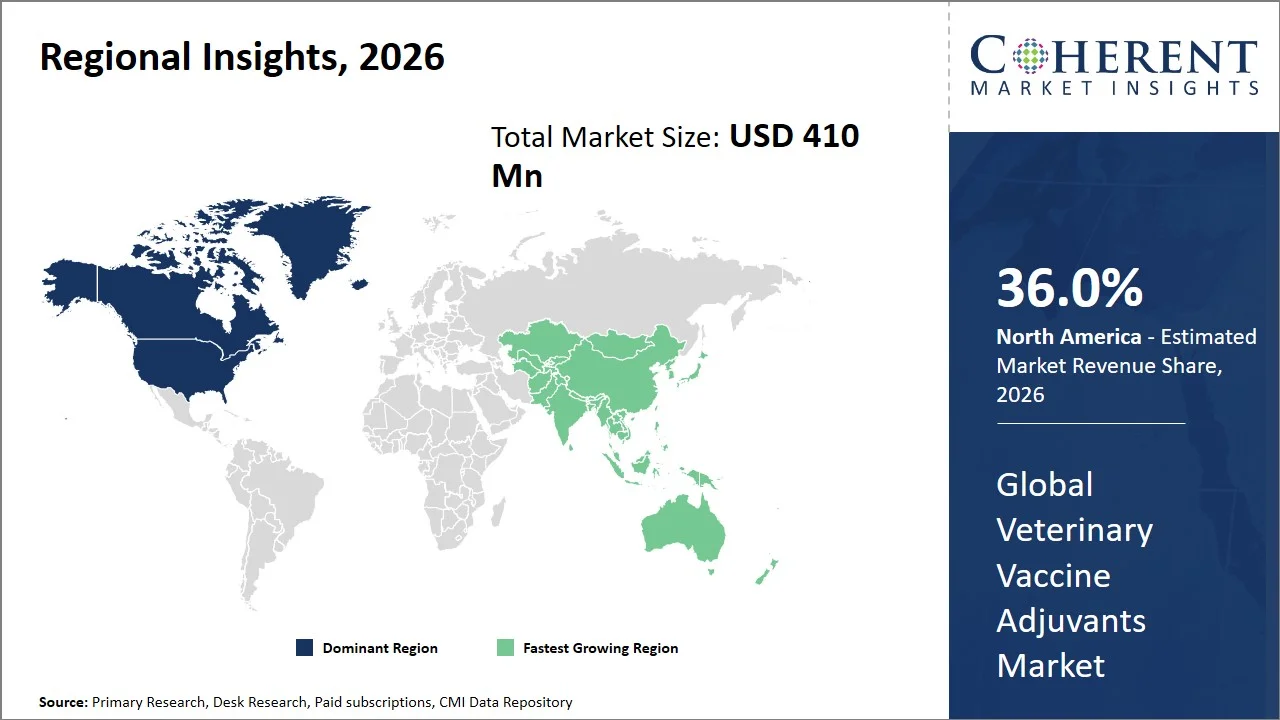
- North America maintains dominance with an expected share of 36.0%in 2026, attributed to advanced R&D pipelines and accelerated licensing pathways for novel biologics. For instance, U.S. Department of Agriculture (USDA) has approved novel products like Oncept Canine Melanoma Vaccine, which is an immunotherapeutic vaccine using DNA technology, under its veterinary biologics program. This particular product received conditional approval from the agency, enabling it to enter the market at an earlier stage, provided that it is safe and reasonably expected to be effective, illustrating how regulatory flexibility can help foster novel vaccine technologies and their adjuvants. (Source: U.S. Department of Agriculture)
- Asia Pacific is expected to exhibit the fastest growth with an estimated contribution of 34.0% share in 2026, fostered by strengthening veterinary health governance and structured biosecurity frameworks. For instance, Australian Animal Health System is implemented by Department of Agriculture, Fisheries and Forestry, consisting of national surveillance, emergency preparedness for animal diseases, and vaccine response to livestock diseases. This framework prioritizes early diagnosis, swift reaction, and immunization, ensuring the continual need for veterinary vaccines and adjuvants across the region. (Source: Department of Agriculture, Fisheries and Forestry)
- Rising Demand for Thermostable Adjuvants in Emerging Markets: The inadequate availability of refrigerated transportation systems, in many parts of Asia Pacific, Africa, and Latin America, is leading to the increased necessity of designing thermostable vaccines. The use of adjuvants that increase the stability of the antigen under elevated temperatures are on the rise especially in developing countries, mainly due to their application in livestock vaccination campaigns in remote areas. This presents an opportunity for firms to manufacture heat-stable adjuvant systems that cut down on logistic costs. Firms that explore this opportunity will tap into the unexploited markets in resource-constrained settings.
- Growing Adoption of Novel Adjuvants for Companion Animal Vaccines: Rising pet humanization and increased investments in prophylactic veterinary services are spurring demands for efficient vaccines with better safety and effectiveness profiles. New types of adjuvants, including saponin-based, polymer-based, and nanoparticle-based adjuvants, are emerging as an area of interest in improving immunity without causing any side-effects. The emerging trend provides new opportunities in premium companion animals, especially North America and Europe. The companies that invest in next-generation adjuvant technologies can secure their positions in premium veterinary therapeutics.
Why Does Aluminum-based Adjuvants Segment Dominate the Global Veterinary Vaccine Adjuvants Market?
Aluminum-based adjuvants are projected to hold the market share of 34.0% in 2026, attributed to their scientifically proven immunogenic effect, a long and consistent history of safety and low costs when used across different vaccines. From a scientific perspective, the aluminum compounds like aluminum hydroxide and phosphate adjuvants improve vaccine efficacy through stimulating of powerful antibody (Th2) response, immune cell activation and antigen absorption, thus providing effective protection even in the presence of purified or inactive antigens. (Source: National Library of Medicine)
Moreover, they can lower the required amount of antigen and decrease the frequency of injections, making their implementation easier at a larger scale in mass vaccination campaigns for cattle. Most importantly, aluminum adjuvants have been utilized for almost a hundred years now in vaccines for both humans and animals, with a proven track record for safety and acceptability, making it their top pick for commercial vaccines (Source: ScienceDirect). Furthermore, in practical veterinary applications, they are commonly used in vaccines against diseases such as Foot-and-Mouth Disease and brucellosis, strengthening their supremacy in large-scale vaccination programs. (Source: Ceva)
Why are Livestock Animals the Largest Segment in the Global Veterinary Vaccine Adjuvants Market?
To learn more about this report, Request Free Sample
Livestock animals are projected to hold a market share of 62.0% in 2026, owing to its significance in food production, economical significance, and vulnerability to diseases due to rearing in large herds. According to the Food and Agriculture Organization, livestock plays an important role in food security and provides almost 40% share of agriculture gross domestic product, thus making animal vaccination crucial (Source: Food and Agriculture Organization). Vaccination against diseases not only helps avoid productivity loss but also prevents potential health risks, especially in densely populated livestock. This is because the animals are highly susceptible to the spread of diseases in large herds; hence, vaccines and adjuvants are used widely in their prevention.
In livestock, diseases like bovine brucellosis are common among the cattle and small ruminants. Governments, therefore, adopt measures that ensure mandatory vaccinations to avoid economic loss due to disease outbreak. For instance, New Zealand government regulates brucellosis in cattle through their biosecurity system, which is overseen by the Ministry for Primary Industries, where stringent herd testing, movement regulations, and vaccination-based disease management protocols have helped to effectively control the disease and nearly eradicate it, safeguarding livestock productivity and export markets (Source: Ministry for Primary Industries). Moreover, the immunization of livestock contributes to the incomes of farmers, as millions rely on the animals for sustenance and economic gain, thereby ensuring continuous demand for vaccines and adjuvants in this category.
Injectable Segment Dominates the Global Veterinary Vaccine Adjuvants Market
The injectable segment is projected to hold a market share of 72.0% in 2026, attributed to its better immunogenicity, dosage accuracy, and fast systemic distribution when compared to other forms. Vaccine administered through injection either intramuscularly, subcutaneously or intravenously allows the antigen to be delivered straight to the tissue or blood, leading to faster absorption and stronger immune response in addition to stimulating both the humoral and cell-mediated immunity (Source: National Library of Medicine). Moreover, this delivery method is most appropriate for the administration of vaccines with adjuvants, considering that the antigen must be delivered at precisely measured rates.
The use of injectable forms of the vaccines are common for diseases such as foot and mouth, brucellosis, and bovine viral diseases in livestock populations due to the high degree of accuracy of the delivery process, as opposed to oral and intranasal administrations. For instance, a systematic review published by DergiPark AKADEMIC, pointed out that injectable vaccines need smaller amounts of antigens and are able to elicit reliable immune responses. Meanwhile, oral vaccines are prone to decomposition in the digestive tract and need higher doses for similar effects. (Source: DergiPark AKADEMIC)
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
EU Veterinary Medicinal Products Regulation (EU) 2019/6 – Ongoing Implementation Updates (2024–2025) |
|
|
U.S. FDA GFI #256 – Antimicrobial Stewardship Expansion to Medically Important Drugs (August 2024) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: European Medicines Agency, U.S. Food and Drug Administration)
Global Veterinary Vaccine Adjuvants Market Dynamics
To learn more about this report, Request Free Sample
Market Drivers
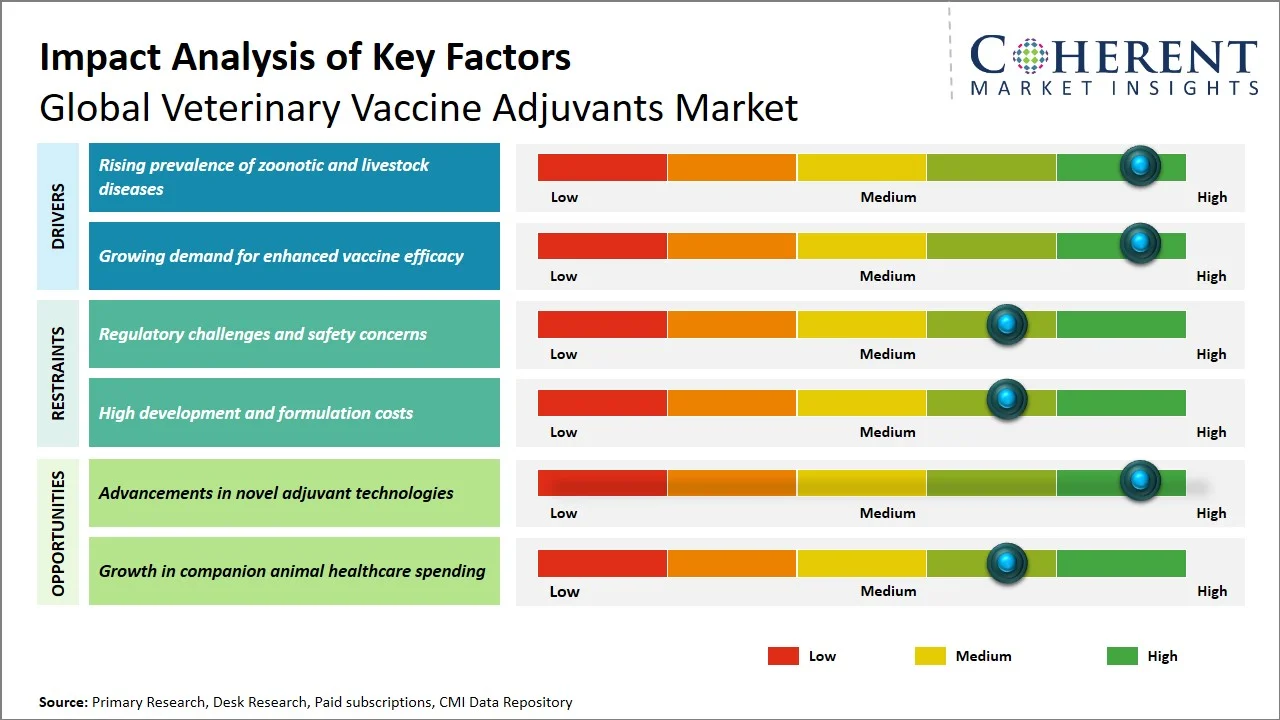
- Rising prevalence of zoonotic and livestock diseases: Increasing incidences of livestock diseases act as a key factor fueling the market growth of veterinary vaccine adjuvants. The increasing number of infections in livestock leads to increased vaccination, as the increase in disease burden correlates directly with higher demand for vaccinations. The Food and Agriculture Organization of the United Nations (FAO) has highlighted that approximately 20% of global livestock production is lost annually owing to infections. This situation threatens food security and adversely impacts the livelihoods of farmers (Source: Tridge). Diseases with high infection rates, such as African swine fever, results in 100% mortality in pigs, and no available vaccine, has led to substantial losses in the pig population worldwide, with major outbreaks in China, Southeast Asia, and parts of the Caribbean affecting hundreds of millions of pigs. Similarly, foot-and-mouth disease causes significant economic damage, with global direct production losses and vaccination costs estimated at USD 21 billion annually (Source: Food and Agriculture Organization). Moreover, endemic diseases like bovine viral diarrhea (BVD) may infect up to 60-80% of cattle herds without any control measures, thus suggesting that there is constant infection pressure within livestock herds. This high prevalence results in poor productivity, hence underscoring the significance of consistent vaccination programs. (Source: ScienceDirect)
- Growing demand for enhanced vaccine efficacy: The need for vaccines that generate highly specific, strong, and prolonged immune response has increased among livestock farmers and veterinarians alike. Modern livestock farming involves the use of highly dense livestock with a fast-spreading disease rate. Vaccines capable of generating strong immunity while using minimal doses and low antigenic load are preferred to ensure better economic and regulatory efficiency. In addition, adjuvants are extensively employed in enhancing the immunogenicity of vaccines that employ inactivated and subunit antigens for effective immunity when administered individually. For instance, in November 2024, study published by Frontiers demonstrated that nanoparticle-based vaccine formulations considerably enhanced immune responses by increasing antibody production, cytokine levels, and lymphocyte proliferation compared to conventional adjuvants, enabling stronger and longer-lasting immunity. (Source: Frontiers)
Emerging Trends
- Shift Toward Precision Adjuvant Formulations for Targeted Immune Responses: There is an increasing tendency to formulate adjuvants that induce particular immune mechanisms, such as Th1 versus Th2, based on the pathogen’s characteristics. It is especially important for challenging pathogens that do not respond well to conventional adjuvants. With advancements in immunology and molecular biology, there is now a possibility to design antigen-adjuvant pairs that improve vaccination efficacy in different species.
- Integration of Adjuvants in Multivalent and Combination Vaccines: Multivalent vaccines that provide immunity against a variety of organisms in one vaccine dose have become the new norm among vaccine producers, particularly for animal production and pets. This trend has led to a growing interest in developing adjuvant systems that will help in maintaining their efficacy while combining several antigens without triggering any immune response. Combination vaccines offer many benefits in terms of compliance, reducing the stress of administering vaccines to animals, and ensuring optimal vaccination regimes.
Regional Insights
To learn more about this report, Request Free Sample
Why is North America a Strong Market for Veterinary Vaccine Adjuvants?
North America leads the global veterinary vaccine adjuvants market, accounting for an estimated 36.0% share in 2026, owing to the presence of well-developed regulatory policies, advanced research & development infrastructure, and proactive programs on animal disease prevention. The regulatory body such as U.S. Department of Agriculture (USDA) plays a significant role, with its center for veterinary biologics playing a vital part in the approval and quality of veterinary vaccines & adjuvants. (Source: U.S. Department of Agriculture)
Furthermore, the companies such as Zoetis Inc. have been making consistent investments for increasing their capacity and innovation abilities for the manufacture of biologics and vaccines. For instance, in November 2025, Zoetis Inc. made an announcement regarding its plans to expand its manufacturing base within the U.S. This move is part of Zoetis' dedication towards innovating the field of monoclonal antibodies for companion animals. This investment seeks to improve production capabilities and increase global competitiveness in next-generation biologics for pet care (Source: Zoetis Inc.). Additionally, the industry has benefited from professional guidelines and recommendations of vaccinations by organizations such as American Veterinary Medical Association, which focuses on providing preventive healthcare to animals (such as, core vs. non-core vaccination classification, species-specific immunization schedules, booster dose intervals, risk-based vaccination protocols, and adverse event reporting guidelines).
Why Does Asia Pacific Veterinary Vaccine Adjuvants Market Exhibit High Growth?
The Asia Pacific veterinary vaccine adjuvants market is expected to exhibit the fastest growth with an estimated contribution of 34.0% share in 2026, owing to the increase in livestock production activities, rise in threats of cross-bred infections, and support from the government for animal health projects. For instance, China continues to maintain extensive mandatory vaccine campaigns against poultry flu and foot-and-mouth disease among other animal illnesses under the supervision of the Ministry of Agriculture and Rural Affairs. (Source: Ministry of Agriculture and Rural Affairs of the People's Republic of China)
Additionally, Australia enforces rigorous animal health policies and practices under the management of the Department of Agriculture, Fisheries and Forestry. This department emphasizes high levels of biosecurity and vaccination, resulting in the promotion of the use of vaccines with superior efficacy in the country (Source: Department of Agriculture, Fisheries and Forestry). Furthermore, vaccines have been successfully incorporated into national biosecurity programs and animal health initiatives to manage and prevent diseases from spreading throughout production units (Source: Meat & Livestock Australia). The coordinated regulations together with an increase in regional production and disease control measures are driving the adoption of veterinary vaccine adjuvants in the Asia Pacific region.
Why is the U.S. Leading Innovation and Adoption in the Veterinary Vaccine Adjuvants Market?
The U.S. is the leader of innovations and adoption in the market of veterinary vaccine adjuvants attributed to the early adoption of innovative technologies, such as recombinant and RNA animal vaccines, that necessitate highly-specialized adjuvants. Universities such as the College of Veterinary Medicine at Iowa State University and Texas A&M College of Veterinary Medicine partner with industry to convert immunology studies to commercially viable vaccines.
For instance, the Iowa State University works together with the U.S. Department of Agriculture (USDA) Agricultural Research Service in conducting studies that are relevant to developing vaccines for diseases affecting pigs. An example is their collaboration in conducting studies regarding the surveillance and development of vaccines related to influenza A viruses. The researchers at agricultural research service in Ames collaborated with the Iowa State researchers in discovering new strains of viruses (Source: U.S. Department of Agriculture). Additionally, strong intellectual property activity around novel adjuvant chemistries supports continuous innovation and market leadership.
Is Japan a Favorable Market for Veterinary Vaccine Adjuvants?
Japan represents a prime target market for veterinary vaccine adjuvants owing to advanced state of its animal health industry, with the existence of strong regulatory standard ensuring high-quality vaccine adoption. The market in the country is relatively advanced with regards to the companion animals’ sector, as there is an increasing emphasis being put on disease prevention through vaccination. Japan is focused on ensuring that livestock is effectively monitored and that diseases are under control; therefore, there is an ongoing demand for high quality vaccines. Additionally, the existence of domestic manufacturers such as Kyoritsu Seiyaku Corporation further strengthens local production and innovation.
Is China Emerging as a Key Growth Hub for the Veterinary Vaccine Adjuvants Market?
China has emerged as one of the growth centers of the veterinary vaccine adjuvants market due to stringent disease control measures undertaken by the government (such as, compulsory vaccination programs for avian influenza and foot-and-mouth disease, nationwide animal disease surveillance systems, movement control and quarantine protocols for livestock transport, culling policies during outbreak containment, and routine farm-level biosecurity inspections), as well as increasing production of vaccines locally (e.g., China’s vaccine supply expanded to ~666 million–1.19 billion doses annually, with the majority produced domestically, alongside growth in GMP-certified manufacturers to over 70 facilities, reflecting significant scale-up in production capacity and infrastructure modernization).
The China government conducts mandatory vaccinations for vital diseases such as avian influenza and foot-and-mouth disease under the oversight of the Ministry of Agriculture and Rural Affairs. Simultaneously, prominent domestic companies such as China Animal Husbandry Industry Co., Ltd. is ramping up their vaccine production capacities alongside biologics, thus requiring better adjuvant formulations. Fast-paced development in intensive livestock farming practices along with increased pet animal ownership in the country will continue to drive vaccine sales, resulting in growth opportunities within the China veterinary vaccine adjuvants market.
Why Does Germany Top the Europe Veterinary Vaccine Adjuvants Market?
Germany is the leader in the veterinary vaccine adjuvants market of Europe because of its stringent animal health policies (such as, mandatory notification of infectious animal diseases under the Animal Health Act, routine surveillance and control programs for diseases such as African swine fever and avian influenza, strict livestock movement and traceability regulations, farm-level biosecurity compliance requirements, and vaccination-based disease prevention strategies aligned with EU animal health frameworks)., compulsory disease prevention measures, and frameworks based on standards set by the European Medicines Agency (EMA).
The nation boasts an efficient livestock industry where vaccinations are regularly carried out for infections, including those that affect cattle and swine, thus guaranteeing constant demand for vaccines using adjuvants. The strong influence of international companies like Boehringer Ingelheim also promotes advancements and manufacturing of biological products. Moreover, Germany’s commitment to lowering antibiotic consumption in animal husbandry hastens the move towards immunization.
Is Veterinary Vaccine Adjuvants Market Developing in Brazil?
Brazil represents one of the emerging markets for veterinary vaccines adjuvants, primarily because of its focus on becoming a disease-free certified zone in which a consistent vaccination process is essential. With such a move towards disease-free zones, Brazil is shifting from a reactive system that addresses diseases as they arise to a preventative approach where animals are vaccinated against potential diseases before they occur.
Furthermore, the adoption of technology in monitoring and tracing the health of livestock has ensured better vaccination programs and increased use of adjuvant products. Additionally, the increased use of export-oriented and high-value meat production chains will increase pressure on farmers to implement more reliable and immunogenic vaccines.
Regulatory Landscape Governing the Global Veterinary Vaccine Adjuvants Market
|
Region
|
Key Regulatory Authorities |
Core Regulations |
Key Requirements & Focus Areas |
|
North America |
U.S. Department of Agriculture (USDA – APHIS CVB) |
Virus-Serum-Toxin Act (VSTA); 9 CFR regulations |
Licensing of veterinary biologics, serial (batch) release testing, potency & safety validation, labeling compliance, and post-market surveillance |
|
Europe |
European Medicines Agency (EMA) |
Regulation (EU) 2019/6; GMP implementing regulations |
Centralized authorization, pharmacovigilance systems, antimicrobial resistance controls, Union Product Database, and harmonized safety/efficacy standards |
|
Asia Pacific |
Central Drugs Standard Control Organization (India); national authorities (e.g., Australia DAFF) |
Central Drugs Standard Control Organization (India); national authorities (e.g., Australia DAFF) |
Approval of veterinary biologics, field trials, stability testing, import licensing, and post-marketing surveillance requirements for vaccines and adjuvants |
|
Latin America |
National regulatory bodies (e.g., MAPA Brazil, SENASA Argentina) |
WHO-aligned and country-specific biologics regulations |
Increasing alignment with WHO guidelines for biotherapeutics, focus on post-approval changes (PACs), quality consistency, and regulatory convergence efforts |
|
Middle East & Africa |
National ministries; supported by World Organization for Animal Health (WOAH) |
WOAH standards; national veterinary legislation |
Emphasis on disease control standards, vaccine quality assurance, and regulatory capacity building, with growing adoption of international guidelines for biologics |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How are the advancements in novel adjuvant technologies creating new growth opportunities in the global veterinary vaccine adjuvants market?
The emergence of new adjuvant technologies are offering ample opportunities for the global veterinary vaccine adjuvants market attributed to the development of next-generation adjuvants that will offer better control over the delivery mechanism and deliver effective delivery of antigens via regulated kinetics. Studies show that newer technologies such as nanoparticle adjuvants, cytokine adjuvants, and ISCOMS allow better vaccine development and enhance the delivery of antigens. The use of nanoparticles as adjuvants will enable the development of better vaccines for animals with improved immune responses and improved antigen uptake and innate immunity. (Source: MDPI)
Furthermore, improvements in cytokines, such as interleukin and interferon adjuvants, have been instrumental in the development of better vaccines due to the enhanced immune response offered by these adjuvants (Source: Frontiers). Developments in adjuvant technologies will aid in the development of better vaccines for diseases such as avian flu, African swine fever, and bovine infections. Traditional vaccines have proven ineffective in treating these diseases; therefore, they require potent adjuvants. Innovations in adjuvant technologies will play a pivotal role in developing advanced vaccines, thereby driving the market.
Market Players, Key Development, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In November 2025, Zoetis Inc. introduced Vanguard Recombishield, which is an injectable vaccine for canines in the U.S. meant to provide protection from the bacteria Bordetella bronchiseptica, one of the leading causes of kennel cough. This new vaccine uses cutting-edge antigen technology that allows for enhanced immune response and better tolerance in dogs. This vaccine is approved for use in dogs eight weeks old and above.
- In March 2025, Boehringer Ingelheim released its latest swine vaccine, INGELVAC CIRCOFLEX AD, in the United States to deliver improved protection against porcine circovirus type 2 (PCV2). This novel vaccine is a unique blend of both PCV2a and PCV2d antigens in a single vaccination dose, providing superior protection against more common viruses. It is designed to enhance the duration and strength of immunity in pigs.
Competitive Landscape
The global veterinary vaccine adjuvants market will likely stay highly competitive due to the continuous developments taking place in the field of immunology, formulation science, and vaccine administration techniques. The market leading players like Zoetis Inc., Merck Animal Health, and Boehringer Ingelheim will emphasize on the development of novel adjuvants and multivalent vaccines in order to gain a competitive advantage over other companies. Partnerships and alliances among the companies are rising, especially in terms of developing novel adjuvants and biologicals. In addition, the firms are adopting efficient production technologies that will help in addressing the requirement of vaccinations for large livestock population. Key focus areas include:
- Development of next-generation adjuvants
- Expansion of multivalent and combination vaccines
- Strengthening R&D collaborations and partnerships
- Scaling up cost-efficient manufacturing and supply chains
- Enhancing regulatory compliance and product standardization
Market Report Scope
Veterinary Vaccine Adjuvants Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 410 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.6% | 2033 Value Projection: | USD 640 Mn |
| Geographies covered: |
|
||
| Segments covered: |
By Application: Bacterial Vaccines, Viral Vaccines, and Parasitic Vaccines By End User: Veterinary Pharmaceutical and Biopharmaceutical Companies, Contract Research and Manufacturing Organizations (CROs/CMOs), Research and Academic Institutes, and Veterinary Clinics and Hospitals |
||
| Companies covered: |
Zoetis Inc., Merck Animal Health, Elanco Animal Health, Ceva Santé Animale, Boehringer Ingelheim Animal Health, HIPRA, Phibro Animal Health Corporation, Virbac, Vaxxinova, and Seppic |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- In the coming years, the prospects of the veterinary vaccine adjuvants market is expected to experience a shift from traditional aluminum and oil-based adjuvants towards advanced next-generation adjuvants capable of controlling the immune system in a disease-dependent manner, either through humoral immunity or cellular immunity. The main emerging trends in the sector include nanotechnology delivery platforms, immunomodulatory molecules like toll-like receptor ligands, and drug delivery systems. This shift guarantees sustained efficacy despite lowering doses of the products. Moreover, the increased pressure to reduce antibiotic use alongside new disease challenges will force the animal health sector into developing more vaccines, requiring innovative adjuvants.
- The largest opportunities are anticipated to be found in the nanoparticle and advanced emulsion adjuvant category among livestock vaccines, especially in Asia Pacific countries like China and India. The high density of livestock and the occurrence of several diseases are the key factors contributing to the increased production of highly efficient vaccines by the government in order to improve productivity. In terms of application, viral vaccines (like FMD, avian influenza, and PCV) will be a good opportunity owing to its economic significance and requirement for a strong response from the immune system.
- In order to establish a competitive advantage, companies must ought to think about investing in R&D in order to come up with innovative adjuvant technology which bears fruits in relation to its immunological impact, durability, and safety. Partnering strategically with biotech firms, research centers, and vaccine manufacturers would definitely be crucial towards innovation and rapid product commercialization. Moreover, the companies should enhance their knowledge about regulations and clinical trials because biological drug and vaccine approval processes are becoming extremely strict around the world. Operation-wise, it will be crucial to take into account cost factors in manufacturing since animals tend to be very sensitive to pricing.
Market Segmentation
- Product Type Insights (Revenue, USD Mn, 2021 - 2033)
- Aluminum-based Adjuvants
- Oil-based Adjuvants
- Emulsion-based Adjuvants
- Others
- Animal Type Insights (Revenue, USD Mn, 2021 - 2033)
- Livestock Animals
- Companion Animals
- Route of Administration Insights (Revenue, USD Mn, 2021 - 2033)
- Injectable
- Oral
- Intranasal
- Application Insights (Revenue, USD Mn, 2021 - 2033)
- Bacterial Vaccines
- Viral Vaccines
- Parasitic Vaccines
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Veterinary Pharmaceutical and Biopharmaceutical Companies
- Contract Research and Manufacturing Organizations (CROs/CMOs)
- Research and Academic Institutes
- Veterinary Clinics and Hospitals
- Regional Insights (Revenue, USD Mn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Zoetis Inc.
- Merck Animal Health
- Elanco Animal Health
- Ceva Santé Animale
- Boehringer Ingelheim Animal Health
- HIPRA
- Phibro Animal Health Corporation
- Virbac
- Vaxxinova
- Seppic
Sources
Primary Research Interviews
- Veterinary vaccine manufacturers and R&D heads
- Academic immunologists and veterinary researchers from institutions working on adjuvant development and animal vaccinology
- Contract research organizations (CROs) and formulation experts specializing in vaccine adjuvant systems
Stakeholders
- Veterinary Vaccine Adjuvant Providers: pharmaceutical companies, specialty adjuvant manufacturers, and biotech firms focused on immunomodulators
- End-use Sectors:
- Veterinary hospitals and clinics
- Livestock farms and poultry producers
- Companion animal healthcare providers
- Research and academic institutes
- Regulatory & Health Bodies: U.S. Food and Drug Administration (FDA) – veterinary biologics safety and approval, European Medicines Agency (EMA) – regulatory frameworks for animal vaccines, World Health Organization – One Health initiatives linking animal and human vaccine development
Databases
- ClinicalTrials.gov – clinical and preclinical studies on veterinary vaccines and adjuvants
- World Health Organization database – zoonotic disease burden and vaccination programs
- Centers for Disease Control and Prevention – animal disease surveillance and zoonotic infection data
- OECD Health Statistics – veterinary healthcare expenditure and biologics usage
Magazines
- Nature (Biotechnology / Immunology sections) – advancements in vaccine adjuvant technologies
- Science Magazine – innovations in immunology, nanotechnology-based adjuvants, and vaccine delivery systems
- NIH Research Matters – updates on immunology and translational vaccine research
Journals
- Veterinary Immunology and Immunopathology – core research on veterinary immune responses and adjuvants
- Frontiers in Immunology – studies on novel adjuvants such as cytokine-based systems
- Journal of Veterinary Internal Medicine – mechanisms and applications of vaccine adjuvants in animals
- Clinical and Vaccine Immunology – research on immune responses and vaccine technologies
Newspapers
- The Times of India – covers government-led animal vaccination drives, zoonotic disease control programs, and livestock immunization initiatives (e.g., anti-rabies campaigns)
- The Hindu – reports on veterinary healthcare policies, animal disease outbreaks, and public vaccination programs in India
- The New York Times – global coverage on zoonotic diseases, pandemic preparedness, and vaccine research impacting animal health
- The Guardian – insights on antimicrobial resistance, livestock health regulations, and One Health initiatives
- Financial Times – business and industry trends in animal health companies, vaccine investments, and biotech developments
- China Daily – coverage on livestock disease control, large-scale vaccination programs, and government initiatives in Asia
Associations
- World Organization for Animal Health (WOAH/OIE) – global animal disease control and vaccination standards
- American Veterinary Medical Association (AVMA) – veterinary healthcare guidelines and insights
- European Federation of Animal Health (AnimalhealthEurope) – industry trends and regulatory developments
Public Domain Sources
- National Institutes of Health (NIH) – funding and research on immunology and vaccine adjuvants
- Centers for Disease Control and Prevention – zoonotic disease monitoring and vaccine-related data
- European Commission – funding programs for animal health and One Health initiatives
Proprietary Elements
- CMI Data Analytics Tool
- Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients