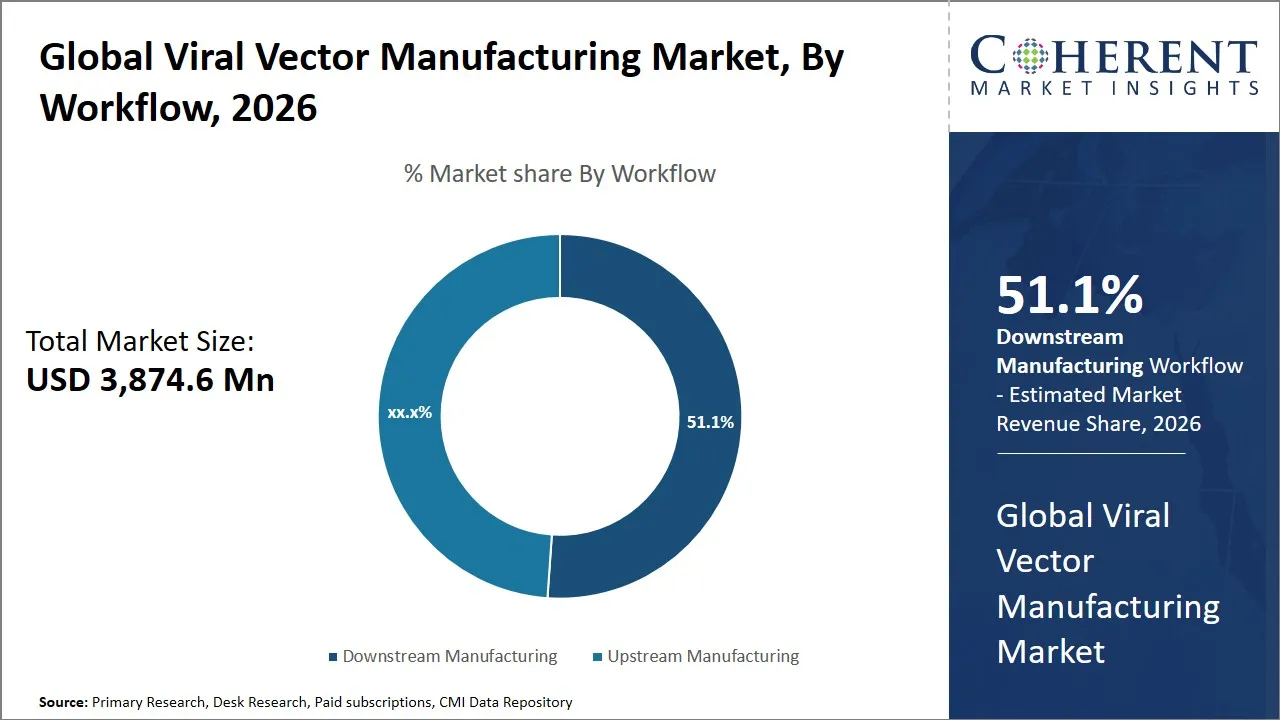
Global Viral Vector Manufacturing Market Size and Forecast – 2026-2033
The global viral vector manufacturing market is estimated to be valued at USD 3,874.6 Mn in 2026 and is expected to reach USD 8,829.1 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 17.6% from 2026 to 2033. This significant growth is driven by increasing demand for gene therapies, rising prevalence of genetic disorders, and advancements in biopharmaceutical technologies, positioning the market for strong expansion over the forecast period.
Key Takeaways of the Global Viral Vector Manufacturing Market
- Downstream manufacturing segment is expected to lead the global viral vector manufacturing market, capturing 51.1% share in 2026.
- Adeno-associated viruses (AAVs) segment is estimated to represent 51.7% of the global viral vector manufacturing market share in 2026.
- Product segment is projected to dominate with 80.0% of the global viral vector manufacturing market share in 2026.
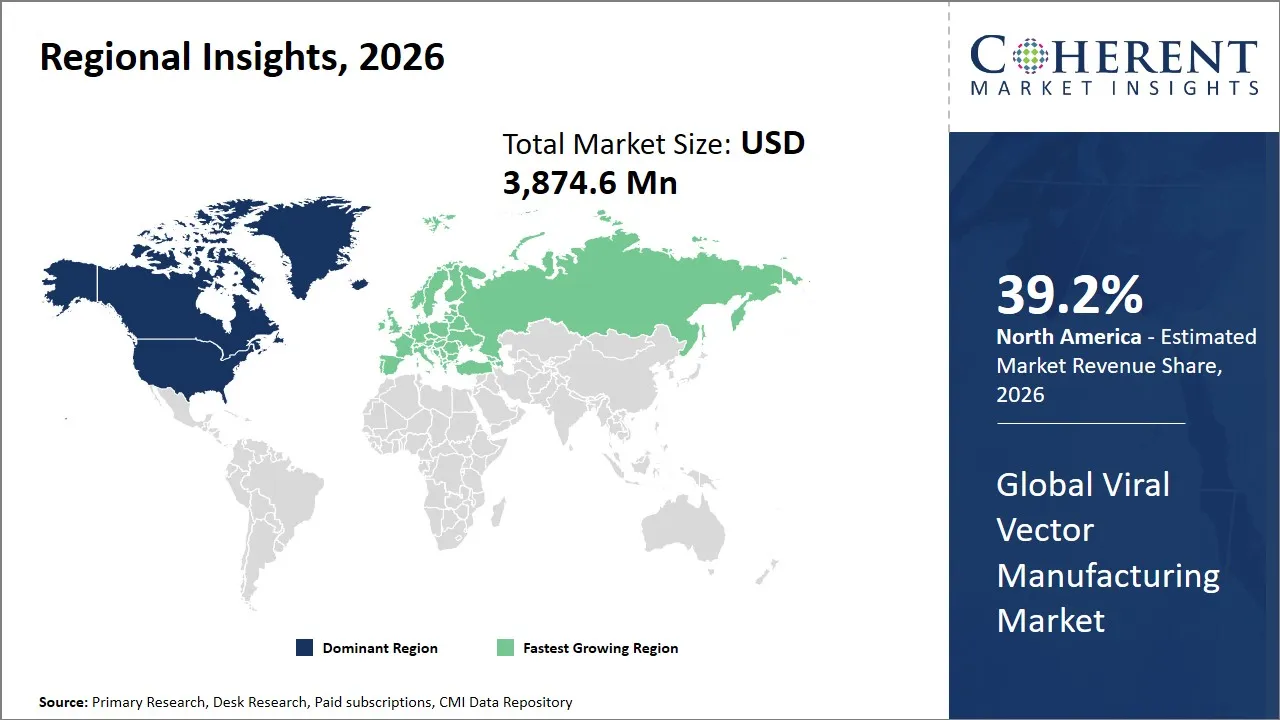
- North America is expected to lead the market, holding a share of 39.2% in 2026. Europe is anticipated to be the fastest-growing region, with 28.5% share in 2026.
Market Overview
- The global viral vector manufacturing market is expanding steadily as gene therapy and cell therapy pipelines move further into late-stage development and commercialization, which is increasing demand for reliable large scale viral vector supply.
- AAV and lentiviral vectors remain the most important platforms in the market because they are widely used across in vivo and ex vivo gene therapy programs. Their growing clinical relevance is directly shaping investment in upstream production, purification, analytics, and fill finish capabilities.
- Manufacturing scalability is one of the biggest priorities in the market. Companies are focusing on suspension-based systems, better purification workflows, and more automated end to end production models to improve yields, consistency, and batch turnaround.
- The market is also benefiting from broader adoption of advanced analytics and tighter process control, as developers and regulators place greater emphasis on product quality, potency, and reproducibility. Stronger analytical capabilities are becoming essential for both regulatory readiness and commercial manufacturing performance.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Regulatory flexibility for cell and gene therapy manufacturing |
|
|
Rising industry focus on standardized analytics and reference materials |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Downstream Manufacturing Segment Dominate the Global Viral Vector Manufacturing Market in 2026?
The downstream manufacturing segment is expected to hold the largest market share of 51.1% in 2026, owing to its indispensable role in ensuring the final product's safety, potency, and efficacy. Downstream manufacturing encompasses processes such as purification, fill-finish, vector recovery, and harvesting, which are essential steps following the initial upstream production and vector amplification phases.
The primary driver behind the dominance of downstream manufacturing is the increasing complexity of viral vectors, which necessitates sophisticated purification strategies to remove impurities, cellular debris, and unwanted viral particles. The high specificity required for therapeutic applications, especially in gene therapy and vaccine development, compels manufacturers to adopt advanced chromatography, filtration, and ultracentrifugation techniques. These processes ensure that viral vectors meet stringent regulatory standards for clinical use, as contamination or inadequate purification could lead to adverse patient outcomes or compromised therapeutic efficacy.
For instance, in May 2024, Charles River launched AAV and lentiviral vector reference materials to streamline GMP transition, strengthen downstream purification workflows, and improve analytical standardization in viral vector manufacturing.
(Source - Charles River)
Why Does the Adeno-associated viruses (AAVs) Segment Dominate the Global Viral Vector Manufacturing Market in 2026?
The Adeno-associated viruses (AAVs) segment is expected to hold 51.7% of the market share in 2026, propelled by their broad applicability across gene therapy and vaccine platforms. The combination of efficacy, low immunogenicity, and non-pathogenic properties makes AAVs a popular choice of vectors, as they are safer options than other viral vectors.
One of the foremost factors behind the dominance of AAVs is their ability to transduce dividing and non-dividing cells reliably, which makes them highly versatile for treating a wide array of genetic disorders affecting different tissues. The stable integration and long-term expression capabilities of AAV-derived vectors enable sustained therapeutic benefits, a critical consideration for chronic diseases requiring durable gene expression.
For instance, in October 2024, Forge Biologics launched the FUEL AAV Manufacturing Platform to enhance productivity, scalability, and manufacturing efficiency for AAV-based gene therapy programs, reinforcing the leadership of the AAV segment in viral vector manufacturing.
(Source - Forge Biologics)
The Product Segment Dominates the Global Viral Vector Manufacturing Market
The product segment is expected to hold the largest market share of 80.0% in 2026, driven by rising demand for ready-to-use viral vectors and bioprocess technologies. The product segment will mainly consist of the viral vectors themselves, raw materials, reagents and special consumables needed to develop and process vectors.
One of the growth opportunities of this segment is due to the growing interest in ready-to-use, high-quality viral vectors products, which save time to clinic and enable a simplified development of therapeutics. To avoid the complicated in-house production that can be very resource intensive and need specialized knowledge, manufacturers and biopharmaceutical firms are looking to buy well-characterized viral vectors products.
Capacity Expansion and Regional Manufacturing Localization in the Global Viral Vector Manufacturing Market
- Capacity Expansion- Capacity continued to be expanded globally in the viral vectors manufacturing sector as companies and CDMOs attempt to minimize bottlenecks in manufacturing plasmid, upstream processing, purification, fill- finish and analytical testing. This growth is being fueled by the increased quantity of gene therapy and advanced therapy programs, as well as the necessity of expedited clinical and commercial supply. The recent industry news also highlights that North America is still the biggest manufacturing hub, followed by Europe and then Asia Pacific is catching up, as businesses seek to install new cell and gene therapy facilities.
- Regional Manufacturing Localization- Regional manufacturing localization is emerging as a strategic focus as sponsors seek to manufacture closer to clinical and commercial demand centers, enhance supply security and ease technology transfer and regulatory coordination. The latter trend is particularly evident in Asia Pacific, with new dedicated facilities being located to meet local and exporting demand, and global manufacturers moving beyond a single region concept, with increased footprint in North America, Europe and Asia Pacific. In the case of the viral vector manufacturing market, this helps to reduce lead times, improve the responsiveness on a regional basis, and reduce reliance on cross-border chains of manufacturing.
Regional Insights
To learn more about this report, Request Free Sample
North America Viral Vector Manufacturing Market Analysis and Trends
The North America region is projected to lead the market with a 39.2% share in 2026, driven by a robust biotech and pharmaceutical ecosystem, advanced infrastructure, and strong government support. The U.S., in particular, benefits from significant investment in gene therapy research and development fueled by the National Institutes of Health (NIH) and Food and Drug Administration (FDA) regulatory frameworks that support accelerated approval pathways. The presence of leading Contract Manufacturing Organizations (CMOs) such as Lonza, Catalent, and Thermo Fisher Scientific enhances manufacturing capabilities and quality standards. The region’s well-established supply chain, access to highly skilled talent, and collaborations between academic institutions and industry players further reinforce its leadership position. Canada also contributes with growing biotech hubs supported by favorable government initiatives and funding.
For instance, in May 2023, AGC Biologics launched its BravoAAV and ProntoLVV platforms to support faster, scalable GMP viral vector manufacturing, strengthening North America’s position in advanced gene therapy production.
(Source - AGC Biologics)
Europe Viral Vector Manufacturing Market Analysis and Trends
The Europe region is expected to exhibit the fastest growth in the viral vector manufacturing market contributing 28.5% share in 2026, due to expanding healthcare infrastructure, increasing government incentives, and rising adoption of gene therapies across emerging economies. Europe is a central center of interest in the viral vectors manufacturing market as it is a region with a robust research base on gene therapies, established biopharma infrastructure and an increase in investment in specialized manufacturing capacity. Rising demand in adeno-associated virus and lentiviral vector production is benefiting the region, and businesses are also increasing localized plants to enhance supply security, accelerate clinical support, and enhance commercial preparedness through the European markets.
For instance, in September 2024, Rentschler Biopharma announced the launch of an expanded service offering at its advanced therapies facility in Stevenage, U.K., introducing a new lentiviral vector manufacturing toolbox. The offering includes process development, cGMP manufacturing, and integrated analytical and regulatory support, complementing its existing adeno-associated viral vector services. The initiative is aimed at supporting growing demand for advanced therapy medicinal products by providing end-to-end manufacturing solutions for gene and cell therapy developers.
(Source - Rentschler Biopharma)
Global Viral Vector Manufacturing Market Outlook for Key Countries
How is the U.S. Helping in the Growth of the Viral Vector Manufacturing Market?
U.S. holds a dominant position in the viral vector manufacturing market through its innovative technology platforms and well laid regulatory structure to speed up clinical adoption. Major industry participants such as Thermo Fisher Scientific, Catalent, and Brammer Bio have considerable manufacturing presence and are still investing in capacity additions. The leadership of the country in gene therapy clinical trials creates ongoing need of production of the vectors in scale. Moreover, the combinations of large pharma companies and dedicated CMOs create innovation and provide quality supply chains.
How is China Helping in the Growth of the Viral Vector Manufacturing Market?
The viral vector manufacturing industry in China is also developing fast, and the government takes serious measures by providing substantial funding to the sector via programs like Made in China 2025 and strategic policies in support of biotechnology. Domestically based firms such as WuXi AppTec and GenScript are building their production capabilities in viral vectors with both research-grade and commercial-level manufacturing. Increasing numbers of rare diseases and cancer therapies in China are creating a need in viral vectors technology, which has led to international collaboration with multinational pharmaceutical firms.
Key Drivers for the Growth of the Germany Viral Vector Manufacturing Market
The Germany viral vector manufacturing market is supported by an excellent biotechnology sector and highly developed regulatory and quality standards in accordance with the European Medicines Agency (EMA). Developers of viral vectors and manufacturers operating in the country include BioNTech and Miltenyi Biotec, which play an important role in the innovation and production of viral vectors. Research and development of technology through academic-industry partnerships is a strength of Germany and the environment through which scale-up and commercialization of manufacturing can occur is supported by government funding and incentives.
Japan Viral Vector Manufacturing Market Trends
Japan continues to lead viral vector manufacturing market in the Asia Pacific due to advanced technological expertise and well-defined regulatory pathways tailored for regenerative medicine and gene therapies. Companies like Takeda Pharmaceutical and AGC Biologics have developed extensive manufacturing infrastructure catering to domestic and international demand. The Japanese government has been a proponent of gene therapy programs which have made treatments more accessible and less time-to-market.
Shift Toward Scalable and Cost-Efficient Production Platforms in the Global Viral Vector Manufacturing Market
- Adoption of Scalable Manufacturing Technologies- The market for viral vector manufacturing is quickly transitioning to scalable manufacturing technologies including suspension cell culture technologies, single use bioreactors, and automated upstream-downstream integration. These technologies allow manufacturers to leave the small batch and labor-intensive production process in favor of higher volume reproducible production. With increased pipeline gene therapy, scalability is no longer a luxury; it is necessary to supply late-stage clinical trials and commercial supply without affecting the quality of the vectors or consistency of yield.
- Focus on Cost Efficiency and Process Optimization- Certainly, the pressure of costs is compelling companies to streamline production by enhancing purification processes via better yield of vectors, simplified purification procedures and minimized manual intervention. Standardized processes, use of platform-based manufacturing processes, and continuous bioprocessing are also becoming commonplace in an effort to reduce cost per dose and turnaround time. This is important because high costs of production are currently one of the greatest obstacles to wider adoption of gene therapy and thus cost-effective production is one of the primary competitive advantages in the market.
Market Players, Key Developments, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In October 2025, DINAMIQS (a Siegfried company) inaugurated Switzerland’s first cGMP viral vector manufacturing facility in Zurich, enabling end-to-end production from R&D to commercial supply.
- In October 2025, Oxford Biomedica (OXB) acquired a commercial-scale, U.S. FDA approved viral vector manufacturing facility in North Carolina from a Resilience subsidiary, strengthening its U.S. GMP capacity and expanding its global CDMO network.
- In February 2025, Novartis opened its first specialized viral vector manufacturing facility (VIFA One) in Slovenia, backed by a USD 46 million (EUR 40 million) investment to enhance production of vectors for advanced gene and cell therapies.
Top Strategies Followed by Global Viral Vector Manufacturing Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Leading players compete by investing heavily in R&D to improve viral vector yield, safety, and scalability while also using partnerships and global expansion to strengthen manufacturing reach and customer access. Their advantage comes from stronger technical capabilities, broader supply networks, and the ability to commercialize faster across major and emerging markets. |
In October 2025, Oxford Biomedica (OXB) acquired a U.S. FDA-approved commercial-scale viral vector manufacturing facility in North Carolina to expand its U.S. GMP capacity and strengthen its global CDMO network, showing how established players use strategic expansion to reinforce market leadership. |
|
Mid-Level Players |
Mid-level companies in the viral vector manufacturing market compete by offering dependable manufacturing services at a more affordable price point than large global players. Their focus is usually on serving smaller biotech firms, emerging therapy developers, and research organizations that need quality support but have tighter budgets. To stay competitive, these companies often strengthen their position through partnerships, technology access deals, and selective capacity expansion that improve efficiency without the cost burden of building a fully integrated global network. |
Matica Biotechnology expanded its viral vector CDMO presence by securing multiple new cell and gene therapy manufacturing projects in 2025 and positioning its U.S. facility and platform capabilities as a cost-effective outsourcing option for therapy developers. |
|
Small-Scale Players |
Small-scale players in the viral vector manufacturing market usually compete by focusing on niche technologies, specialized vector platforms, or process innovations that improve yield, purity, or flexibility. Since they do not have the scale of large manufacturers, they often rely on local partnerships and targeted collaborations to access technical expertise, regional customers, and regulatory support. |
PCI Biotech has been developing a photochemical technology to improve adeno-associated virus manufacturing by increasing extraction efficiency and reducing impurities, showing how smaller players try to stand out through specialized innovation rather than scale. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
Viral Vector Manufacturing Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 3,874.6 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 17.6% | 2033 Value Projection: | USD 8,829.1 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Lonza, Catalent, Thermo Fisher Scientific, FUJIFILM Diosynth Biotechnologies, Merck KGaA, AGC Biologics, Charles River Laboratories, Oxford Biomedica, Viralgen, Minaris Advanced Therapies, WuXi Advanced Therapies, SK pharmteco, Resilience, uniQure, and Novartis Gene Therapies |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Viral Vector Manufacturing Market Dynamics
To learn more about this report, Request Free Sample
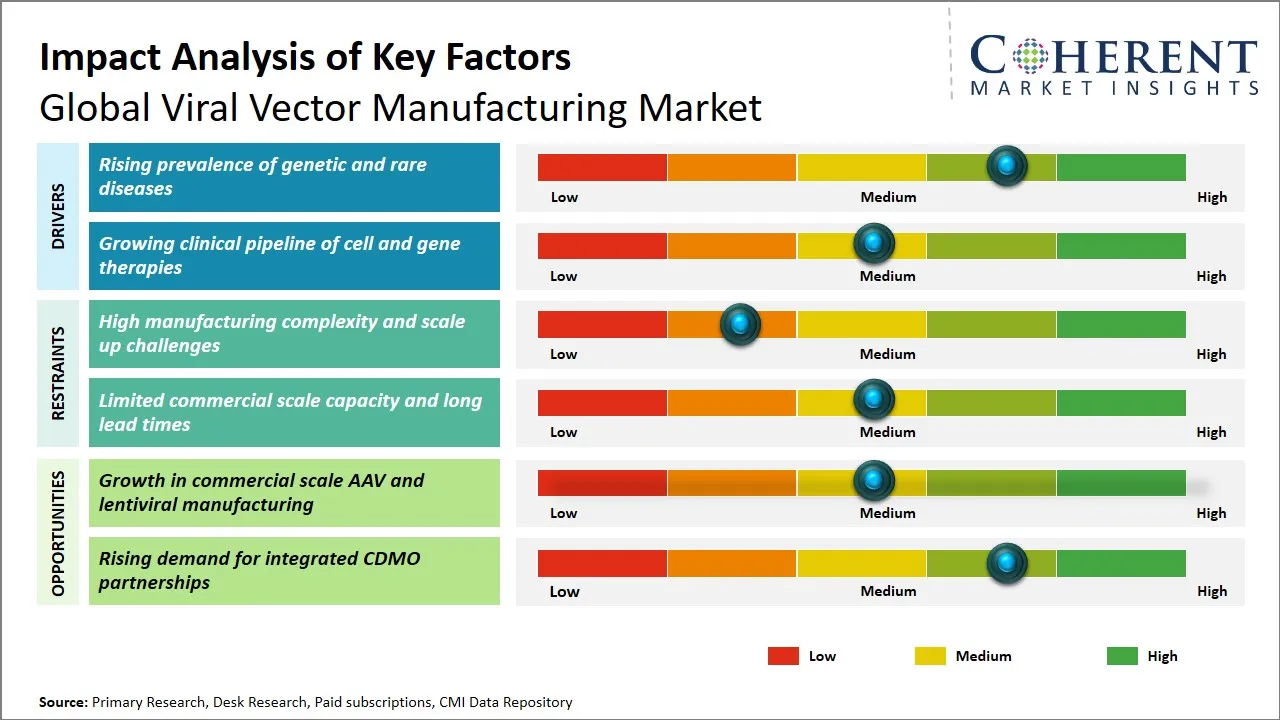
Global Viral Vector Manufacturing Market Driver - Rising Prevalence of Genetic and Rare Diseases
The rising rate of genetic and rare diseases across the globe is a major force that is gaining momentum to drive the need to manufacture viral vectors. With the progress of genomics and molecular biology that has enhanced the capacity to diagnose such ailments at an earlier and more precise stage, the advancement of gene therapies to treat such diseases has grown accordingly. The delivery vehicles, known as the viral vectors, have become an essential tool that can cure the disorders that were considered untreatable or poorly controlled in the past, focusing on their underlying causes. This increase in patient numbers, in conjunction with the greater levels of awareness and greater diagnostic abilities, has led to increased focus on creating tailored, focused gene therapies. Also, several rare diseases, some of which typically result from single-gene defects, are especially susceptible to viral vectors-based therapies which further spur investment and innovation in this manufacturing sector.
For instance, on February 24, 2026, Sarepta Therapeutics announced the commercial launch of ELEVIDYS in Japan for Duchenne muscular dystrophy, a rare genetic disease. This matters for the viral vector manufacturing market because ELEVIDYS is an adeno-associated virus vector-based gene therapy, and launches like this directly increase demand for high quality large scale viral vector production, analytical testing, and commercial supply support for rare disease treatments.
(Source- Sarepta Therapeutics)
Global Viral Vector Manufacturing Market Opportunity - Growth in Commercial-Scale AAV and Lentiviral Manufacturing
The viral vector manufacturing market production is experiencing strong growth prospects due to the rising need to produce adeno-associated virus (AAV) and lentivirus vectors in commercial scale. Such viral vectors play a significant role in gene therapy, immunotherapy, and vaccine development and form the key component of developed biopharmaceutical modalities. As clinical trials of gene therapy have increased and commercialization has begun, there is a transformation in manufacturing capacity and capabilities in production through research batches to commercial production in large scale. Such a shift is driving investments in scalable, robust and cost-effective manufacturing platforms which can will satisfy high regulatory demands. There is a growing need in AAV vectors, especially, because of their good safety profile and ability to target genetic disorders, which prompts manufacturers to increase their infrastructure and yield optimal production.
For instance, in May 2024, Donaldson’s Isolere Bio and Univercells Technologies, announced the development of an integrated lentiviral vector manufacturing platform designed to improve recoveries with a scalable process while keeping the manufacturing footprint smaller. This fits the market shift toward commercial scale lentiviral manufacturing with stronger efficiency and process integration.
(Source - Donaldson’s Isolere Bio)
Analyst Opinion (Expert Opinion)
- The viral vector manufacturing market is on the move towards a global scale as gene and cell therapy pipelines are growing and the demand to produce at a reliable clinical and commercial level. Improvements in production via suspension-based production, single use bioprocessing and purification efficiency, and a more conducive regulatory environment are supporting growth and getting developers programs onwards with a clear understanding of the way forward. Simultaneously, the market is providing new opportunities related to platform manufacturing, regional manufacturing centers and process standardization which can enhance the supply security and turnaround time decreasing.
- However, the market cannot be scaled easily. The barriers include high manufacturing costs, restricted GMP capacity, technology transfer complexity, and batch consistency challenge which are significant factors to consider as more of the drug therapies advance to late-stage development and commercialization. The events in the industry that have been conducted in the past two to three years have influenced this market by bringing together the manufacturers, therapy developers, regulators, and technology providers on a common platform of priorities scalability, compliance and innovation. Consequently, this is leading to the market being increasingly structured, increasingly regional and increasingly concerned with the long-term manufacturing efficiency as opposed to merely the early-stage ability.
Market Segmentation
- Workflow Insights (Revenue, USD Mn, 2021 - 2033)
- Downstream Manufacturing
- Purification
- Fill Finish
- Upstream Manufacturing
- Vector Amplification, Editing and Expansion
- Vector Recovery/Harvesting
- Downstream Manufacturing
- Type Insights (Revenue, USD Mn, 2021 - 2033)
- Adeno-associated viruses (AAVs)
- Lentiviruses
- Retroviruses
- Adenoviruses
- Others
- Product & Services Insights (Revenue, USD Mn, 2021 - 2033)
- Product
- Services
- Application Insights (Revenue, USD Mn, 2021 - 2033)
- Cell and Gene Therapy
- Vaccine Development
- Research
- Disease Indication Insights (Revenue, USD Mn, 2021 - 2033)
- Cancer
- Generic Disorders
- Infectious Diseases
- Others
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Pharmaceutical and Biopharmaceutical Companies
- Academics and Research Institutes
- Others
- Regional Insights (Revenue, USD Mn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Lonza
- Catalent
- Thermo Fisher Scientific
- FUJIFILM Diosynth Biotechnologies
- Merck KGaA
- AGC Biologics
- Charles River Laboratories
- Oxford Biomedica
- Viralgen
- Minaris Advanced Therapies
- WuXi Advanced Therapies
- SK pharmteco
- Resilience
- uniQure
- Novartis Gene Therapies
Sources
Primary Research Interviews
Industry Stakeholders list
- CDMO executives
- Bioprocess development heads
- Viral vector manufacturing directors
- Quality assurance and quality control leaders
- Regulatory affairs specialists
- Technology transfer managers
End Users list
- Gene therapy developers
- Biopharmaceutical companies
- Academic research institutes
- Hospital based cell and gene therapy centers
- Clinical trial investigators
- Translational medicine laboratories
Government and International Databases
- U.S. Food and Drug Administration
- European Medicines Agency
- ClinicalTrials.gov
- World Health Organization
- OECD
Trade Publications
- BioProcess International
- Pharmaceutical Technology
- American Pharmaceutical Review
- GEN
- BioPharm International
- European Pharmaceutical Review
Academic Journals
- Molecular Therapy
- Molecular Therapy Methods and Clinical Development
- Human Gene Therapy
- Nature Biotechnology
- Molecular Therapy Oncolytics
- Cytotherapy
Reputable Newspapers
- Financial Times
- The Wall Street Journal
- The New York Times
- The Guardian
- Nikkei Asia
Industry Associations
- American Society of Gene and Cell Therapy
- Alliance for Regenerative Medicine
- Cell and Gene Therapy Catapult
- International Society for Cell and Gene Therapy
- BioIndustry Association
- International Society for Pharmaceutical Engineering
Public Domain Resources
- FDA guidance documents
- EMA scientific guidelines
- Clinical trial registries
- Patent databases
- Company annual reports
- Conference proceedings and abstracts
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 10 Years
Share
Share
About Author
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients