The acute vulvovaginal candidiasis treatment market size is anticipated to grow at a CAGR of 5.2% with USD 1,050 Mn in 2026 and is expected to reach USD 1,470 Mn in 2033. The market is growing with the increasing prevalence of vulvovaginal candidiasis (VVC) and the rising adoption of both oral and topical antifungal treatments. As per the CDC, approximately 75% of women are expected to experience at least one episode of VVC in their lifetime, thereby driving demand for effective treatment solutions
To learn more about this report, Request Free Sample
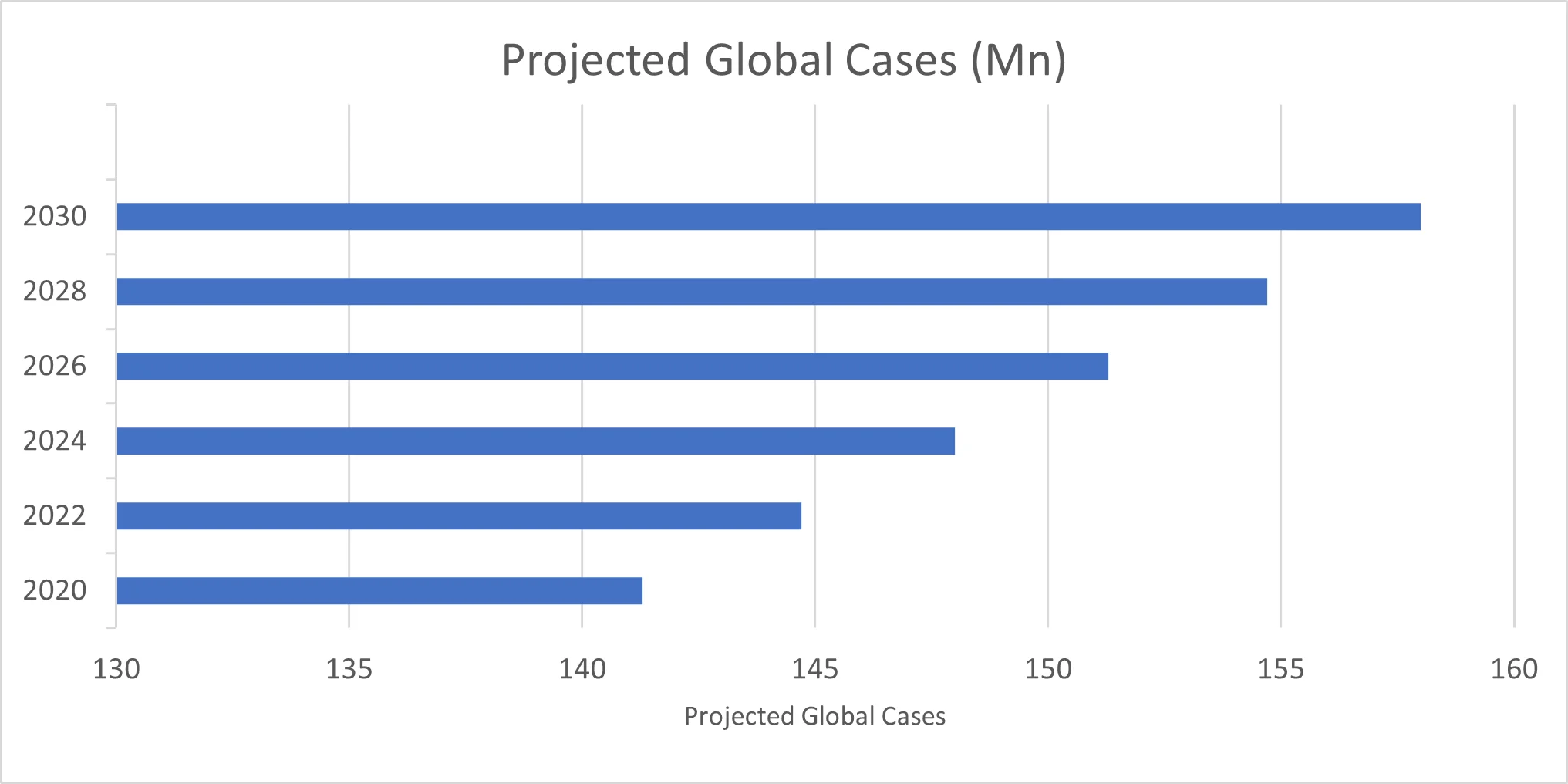
| Year |
Projected Global Cases (Millions) |
| 2020 |
141.3 |
|
2022 |
144.7 |
|
2024 |
148.0 |
|
2026 |
151.3 |
|
2028 |
154.7 |
|
2030 |
158.0 |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
U.S. demand is strengthening as acute vulvovaginal candidiasis remains a high-frequency women’s health condition and treatment expectations shift beyond traditional azole creams and single-dose fluconazole. The CDC states that an estimated 75% of women have at least one VVC episode, 40%–45% have two or more episodes, and 10%–20% develop complicated VVC, creating a large recurring treatment pool.
This shift is also supported by new FDA-approved oral options. BREXAFEMME (ibrexafungerp) is approved for treatment of VVC in adult and post-menarchal pediatric females, with a one-day 600 mg oral regimen, while FDA-approved VIVJOA (oteseconazole) lowered culture-verified acute VVC recurrence versus placebo through Week 48 in recurrent VVC trials.
The development pipeline is further improving market confidence. ClinicalTrials.gov lists a Phase 3 study evaluating boric acid inserts for VVC, while the completed CANDLE Phase 3 study evaluated oral ibrexafungerp for recurrent VVC prevention. This is making U.S. treatment adoption more innovation-led, recurrence-focused, as well as patient-convenient.
A key recent innovation in Acute Vulvovaginal Candidiasis Treatment is the approval and clinical use of ibrexafungerp, marketed as BREXAFEMME. It is among the important innovations because it introduced a new oral, non-azole antifungal mechanism for vaginal yeast infections, unlike traditional azole therapies like fluconazole, clotrimazole, or miconazole. The U.S. FDA listed BREXAFEMME as the first approved drug in a new antifungal class in more than 20 years for vulvovaginal candidiasis, thereby making it a major therapeutic breakthrough for the market.
Ibrexafungerp is a triterpenoid antifungal which works by inhibiting glucan synthase. It is an enzyme needed for fungal cell wall formation. This makes it different from azoles, which mainly target ergosterol synthesis. This advancement is especially relevant as CDC guidance still shows that most conventional uncomplicated VVC regimens are azole-based like the topical azoles and single-dose oral fluconazole.
For acute VVC, the current prescribing information states that BREXAFEMME is indicated for adult and post-menarchal pediatric females and is given as a one-day oral regimen, with 300 mg taken about 12 hours apart for a total 600 mg dose. This supports better treatment convenience compared with multi-day topical regimens, while also creating a premium oral alternative for patients where azole resistance, intolerance, drug interactions, or treatment failure may be a concern.
|
Current Event |
Description and its Impact |
|
First‑in‑Class Non‑Azole Oral Antifungal (ibrexafungerp) Description: |
|
|
CDC Treatment Guidance on Antifungal Strategy for VVC |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
On the basis of drug type, the fluconazole segment accounts for the largest acute vulvovaginal candidiasis treatment market share of 35.2% in 2026. The segment’s growth is because fluconazole remains a preferred oral azole option for uncomplicated VVC, supported by simple dosing, strong physician familiarity, and wide outpatient usability. CDC treatment guidelines list fluconazole 150 mg orally in a single dose as the recommended oral regimen for vulvovaginal candidiasis. Azole treatment is also reported to provide symptom relief and negative cultures in 80%–90% of patients who complete therapy.
In recurrent cases, fluconazole continues to hold strong clinical relevance because CDC recommends fluconazole every third day for three doses before maintenance therapy, followed by weekly oral fluconazole for six months as the indicated maintenance regimen for recurrent VVC. DailyMed also confirms the approved fluconazole dosage for vaginal candidiasis as 150 mg as a single oral dose.
The recent FDA-approved alternatives are expanding the treatment industry. In April 2022, the U.S. Food and Drug Administration (FDA) approved VIVJOA, an azole antifungal therapy developed to lower the incidence of recurrent vulvovaginal candidiasis (RVVC) in females with a history of RVVC who are not of reproductive potential. VIVJOA became the first and only FDA-approved treatment which was specifically indicated for this condition.
To learn more about this report, Request Free Sample
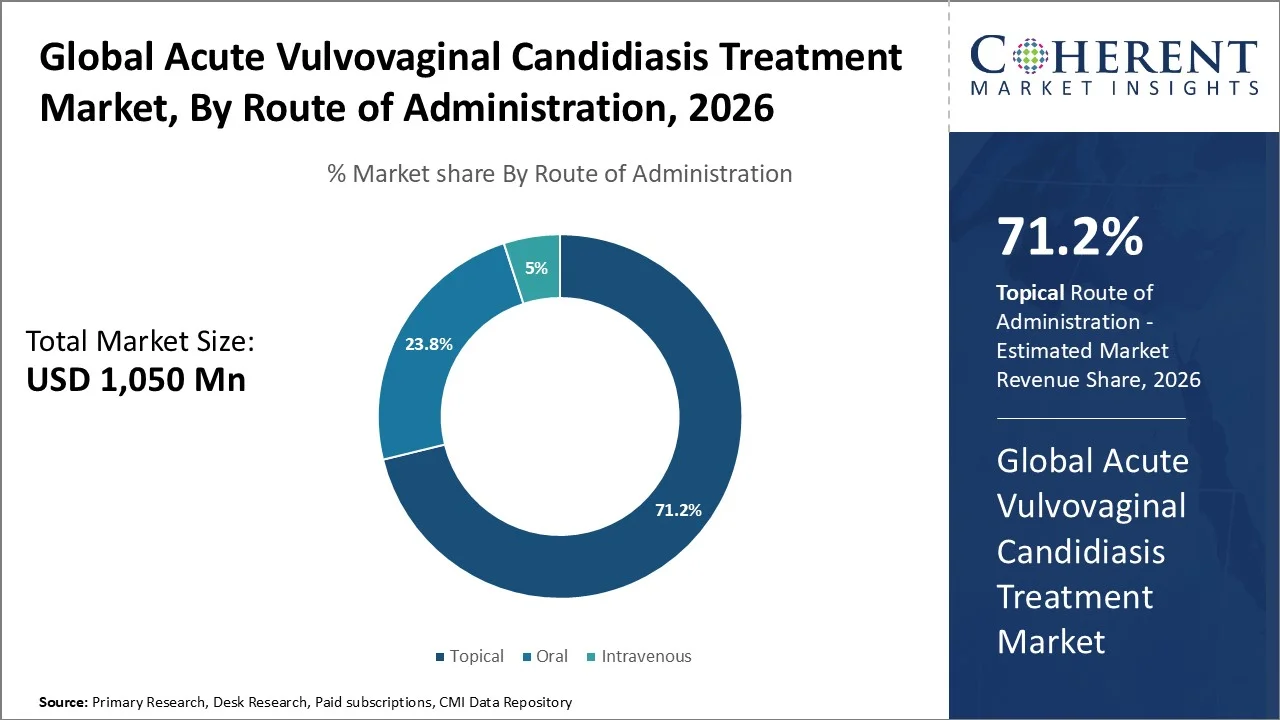
Topical segment accounts for the largest acute vulvovaginal candidiasis treatment market share of 71.2% in 2026, owing to widespread use of topical antifungals as first‑line, localized therapy for uncomplicated VVC. Short‑course topical formulations, including intravaginal azole creams and suppositories, are recommended by treatment guidelines and offer high local drug concentration, symptom relief, and reduced systemic exposure compared with oral therapies.
Topical regimens (singleIn clinical practice, topical options such as clotrimazole, miconazole, and tioconazole intravaginal therapies remain standard therapy for mild to moderate acute episodes due to ease of use and favorable tolerability.

To learn more about this report, Request Free Sample
The North America region accounts for 44.20% of the market in 2026. The region’s growth is backed by the region’s robust healthcare infrastructure, increased awareness, and high prevalence of VVC cases. The substantial patient base is fueling the demand for effective treatments, particularly oral and topical anti fungal drugs.
The recent regulatory progress has diversified treatment options. The U.S. Food and Drug Administration (FDA) approved ibrexafungerp (Brexafemme) in June 2021 as the first oral non In addition, the oteseconazole (Vivjoa) was approved in April 2022 for recurrent VVC in select populations and is under late
The top pharmaceutical players influencing this space are Scynexis/GSK (Brexafemme), Mycovia Pharmaceuticals (Vivjoa), and traditional antifungal providers like Pfizer and Teva Pharmaceuticals, which supply generic and branded azoles used in acute VVC management. These developments reflect evolving treatment patterns, with newer therapies complementing established agents in North America’s acute VVC therapeutic landscape.
The Asia Pacific region is poised to be as the fastest-growing region through 2026-2033. The government and public‑health data specific to acute vulvovaginal candidiasis (VVC) are limited in the region but available epidemiologic and clinical trial evidence suggests a substantial treatment burden and evolving therapeutic landscape. A systematic review of Southeast Asian women estimated a pooled VVC prevalence of ~23% (95% CI 18Local studies also report prevalence ranging from 10–35% in India among women of reproductive age, underscoring the clinical relevance in South Asia.
While worldwide regulatory approval of ibrexafungerp (BREXAFEMME®) occurred in June 2021, Asia Pacific regulatory statuses vary and localized approvals or submission activity are ongoing. According to the World Health Organization’s 2025 fungal disease fact sheet, candidiasis remains a common yeast infection requiring antifungal therapy, with concerns about antifungal resistance and gaps in diagnostic capacity across low‑ and middle‑income settings including South‑East Asia and Western Pacific Regions.
The United States Acute Vulvovaginal Candidiasis Treatment Market is witnessing steady growth. In the U.S., acute vulvovaginal candidiasis (VVC) remains one of the most prevalent gynecologic infections, affecting >50% of women over their lifetime, with many experiencing ≥2 episodes requiring treatment with short
The growth in the treatment market is underpinned by epidemiological drivers such as increasing rates of diabetes and antibiotic use, both recognized risk factors elevating candidiasis susceptibility as well as heightened healthcare‑seeking behavior among symptomatic women. In addition, greater patient awareness and provider diagnosis contribute to consistent prescription and over‑the‑counter antifungal utilization, thereby sustaining sustained demand for effective VVC therapies.
The China Acute Vulvovaginal Candidiasis Treatment Market is poised for significant growth, driven by the increasing prevalence of fungal infections among women and the expanding availability of advanced antifungal therapies. The National Medical Products Administration (NMPA) approved Oteseconazole (Vivjoa) capsules in June 2023 for severe VVC, where Phase 3 data showed therapeutic cure rates of 66.9 % vs. 45.9 % with fluconazole at Day 28 (p = 0.0002). A phase III trial (CTR20220918) evaluating oral ibrexafungerp in Chinese women reported 51.0 % clinical cure vs. 25.6 % placebo, with significant mycological eradication (55.6 % vs. 18.2 %). China’s 2024 clinical practice updates emphasize diagnostic specificity and resistance monitoring per national guideline revisions.
Some of the major key players in Acute Vulvovaginal Candidiasis Treatment are Mycovia Pharmaceuticals, Inc., Scynexis, Inc., Basilea Pharmaceutica Ltd., Astellas Pharma Inc., Grupo Ferrer Internacional, S.A., Pacgen Life Science Corporation, NovaDigm Therapeutics, Inc., Cidara Therapeutics, Inc., Amplyx Pharmaceuticals Inc., and Pfizer, Inc.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,050 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.2% | 2033 Value Projection: | USD 1,470 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Mycovia Pharmaceuticals, Inc., Scynexis, Inc., Basilea Pharmaceutica Ltd., Astellas Pharma Inc., Grupo Ferrer Internacional, S.A., Pacgen Life Science Corporation, NovaDigm Therapeutics, Inc., Cidara Therapeutics, Inc., Amplyx Pharmaceuticals Inc., and Pfizer, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients