Allergic Asthma Therapeutics Market is estimated to be valued at USD 7.95 Bn in 2026 and is expected to reach USD 9.91 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 3.2% from 2026 to 2033.
Analysts’ Views on Global Allergic Asthma Therapeutics Market:
Over the projected period, increased respiratory diseases, and strategic expansions by key players to strengthen its market position, are anticipated to drive the growth of the global allergic asthma therapeutics market.diagnosticand medical device technologies, announced that it has entered into an agreement with Ontario Inc., operating as August Therapeutics, Inc., a biotechnology company and Ketiko Bio Corp, a global pharmaceutical company to acquire a 25% interest in each of InStatin, Inc.
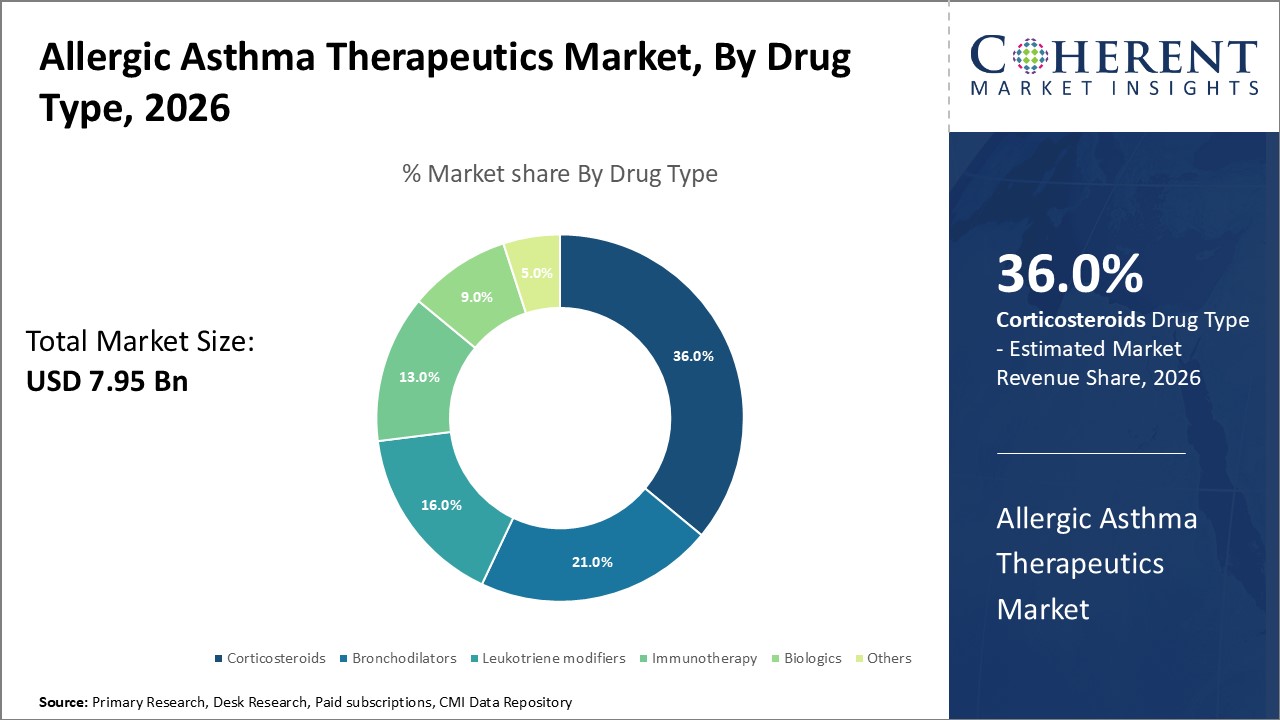
Figure 1. Global Allergic Asthma Therapeutics Market Share (%), By Drug Type, 2026
To learn more about this report, Request Free Sample
Global Allergic Asthma Therapeutics Market– Drivers
High prevalence of respiratory diseases
Increasing prevalence of respiratory diseases is expected to the growth of global allergic asthma therapeutics market over the forecast period. For instance, in June 2020, according to the Global Asthma report 2022, the Global Burden of Disease Study (GBD) estimated that in 2020, there were 262 million people affected by asthma, equating to an age-standardized rate of 3,416 cases per 100,000 population.
Increasing awareness about the asthma treatment
Increasing awareness about the asthma treatment is expected to drive the market over the forecast period. For instance, on May 02 2023, Alkem Labs, an India-based multinational pharmaceutical company, has announced the launch of its awareness campaign #RelieverFreeIndia to raise awareness about asthma among physicians and asthma patients. The company statement informed, “This campaign aims to spread awareness of the hazards of over-usage of reliever medications and substitute it with SMART therapy i.e., needing only one inhaler for both ‘daily’ and ‘reliever’ asthma treatment.”
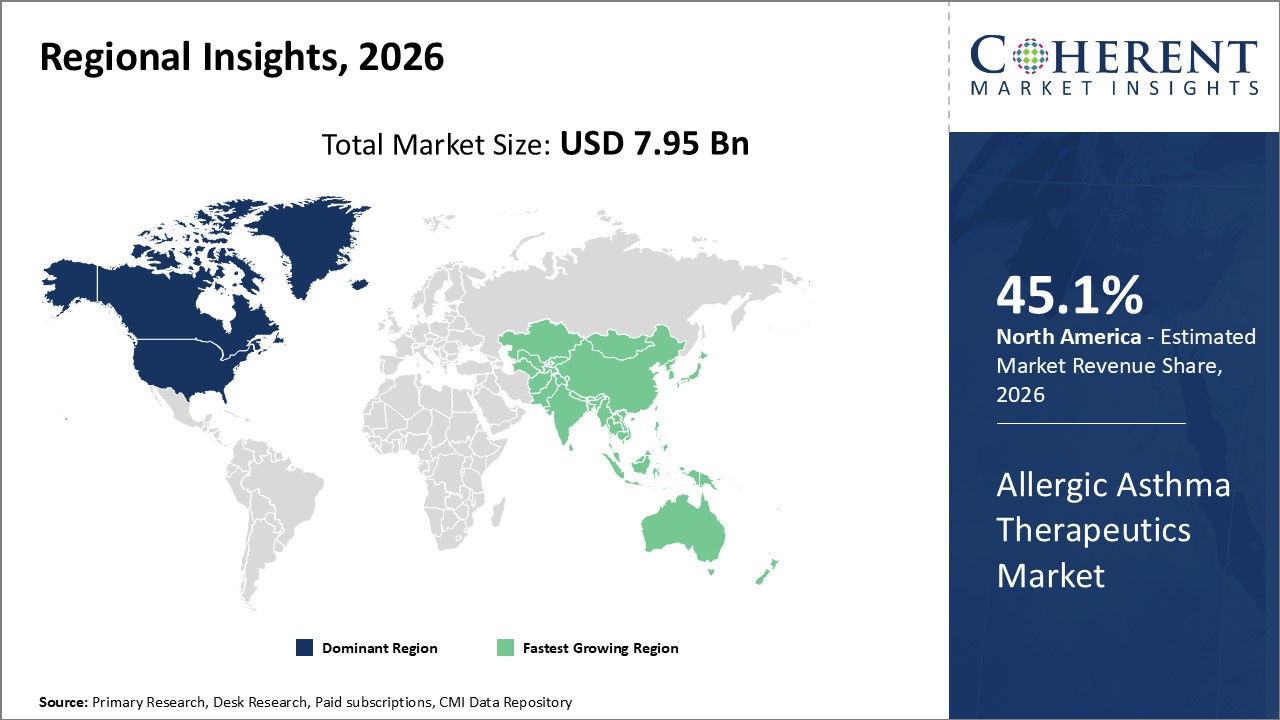
Figure 2. Global Allergic Asthma Therapeutics Market Share (%), By Region, 2026
To learn more about this report, Request Free Sample
Global Allergic Asthma Therapeutics Market- Regional Analysis
Among region, North America is estimated to hold a dominant position in the global allergic asthma therapeutics market over the forecast period owing to the increase in chronic respiratory diseases. For instance, on February 02, 2026, the Centers for Disease Control and Prevention (CDC), estimated that there are 142,342 deaths for every 100,000 people in 2021 in 2021 in the U.S.
respiratoryallergies, including allergic rhinitis (AR) and sinusitis. These increase the hospitalization rates due to the increasing prevalence of chronic respiratory diseases.
Allergic Asthma Therapeutics Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 7.95 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 3.2% | 2033 Value Projection: | USD 9.91 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
ASLAN Pharmaceuticals, Takeda Pharmaceutical Company Limited, CHIESI Farmaceutici S.p.A., CSL, NIOX, Fountain Therapeutics, Eli Lilly and Company, GSK plc., Infinity Pharmaceuticals, Inc., Mabtech, Kineta Inc., Marinomed Biotech AG, Mycenax Biotech Inc., AstraZeneca, and Panacea Biotec. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Allergic Asthma Therapeutics Market- Segmentation
The global allergic asthma therapeutics market report is segmented into drug type, route of administration, and region.
Among Drug Type, the global allergic asthma therapeutics market is segmented into corticosteroids, bronchodilators, leukotriene modifiers, immunotherapy, biologics, and others. Out of which, the corticosteroids is expected to dominate the global allergic asthma therapeutics market during the forecast period and this is due to the action of it directly on the airways and have fewer side effects.
Among Route of Administration, the global allergic asthma therapeutics market is segmented into oral, inhaled, and others. The inhaled segment is expected to dominate the market over the forecast period and this is due to the low side effects as compared to the oral route and faster onset of action.
Among Region, the global allergic asthma therapeutics market is segmented into North America, Latin America, Europe, Asia Pacific, Middle East, and Africa. Out of which, the North America is expected to dominate the market over the forecast period owing to the increase in launch of product by market key players.
Among all segmentation, the route of administration segment has the highest potential to dominate over the forecast period due to the low side effects as compared to the oral route and faster onset of action. For instance, in May 2020, ASLAN Pharmaceuticals, a Singapore-based clinical-stage immunology and oncology-focused biopharma company, amended its license agreement with CSL Limited (CSL). ASLAN’s aim behind this agreement was to have full global rights to develop, manufacture, and commercialize ‘ASLAN004’ in all indications. ASLAN004 is a fully human monoclonal antibody that helps in triggering the symptoms of allergic asthma such as coughing, wheezing, exacerbations of the disease, and shortness of breath.
Global Allergic Asthma Therapeutics Market- Cross Sectional Analysis
Among drug type, the corticosteroid segment held a dominant position in North America region over the forecast period due to increasing number of respiratory diseases which lead to the growth of allergies. For instance, in 2021, the Asthma and Allergy Foundation of America (AAFA), reported that each year in the U.S, anaphylaxis (a severe allergic reaction) to food results in 90,000 emergency room visits, and more than 50 million people in the U.S., experience various types of allergies each year. Asthma and Allergy Foundation of America (AAFA) 2021 data also reported that approximately 24 million people in the U.S. were diagnosed with seasonal allergic rhinitis. This equals around 9% (19.2 million) of adults and 8% (5.2 million) of children, and about 32 million people have food allergies. Thus, the growing prevalence of allergies is expected to increase demand for the treatment, thereby boosting the market's growth in North America.
Global Allergic Asthma Therapeutics Market- Key Developments
In March 2022, the U.S FDA, announced the approval of general drug-device combination product approval of the first generic for one of the most commonly prescribed complex drug-device combination products to treat asthma and (Chronic obstructive pulmonary disease) COPD is another step forward in our commitment to bring generic copies of complex drugs to the market, which e improve quality of life and help reduce the cost of treatment.
On April 18 2023, GSK plc., a global biopharma company and BELLUS Health Inc., a drug development company, announced that they have entered into an agreement under which GSK will acquire BELLUS, a Canada-based, late-stage biopharmaceutical company, working to better the lives of patients suffering from refractory chronic cough (RCC) for US$ 14.75 per share of common stock in cash representing an approximate total equity value of US$ 2 billion. The acquisition provides GSK, plc., access to camlipixant, a potential best-in-class and highly selective P2X3 antagonist currently in phase III development for the first-line treatment of adult patients with RCC.
In December 2021, AstraZeneca, a global biopharmaceutical company developing innovative medicines and Amgen, Inc., a multinational biopharmaceutical company Tezspire (tezepelumab-ekko) has been approved in the U.S. for the add-on maintenance treatment of adult and pediatric patients aged 12 years and older with severe asthma. Tezspire was approved following a Priority Review by the U.S. Food and Drug Administration (FDA) and based on results from the PATHFINDER clinical trial programme. The application included results from the pivotal NAVIGATOR Phase III trial in which Tezspire demonstrated superiority across every primary and key secondary endpoint in patients with severe asthma, compared to placebo, when added to standard therapy.
In February 2021, CHIESI Farmaceutici S.p.A., an international research-focused healthcare group, announced that the European Commission has granted the marketing authorisation for Trimbow (beclometasone/formoterol/glycopyrronium), an extrafine formulation triple fixed combination therapy in a single inhaler, as a maintenance treatment for adult asthma patients not adequately controlled with a maintenance combination of a long-acting beta2-agonist (LABA) and medium dose of inhaled corticosteroid (ICS), who experienced one or more asthma exacerbations in the previous year
Global Allergic Asthma Therapeutics Market- Key Trends
Research and Development in biologics is expected to grow over the forecast period
The research and development in the field of biologics is expected to grow over the forecast period. For instance, in July 2021, AstraZeneca, a global biopharmaceutical company developing innovative medicines, Biologics License Application (BLA) for tezepelumab has been accepted and granted Priority Review for the treatment of asthma from the U.S. Food and Drug Administration (FDA). Tezepelumab is being developed by AstraZeneca in collaboration with Amgen. The U.S. FDA grants Priority Review to applications for medicines that offer significant advantages over available options by demonstrating safety or efficacy improvements, preventing serious conditions, or enhancing patient compliance
Global Allergic Asthma Therapeutics Market: Restraints
High cost of asthma treatment
Certain factors such as high cost of asthma treatment is expected to restrict the growth of global allergic asthma therapeutics market over the forecast period. For instance, in January 2020, according to the Asthma and Allergy Foundation of America, the yearly cost of asthma in the U.S is around US$ 56 billion, and each year, asthma accounts for more than 439,000 hospitalizations, 1.7 million emergency department (ED) visits, and 13.8 million missed school days. So, the use of orals drugs to be employed for the treatment as compared inhalers due to its low cost.
Global Allergic Asthma Therapeutics Market- Key Players
Major players operating in the global allergic asthma therapeutics market include ASLAN Pharmaceuticals, Takeda Pharmaceutical Company Limited, CHIESI Farmaceutici S.p.A., CSL, NIOX, Fountain Therapeutics, Eli Lilly and Company, GSK plc., Infinity Pharmaceuticals, Inc., Mabtech, Kineta Inc., Marinomed Biotech AG, Mycenax Biotech Inc., AstraZeneca, and Panacea Biotec.
*Definition: When an allergen is inhaled, it causes the airways to constrict, causing allergic asthma. Common allergens include pollen, dander and mold spores. The cause of allergic asthma is complex. It is likely that a combination of multiple genetic and environmental factors contribute to development of the condition. Doctors believe genes are involved because having a family member with allergic asthma or another allergic disorder increases a person's risk of developing asthma.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients