The global digital biomarker market is expected to grow from USD 4,850 Mn in 2026 to USD 16,900 Mn by 2033, registering a compound annual growth rate (CAGR) of 19.5%. The global digital biomarker market is poised for significant expansion fueled by growing government emphasis on digital health care and remote patient monitoring for addressing the growing burden of chronic diseases.
According to the World Health Organization, noncommunicable diseases (NCDs), including heart disease, stroke, cancer, diabetes, and chronic lung disease, are collectively responsible for 74% of all deaths worldwide. More than three-quarters of all NCD deaths, and 86% of the 17 million people who died prematurely, or before reaching 70 years of age, occur in low- and middle-income countries.
(Source: World Health Organization)
Monitoring biomarkers are projected to hold a market share of 46.0% in 2026, due to their ability to provide continuous and real-time tracking of both physiological and behavioral data. This is evidenced by numerous case studies conducted by various organizations, in which data collection was achieved through the use of wearable devices. For instance, the National Institutes of Health (NIH) supported the All of Us Research Program which involves the collection of continuous data on activity and sleep, among other health parameters, from hundreds of thousands of people for the purpose of disease development research. (Source: Vibrent Health)
Moreover, a study published in September 2023 in National Library of Medicine demonstrates that wearable biosensors are capable of providing continuous real-time tracking of biomarkers such as glucose, lactate, and physiological data (Source: National Library of Medicine). Additionally, the National Cancer Institute points out that new developments in wearable sensors have made it possible to measure "the quantity, timing, and quality of sleep, light exposure, and physical activity," thereby facilitating prolonged assessment of behaviors and exposures in health studies (Source: National Cancer Institute). The robustness of the body of clinical and governmental evidence available is an indication of the increased usefulness of continuous monitoring, which makes monitoring biomarkers the most dominant segment within the digital biomarker market.
To learn more about this report, Request Free Sample
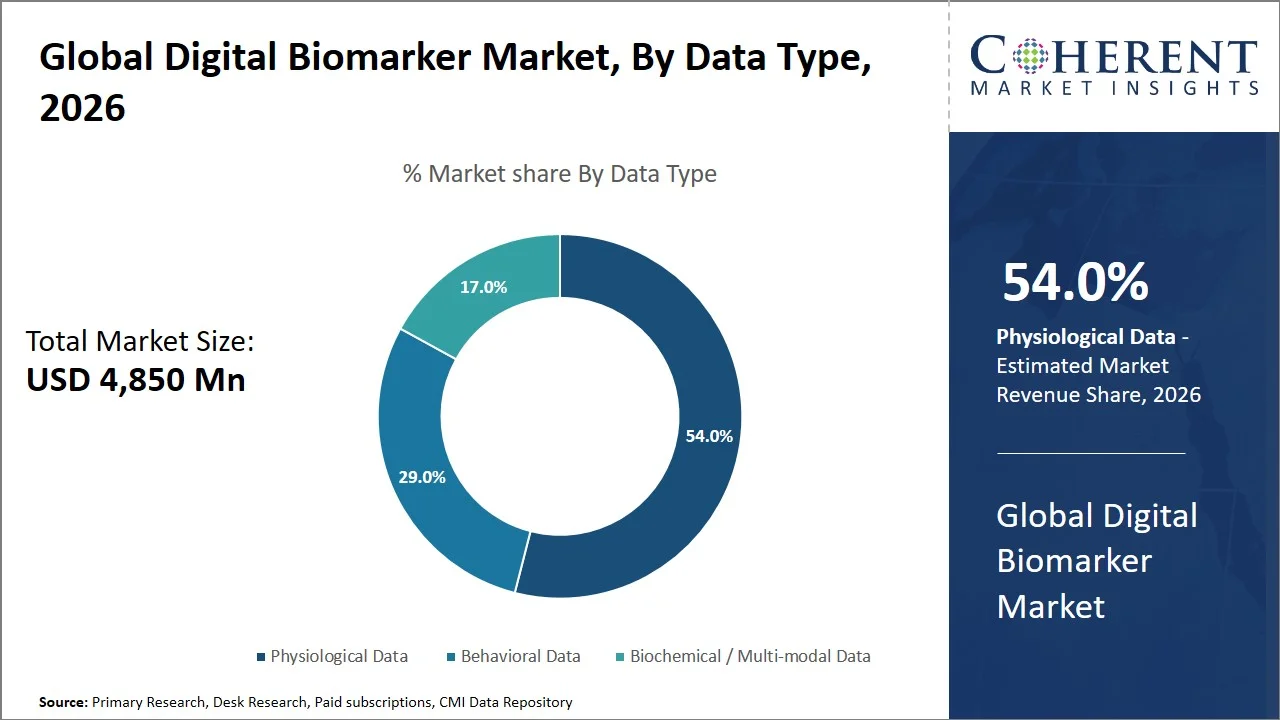
Physiological data is projected to hold a market share of 54.0% in 2026, since it captures objective and quantifiable measures of important functions of the body including heart rate, blood pressure, oxygen saturation, and physical activity. Behavioral and self-reporting data types lack objectivity and therefore cannot be used to accurately monitor disease progression and diagnose patients. Physiological measures are continually captured using validated medical-grade and consumer-grade wearable technology and can be utilized for clinical and research purposes reliably compared to other data types.
Regulatory and public health agencies encourage the use of physiological data gathered from wearable devices in their activities. For instance, the Centers for Medicare & Medicaid Services endorses the use of remotely captured physiological data using wearables such as heart rate and blood pressure measurement in clinical decisions and payment decisions (Source: Centers for Medicare & Medicaid Services). Moreover, other agencies such as the National Institutes of Health (NIH) encourages the collection of physiological measures using wearable devices continuously in major research programs aimed at studying disease progression and understanding disease trends in populations. (Source: National Institutes of Health)
The wearable devices segment is projected to hold a market share of 51.0% in 2026, due to its higher degree of accessibility, passive data collection, and seamless integration into the lifestyle of individuals. This dominance has been further bolstered by their growing adoption in large-scale health studies; for instance, the UK Biobank has utilized wearable physical activity trackers on more than 100,000 individuals in order to gain objective health data on a large scale. (Source: UK Biobank)
Moreover, the World Health Organization recognizes the importance of digital technology and its applications such as wearables in building up people-centered and evidence-based health care systems (Source: World Health Organization). This has made wearable devices the ideal choice when it comes to the deployment of digital biomarkers in both research and health care environments due to their scalability and frequency of data collection.
|
Current Events |
Description and its Impact |
|
EU European Health Data Space (EHDS) Regulation Advancement (2025) |
|
|
U.S. FDA Guidance on Digital Health Technologies in Clinical Investigations (2023–ongoing adoption) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: European Commission, U.S. Food and Drug Administration)
To learn more about this report, Request Free Sample
To learn more about this report, Request Free Sample
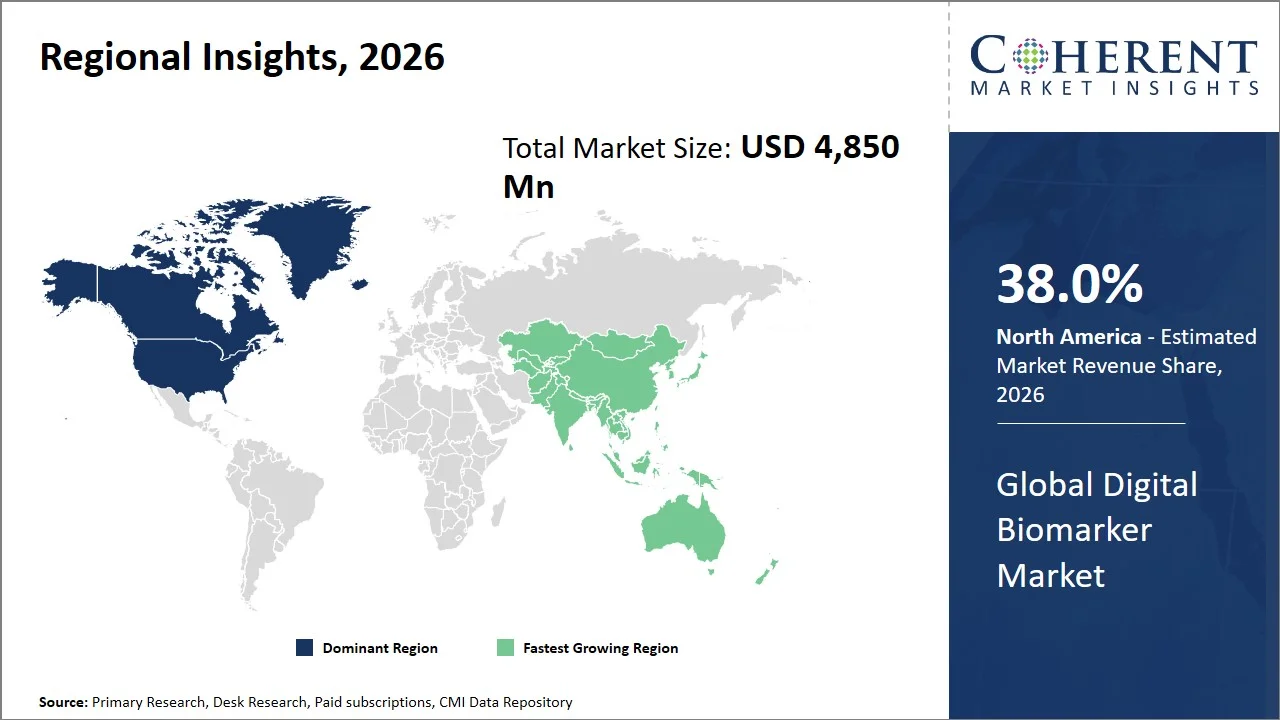
North America leads the global digital biomarker market, accounting for an estimated 38.0% share in 2026, owing to its well-established infrastructure for conducting clinical research along with an early adopter attitude toward healthcare based on data-driven approaches that allows for the swift deployment of digital health solutions. The region is known for taking part in major scientific studies; for instance, the National Institute of Health (NIH) heads up a program called All of Us Research Program that involves collecting health data on over one million individuals to support the development of personalized medicine treatments. (Source: National Institute of Health)
Furthermore, as reported by the Centers for Disease Control and Prevention, chronic diseases continue to be widespread in the U.S. and a considerable number of people require prolonged supervision. Moreover, 90% of the nation's USD 4.9 trillion in annual health care expenditures are for people with chronic and mental health conditions (Source: Centers for Disease Control and Prevention). This convergence of a solid research framework, huge patient databases, and high prevalence of the disease makes for an ideal environment for digital biomarker to be deployed quickly and effectively.
The Asia Pacific digital biomarker market is expected to exhibit the fastest growth with an estimated contribution of 24.0% share to the market in 2026, owing to its rapid build-up of digital infrastructure and a highly varied base of patients. At the same time, the governments in the region are keen on introducing technology-based solutions for healthcare delivery through their healthcare digitization initiatives.
Countries like India have plans to set up an ecosystem of digital health solutions at the national level. For example, the Indian National Health Authority has launched the Ayushman Bharat Digital Mission that involves building a complete ecosystem that allows the creation of big health data and its interoperability (Source: National Health Authority). This confluence of digitally-enabled policies, rising health needs, and higher rates of device connectivity has led to an uptick in the use of digital biomarkers, thereby positioning Asia Pacific as the fastest-growing regional market for digital biomarkers.
The U.S. is the leader in innovations and implementations within the digital biomarker market, due to the country's significant capabilities of translating innovations from the laboratory into clinical settings and federal support of digital health innovations. For instance, the National Institute of Health (NIH) is engaged in launching the Bridge2AI project, which targets creating high-quality biomedical datasets to develop artificial intelligence technologies in healthcare, particularly by utilizing digital biomarkers. (Source: National Institute of Health)
Moreover, the Advanced Research Projects Agency for Health works on promoting health technologies through conducting innovative projects that seek to address various health issues with the help of advanced data analysis techniques (Source: Advanced Research Projects Agency for Health). The U.S., with its strong focus on integrating artificial intelligence, federally-funded innovation programs (e.g., ARPA-H programs such as Platform Accelerating Rural Access to Distributed & Integrated Medical Care (PARADIGM), Scalable Solutions for Healthcare Delivery (SSHD), and Precision Surgical Interventions (PSI)), and quickly translating research to practice, is well-positioned to be one of the leading nations in developing digital biomarkers and their widespread use.
The U.K. represents an ideal environment for the digital biomarker market due to the presence of a robust public healthcare system and a digital health uptake framework. The National Health Service allows the deployment of digital solutions in a centralized manner, thus facilitating real-world data collection and verification. Additionally, the National Institute for Health and Care Excellence has put forward the Evidence Standards Framework for Digital Health Technologies, which clearly defines standards on the assessment of digital products, including digital biomarker generators (Source: National Institute for Health and Care Excellence). Clinical research facilities, along with the availability of longitudinal data from patients, add to the value of the U.K. digital biomarker market in relation to digital biomarkers.
China stands out as one of the most important countries with respect to the future growth of the digital biomarker market owing to an increased adoption of digital technologies in the healthcare sector, as well as wide use of smart health devices. The policies initiated by the National Health Commission of the People’s Republic of China to promote “Internet + Healthcare” aim at embedding digital technologies into the healthcare system and facilitating remote monitoring. (Source: National Health Commission of the People’s Republic of China)
Additionally, the National Medical Products Administration is enhancing the regulation of digital health and Software as a Medical Device (SaMD) product to facilitate their approval and commercial availability. (Source: China Med Device) China's large population and rising number of people with chronic diseases (over 300 million people in China are diagnosed with chronic diseases, accounting for ~86.6% of total deaths) make the need for continuous monitoring solutions even greater. The country also benefits from the widespread use of smartphones and wearables, which makes it easier to collect large amounts of real-world data. This combination of policy support, regulatory changes, and growth in digital infrastructure makes China a promising market for digital biomarker expansion.
Germany emerges as a leader in the Europe digital biomarker industry owing to the availability of a well-established system for reimbursement of digital health services along with regulations that facilitate smooth adoption of digital therapeutics and monitoring systems (such as Digital Healthcare Act – DVG; Digital Health Applications Ordinance – DiGAV). The country has the approval and reimbursement system of digital health applications (DiGAs), managed by the Federal Institute for Drugs and Medical Devices. This allows clinically proven digital technology to be prescribed by physicians and also makes the reimbursement process possible through statutory health insurance plans. (Source: Federal Institute for Drugs and Medical Devices)
Furthermore, Germany has an advanced healthcare environment along with high penetration of electronic medical records, thereby enabling the generation of large amounts of digital data. Besides this, the country also has an active clinical research environment that can help in validating digital biomarkers.
India is witnessing notable progress in the digital biomarker market, driven by advancements in the regulatory framework, expansion of clinical research infrastructure, and increasing integration of digital technologies in healthcare. Medical devices available in the country are regulated by Central Drugs Standard Control Organization through Medical Device Rules, 2017, with a proper structure to authorize software-enabled and interconnected devices.
Additionally, ethical guidelines formulated by the Indian Council of Medical Research have also been provided to ensure that ethical considerations are kept in mind while conducting biomedical and healthcare research, including the use of digital methods for capturing real-world data from the patients in clinical trials. India is also known for its burgeoning landscape for conducting clinical trials and more use of digital tools for diagnosis and monitoring patients. All of this implies that the country has the potential to become a developing market for digital biomarkers. For instance, digital biomarker-powered remote monitoring technology was extensively used during the pandemic via interventions like the Aarogya Setu, where data from real-time monitoring could be collected in massive quantities, thus proving the potential of digital technologies in health surveillance at scale.
|
Region |
Key Regulatory Bodies |
Key Regulations |
Regulatory Focus |
Impact on Market |
|
North America |
U.S. Food and Drug Administration, Health Canada |
FDA Digital Health Policies, Software as a Medical Device (SaMD), Health Canada SaMD Guidance |
Software validation, real-world evidence, device classification, reimbursement frameworks |
Mature regulatory ecosystem with clear pathways accelerates commercialization and clinical adoption of digital biomarkers |
|
Europe |
European Medicines Agency, European Commission |
Medical Device Regulation (MDR 2017/745), General Data Protection Regulation (GDPR) |
Data privacy, cybersecurity, clinical validation, cross-border data interoperability |
Strong data protection laws enhance trust and enable secure deployment of digital biomarkers at scale |
|
Asia Pacific |
Pharmaceuticals and Medical Devices Agency, National Medical Products Administration, Ministry of Health and Family Welfare |
SaMD guidelines, national digital health missions (e.g., ABDM), evolving device regulations |
Digital health infrastructure development, regulatory harmonization, real-world data usage |
Rapidly evolving regulatory landscape supports large-scale adoption driven by government-led digital health initiatives |
|
Latin America |
National Health Surveillance Agency, Federal Commission for the Protection against Sanitary Risk |
Medical device regulations, emerging digital health guidelines |
Device approval processes, regulatory modernization, telehealth expansion |
Gradual regulatory development is improving market access, though variability across countries may slow adoption |
|
Middle East & Africa |
Saudi Food and Drug Authority, Department of Health, South African Health Products Regulatory Authority |
National digital health strategies, medical device regulations, telemedicine frameworks |
Digital transformation, healthcare accessibility, regulatory capacity building |
Increasing government investments in digital health are creating new opportunities, though regulatory frameworks are still maturing |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The adoption of decentralized clinical trials (DCTs) and personalized medicine approaches are presenting a number of growth opportunities in the digital biomarker market by enabling continuous and objective data generation using real-world evidence. Digital biomarkers make it possible for patients to be continuously monitored remotely by way of the use of wearables and other connected devices. The above-mentioned changes in the clinical trials industry have been encouraged by regulatory authorities; for example, the U.S. Food and Drug Administration has published recommendations for the incorporation of digital technology into decentralized clinical trials (DCTs) in order to enable the collection and analysis of remote data. (Source: U.S. Food and Drug Administration)
Moreover, European Medicines Agency framework highlights that real-world evidence is increasingly used to study treatment effectiveness, safety, and clinical management in real-world settings, which is critical for personalized medicine approaches and decentralized trials (Source: European Medicines Agency). As a result, there is an acceleration of digital biomarker adoption as essential instruments that make it possible to collect high-frequency patient data.

To learn more about this report, Request Free Sample
In the global digital biomarker market, the competitive nature is expected to persist as there has been significant development within digital health technologies, rising demand for real-time monitoring of patients, and the application of artificial intelligence within healthcare analytics. The organizations that have an established knowledge base in areas such as analytics, wearables, and clinical validation are able to leverage their advantages effectively. Key focus areas include:
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 4,850 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 19.5% | 2033 Value Projection: | USD 16,900 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
AliveCor Inc., BioSensics, Evidation Health, Empatica Inc., Koneksa Health, Huma Therapeutics, Altoida AG, IXICO plc, Oura Health, and Verily Life Sciences |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients