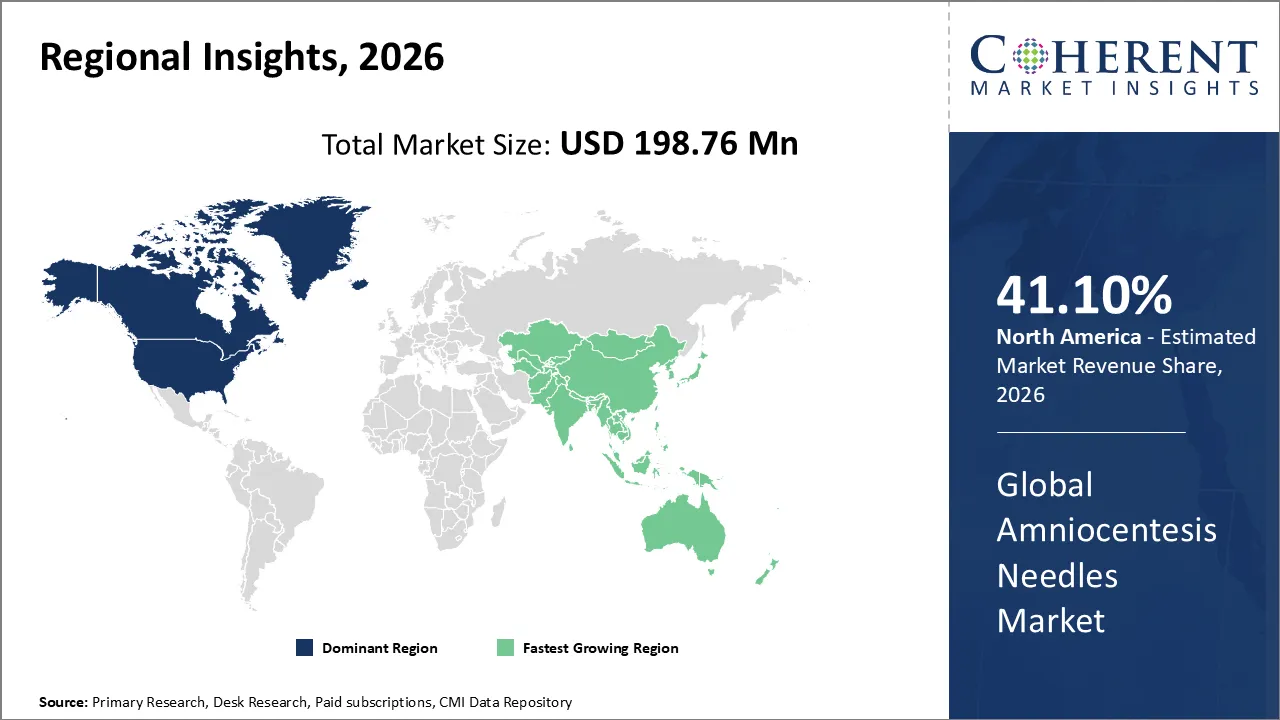
The Amniocentesis Needles Market is estimated to be valued at USD 198.76 Mn in 2026 and is expected to reach USD 220.78 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 7.86% from 2026 to 2033.
The global market for amniocentesis needles is expanding because more healthcare practitioners are doing prenatal diagnostic procedures and raising awareness of fetal health. Hospitals and maternal-fetal care facilities actively boost demand by using advanced healthcare infrastructure and qualified professionals. To make procedures safer and more efficient, manufacturers come up with new ways to construct needles and combine them with imaging equipment. The market grows even more as doctors deal with high-risk pregnancies, genetic diseases, and do therapeutic procedures like fetal blood transfusions. North America and Europe are the most advanced adopters, but other areas provide new chances.
|
Current Events |
Description and its impact |
|
Geopolitical Developments Affecting Medical Device Trade |
|
|
Technological Innovations and Advancements |
|
|
Economic Factors Influencing Healthcare Spending |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
100mm to 150mm hold the largest market share of 38.9% in 2026. Clinicians are increasingly driving the demand for 100 mm to 150 mm amniocentesis needles since they are flexible and work well for a variety of prenatal procedures. They like this length because it gives them the best balance of reach and control, which makes sure it works for all types of maternal anatomies. The usage of these tests is also increasing because prenatal screening programs are becoming more common and early genetic tests are being given more attention. Hospitals and diagnostic centers actively use this size because it is easy to use, works with ultrasound, and is always reliable in both routine and high-risk procedures. This makes it a standard clinical choice.
Fetal Blood Transfusion expected to hold largest market share of 43.6% in 2026. The amniocentesis needles market is growing because more and more doctors are finding fetal blood abnormalities during prenatal treatment. They employ these methods to treat problems like fetal anemia and stop problems from happening before birth. Advanced needle designs and real-time imaging make interventions more precise and safer, which encourages more doctors to use them. Hospitals and specialist maternal-fetal clinics actively administer these medicines, and as more people learn about the benefits of in-utero treatment, the demand for needles made just for therapeutic fetal blood transfusions is rising. For instance, in August 2025, Fayette County Fire and Emergency Services launched its Pre-Hospital Blood Transfusion Program.
Hospitals acquired the prominent market share of 42.2% in 2026. Hospitals are a big part of the amniocentesis needles market since they handle a lot of patients and give full prenatal care. Obstetrics and maternal-fetal medicine teams do both routine and high-risk procedures, which keeps the demand for trustworthy needles high. They can do procedures safely and quickly because they have skilled specialists, advanced imaging technology, and clean rooms. Hospitals also make sure they always have enough needles by streamlining their purchasing processes and building long-term relationships with suppliers. As referral facilities for complicated pregnancies, they use new needle designs to make the procedures go better.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 41.1% in 2026. Healthcare professionals are having a big impact on the North American amniocentesis needles market by using new prenatal diagnostic methods and raising awareness about fetal health. They are using high-precision needles and imaging technology more and more to make procedures safer and more accurate. Hospitals and maternal-fetal clinics help people adopt by providing well-established infrastructure and qualified professionals. The market is also getting busier because more people want genetic testing, help with high-risk pregnancies, and therapeutic operations like fetal blood transfusions. At the same time, producers in different parts of the world are coming up with new ways to make needles work better and give doctors more confidence.
By making advanced diagnostic services more widely available, hospitals and prenatal care centers are helping the Asia Pacific amniocentesis needles market grow. More and more doctors are doing amniocentesis procedures because people are becoming more conscious of fetal health and prenatal screening programs are becoming more common. Better healthcare infrastructure, better-trained specialists, and the use of imaging technologies make treatments safer and more efficient. The growing number of high-risk pregnancies and genetic testing programs is driving up demand even more. At the same time, local producers and distributors are making cheap, high-quality needles to help more people in developing nations use them.
Hospitals and maternal-fetal medical facilities are helping the US amniocentesis needles market develop by doing advanced prenatal tests. More and more, doctors are using precision needles with real-time imaging to do treatments safely and correctly. More people are learning about genetic diseases and high-risk pregnancies, which makes routine adoption more likely. Therapeutic interventions like fetal blood transfusions also increase demand. A well-established healthcare system, trained specialists, and a large network of hospitals make sure that needles are used consistently. Domestic producers are also always coming up with new ways to make needles safer, better quality, and more trustworthy for doctors across the country.
Hospitals and prenatal care centers are helping the China amniocentesis needles market grow by making it easier for people to get advanced fetal diagnostic services. More and more doctors are doing amniocentesis because people are becoming more conscious of prenatal health and the government is backing screening initiatives. Better healthcare infrastructure, more trained obstetricians, and the use of imaging-guided techniques make treatments safer and more accurate. The market is growing because there are more high-risk pregnancies and more people want genetic testing. Local producers also make cheap, high-quality needles to help more people use them in cities and new locations.
The market is moving toward needles that work well with real-time imaging technologies like high-resolution ultrasonography. This new technology lets doctors do amniocentesis and other similar operations with more accuracy and less risk. Better sight when inserting a needle makes the procedure safer, lowers the risk of complications, and gives clinicians more confidence. More and more, hospitals and maternal-fetal centers want these high-tech solutions, which is pushing manufacturers to come up with new ideas and make needles that work with the changing needs of procedures.
Needles used for medical procedures including fetal blood transfusions and amnioreduction are getting more attention. More and more, doctors are using needles that can be used for both diagnostic sampling and focused therapy inside the womb. This development is part of a bigger change in how we care for mothers and babies, moving toward proactive interventions for high-risk pregnancies. Hospitals and specialized centers are starting to use multi-functional needles. This has led manufacturers to focus on making needles that are safe, precise, and flexible enough to suit both diagnostic and therapeutic purposes.
There is an increasing demand for needles that integrate diagnostic and therapeutic functions, such as facilitating both amniocentesis and fetal blood transfusions. Clinicians like tools that make procedures easier and faster. Manufacturers can meet the changing needs of maternal-fetal specialists, especially in high-risk pregnancies, by making multi-functional needles that are safe, precise, and easy to use. This will broaden the range of uses and increase market penetration in hospitals and specialized prenatal care centers.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 198.76 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.86% | 2033 Value Projection: | USD 220.78 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Becton, Dickinson and Company, Biopsybell S.R.L., Cook Medical Inc, CooperSurgical Inc., Integra Lifesciences, Laboratoire CCD, Medline Industries Inc., Medtronic Plc, RI.MOS. S.R.L., Rocket Medical Plc., Smiths Medical Inc., and Tsunami Medical S.R.L. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients