The Europe animal healthcare market is estimated to be valued at USD 16,965.7 Mn in 2026 and is expected to reach USD 24,679.6 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 5.5% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
The Europe animal healthcare market is expected to witness positive growth over the forecast period. Rising pet adoption in European countries such as the U.K., France, and Germany is driving the demand for animal healthcare products and services. Additionally, growing awareness among pet owners about animal health and increasing pet humanization will propel the demand for vaccinations and nutritional products. However, high costs associated with veterinary services and treatments may hamper the market growth to some extent. Overall, increasing focus of key players on product innovation and development of advanced medical devices for veterinary usage is expected to create new growth opportunities for this market.
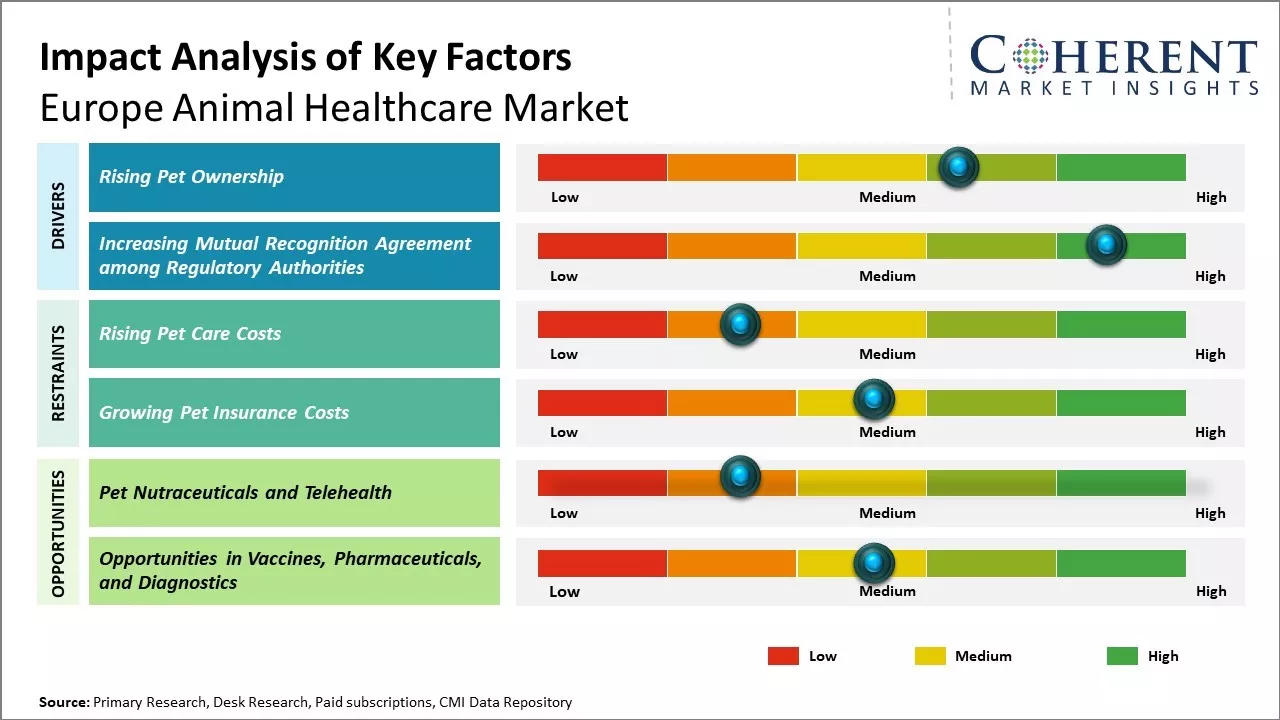
Market Driver – Rising Pet Ownership
With growing urbanization and modern lifestyles, ownership of pets is increasing at a significant pace across Europe. People are increasingly adopting pets such as dogs, cats, and other small animals due to companionship and need for affection. This has led to dogs and cats far outnumbering children in many European households in recent years. As a result, there is rising awareness about the health and wellbeing of pets. Pet owners understand that their pets also need proper healthcare, nutrition and preventive care similar to humans. The growing expenditure on premium food, insurance, treatment, and services indicates that pet owners want their companions to live long and healthy lives through quality healthcare interventions. As the trend towards nuclear families continues, citizens particularly in urban centers who lack extended family support find pets as a dependable source of unconditional affection. This will further fuel the demand for animal healthcare products and services.
Get actionable strategies to beat competition: Request Free Sample
Increasing Mutual Recognition Agreement among Regulatory AuthoritiesIncreasing mutual recognition agreement among regulatory authorities for the inspections of animal healthcare drugs is expected to drive the Europe animal healthcare market growth over the forecast period. For instance, in June 2023, the U.S. Food and Drug Administration and European Medicines Agency announced a U.S.-EU Mutual Recognition Agreement (MRA) for pharmaceutical good manufacturing practices inspections of animal drugs. By extending coverage to animal drug manufacturers, the FDA will be able to rely on inspection reports created by certain European authorities minimizing the need for additional animal drug facility inspections and freeing up regulatory resources. The MRA extends only to surveillance inspections of drug establishments, not to pre-approval inspections.

To learn more about this report, Request Free Sample
Market Challenges – Rising Pet Care CostsRising pet care costs are putting financial strain on pet owners across the region. Generic competition is increasing for many off-patent drugs, reducing prices and profit margins for branded products. Strict regulations from the European Medicines Agency also raise compliance costs. Demonstrating safety and efficacy of new products through clinical trials takes significant time and investment.
Market Opportunities – Pet Nutraceuticals and Telehealth
Pet nutraceuticals and telehealth present a compelling growth opportunity in the European animal healthcare market. Pet owners are showing greater willingness to explore nutraceutical supplements and functional foods that can provide additional health benefits to pets. The market has seen the launch of various supplement products targeted at joint health, skin and coat, digestive health, heart health, weight management and more for dogs and cats. The availability of these supplements through e-commerce websites and apps as well as mainstream retail stores has made them highly accessible solutions for pet owners.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
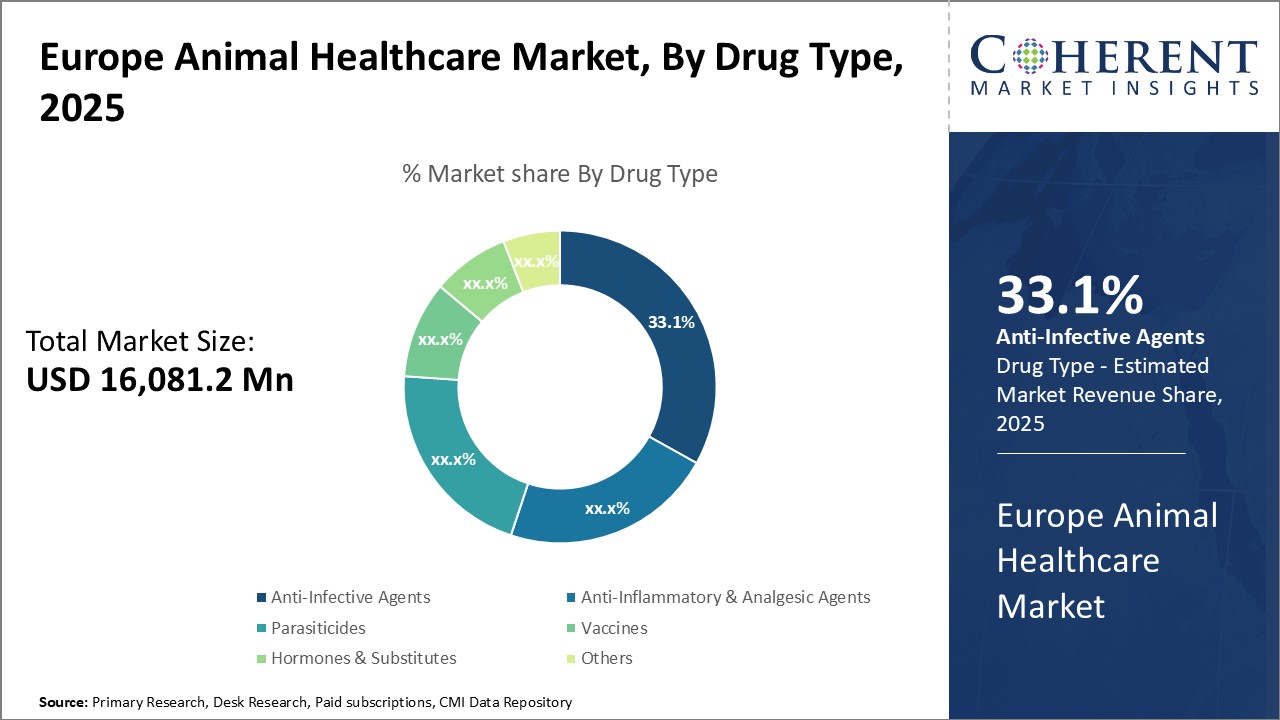
Insights, By Drug Type: Increasing Prevalence of Infectious Diseases in AnimalsThe drug type segment includes anti-infective agents, anti-inflammatory & analgesic agents, parasiticides, vaccines, hormones & substitutes, and others. The anti-infective agents subsegment is estimated to hold 34.1% of the market share in 2026, owing to the increasing prevalence of infectious diseases in animals. Anti-infective drugs remain in high demand due to the constant threat of bacterial, viral, and parasitic pathogens. Livestock are especially vulnerable to contagious illnesses due to their close quarters in agricultural facilities. Even with prophylactic vaccination programs, outbreaks continue to occur, requiring rapid medical intervention. Anti-infectives act as the frontline treatment during outbreaks, helping to control disease spread. Their utility in both disease response and long-term prevention strategies has made them a staple of veterinary practices. Veterinarians commonly prescribe anti-infectives for routine illnesses and to treat secondary infections stemming from other conditions. As new disease strains emerge and develop antimicrobial resistance, innovative anti-infectives addressing resistant pathogens will be crucial to sustaining this market segment's leadership.
To learn more about this report, Request Free Sample
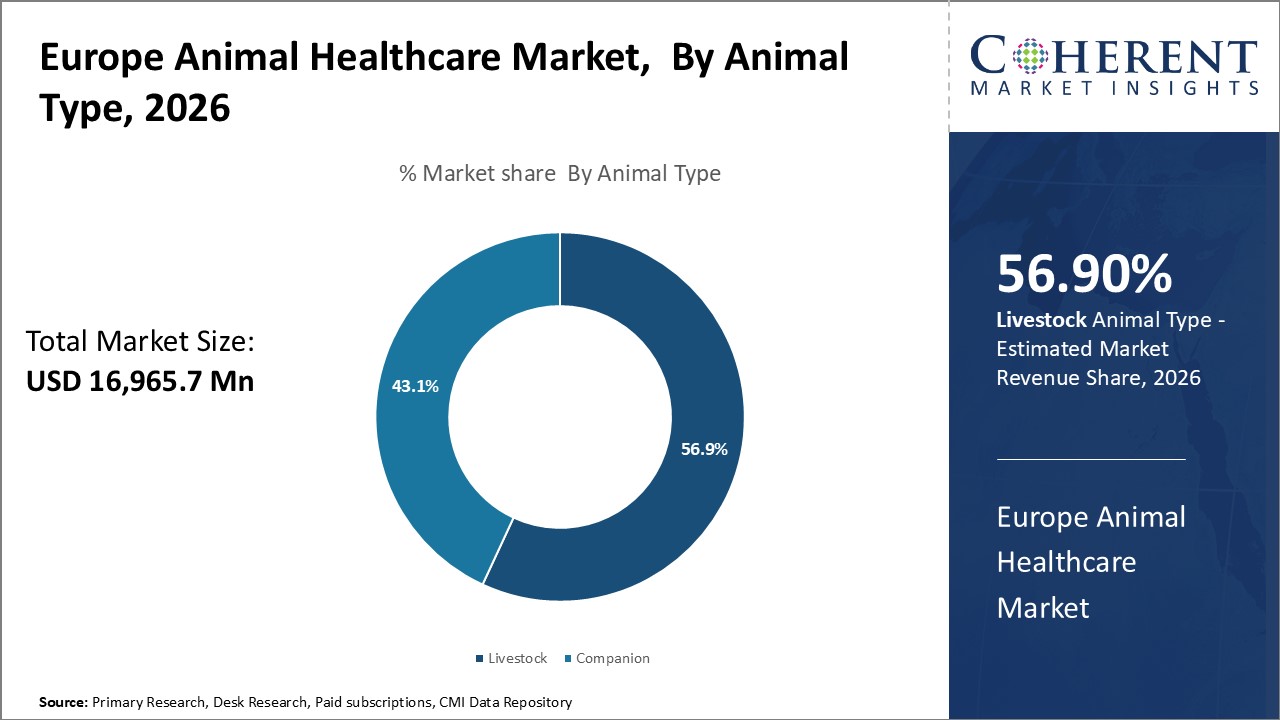
Insights, By Animal Type: Increasing Caring for Companion Animals
The animal type segment includes livestock and companion. The livestock subsegment is estimated to hold 56.9% of the market share in 2026 driven by increasing caring for companion animals. While companion animal healthcare is a growing field, livestock production remains a cornerstone of European agriculture. Treating livestock is a massive undertaking given the huge animal populations involved in meat, egg, and dairy production. Proper animal healthcare is necessary to maximize productivity and minimize economic losses from disease and mortality. Routine treatments for viral and bacterial infections are ubiquitous in livestock operations to prevent outbreaks. Vaccinations are also extensively used to provide immunization against common illness threats. Beyond disease prevention, growth promoters and endocrine treatments are utilized to accelerate maturation and optimize production attributes such as milk yield or lean muscle growth. Reproductive medications further aid in efficient breeding. The scale of livestock agriculture in Europe sustains heavy demand for these types of animal health products and services. Ensuring livestock health supports the livelihoods of farmers and the provision of inexpensive animal proteins.
Insights, By Indication: Animals’ Natural Susceptibility to Pathogens
The indication segment includes infectious diseases, pain management, auto-immune disease, and others. The infectious diseases subsegment is expected to have 30.2% of the market share in 2026 due to animals’ natural susceptibility to pathogens. Both livestock and companion animals lack complex medical infrastructures and are often housed in settings conducive to disease transmission. Communal living arrangements coupled with susceptibility to zoonotic diseases like rabies, avian influenza, and others demand prophylactic tools. Vaccines play a critical role in preventing infectious illness but are not a replacement for drug therapeutics.
Europe Animal Healthcare Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 16,965.7 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.5% | 2033 Value Projection: | USD 24,679.6 Mn |
| Segments covered: |
|
||
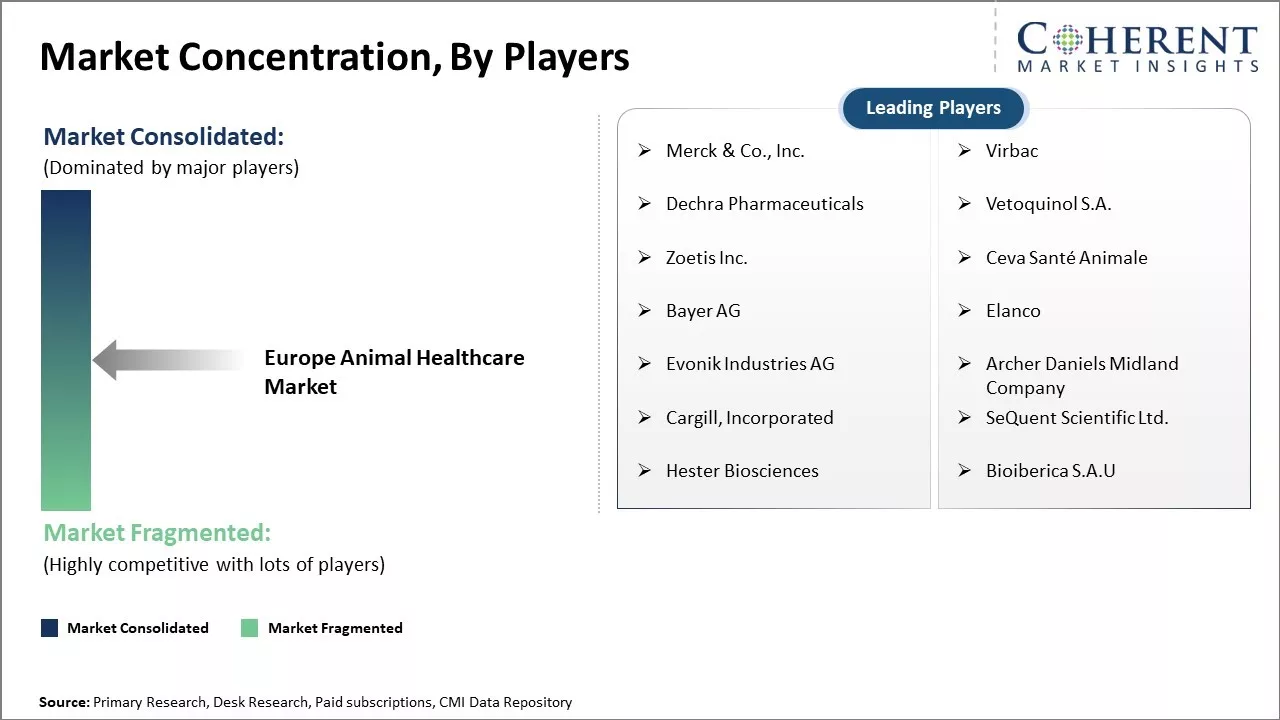
| Companies covered: |
Merck & Co., Inc., Virbac, Dechra Pharmaceuticals, Vetoquinol S.A., Zoetis Inc., Ceva Santé Animale, Bayer AG, Elanco, Evonik Industries AG, Archer Daniels Midland Company, Cargill, Incorporated, SeQuent Scientific Ltd., Hester Biosciences, and Bioiberica S.A.U |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: Animal healthcare focuses on effective animal health management by diagnosing and treating diseases. The outbreak of animal diseases restricts the trading of animal-origin products, increases the cost of disease management, and hampers the global economy. The Europe animal healthcare market refers to the market for various veterinary healthcare products and services for livestock animals as well as companion animals in the European countries. It includes pharmaceuticals, vaccines, parasiticides, feed additives, and other animal care products that improve animal health and productivity.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients