The Anterior Cruciate Ligament and Posterior Cruciate Ligament reconstruction devices market is estimated to be valued at USD 14,450 Mn in 2026 and expected to reach USD 23,660 Mn by 2033, witnessing a CAGR of 7.2% over the forecast period (2026-2033).
The anterior cruciate ligament (ACL) and posterior cruciate ligament (PCL) reconstruction devices market is driven by the increasing incidence of sports-related knee injuries, aging populations, and advancements in minimally invasive surgical techniques.
The anterior cruciate ligament (ACL) and posterior cruciate ligament (PCL) reconstruction is an operation to replace torn ligament to restore the stability of the knee joint. The ACL and PCL are tough bands of tissues in the middle portion of the knee that joins the tibia and the femur and prevents the two imperative bones from sliding or pivoting in an uncontrolled fashion.
|
Current Event |
Description and its Impact |
|
Technological Advancements in Orthopedic Biomaterials |
|
|
Regulatory Environment Changes and Market Access |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
|
Category |
ACL (Anterior Cruciate Ligament) |
PCL (Posterior Cruciate Ligament) |
|
Relative Injury Frequency |
Occurs 4–6× more frequently than PCL injuries globally |
Occurs far less often; represents a small fraction of ligament injuries |
|
Annual Global Injury Incidence |
2–2.5 million ACL injuries per year |
Significantly lower; no more than 5% of knee ligament injuries |
|
Annual Reconstruction Volume |
>400,000 ACL reconstructions performed worldwide each year |
Reconstruction volume is minimal due to low injury incidence |
|
Share of Knee Ligament Injuries |
Represents the majority of ligament injuries requiring surgery |
Accounts for <5% of all knee ligament injuries |
|
Impact on Device Demand |
Drives dominant demand for reconstruction devices |
Generates limited device demand due to low case numbers |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of product type, the Anterior Cruciate Ligament (ACL) procedure segment is expected to hold 83.4% share in 2026, due to there are substantially more ACL injuries around the world than PCL tears. Demand is driven by sports-related injuries, more people doing high-impact activities, and a strong clinical preference for surgical repair. Fixation devices that are always getting better help ACL stay at the top of the market.
In terms of end user, the hospitals segment is expected to lead the market with 68.2% share in 2026, because they have advanced surgical infrastructure, specialized orthopedic teams, and comprehensive postoperative care. Most ACL and PCL reconstruction surgeries are done in hospitals as fixing ligaments is difficult. Hospitals' dominant market share is strengthened by a high number of patients and high-quality equipment.
For instance, in September 2025, ValleyOrtho launched the BEAR implant, a new way to fix ACLs that is now used at Valley View Hospital and its surgery centers. Dr. Tomas Pevny leads the procedure, which allows for natural ligament regrowth with a less invasive approach. This leads to better outcomes and more advanced ACL treatment options in hospitals.
To learn more about this report, Request Free Sample
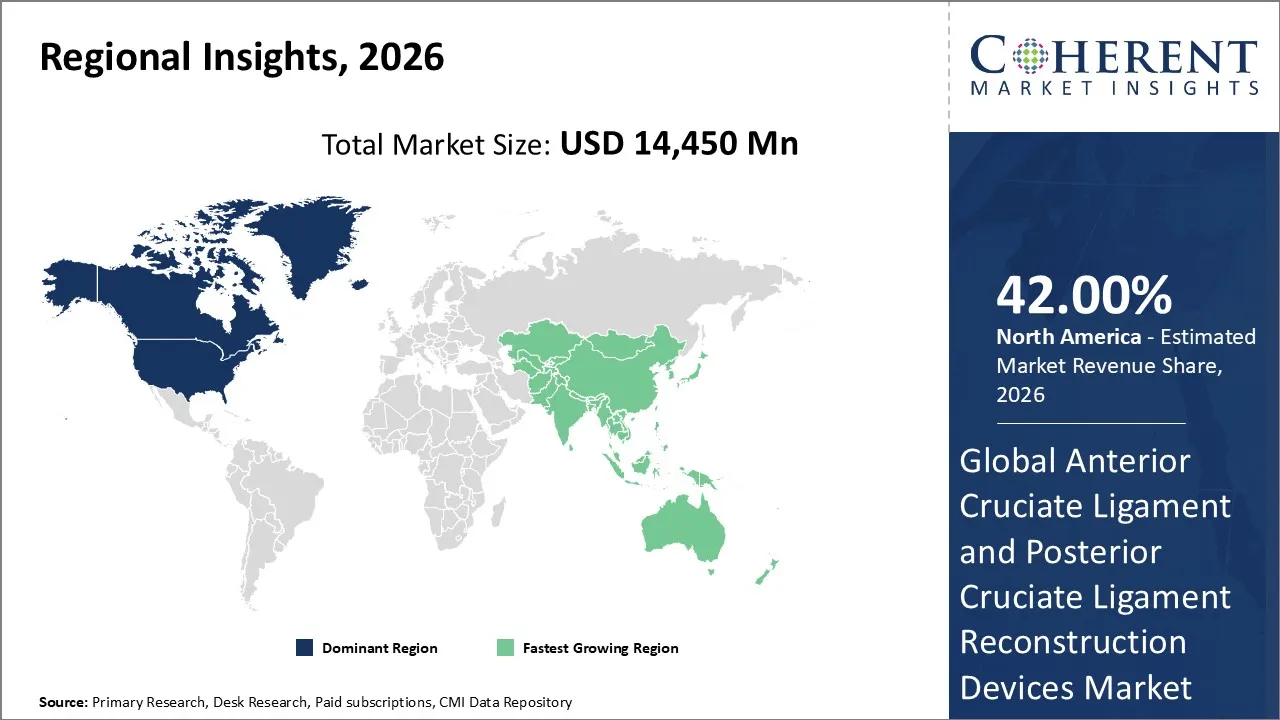
North America is expected to dominate the Anterior Cruciate Ligament and Posterior Cruciate Ligament reconstruction devices market with 42% share in 2026, because it has advanced healthcare systems, an excessive amount of people plays sports, and there are a lot of important medical device manufacturers there. Most ACL and PCL reconstruction surgeries are performed in hospitals and orthopedic clinics. This is because they have good insurance coverage and established rehabilitation programs.
At the same time, new bioabsorbable fixation systems and synthetic grafts are making them more popular, which is helping North America stay on top in the Anterior Cruciate Ligament and Posterior Cruciate Ligament Reconstruction Devices Market.
For instance, in March 2025, The FDA provided Miach Orthopaedics 510(k) approval, which means that the BEAR implant can now be used on kids, teens, and people with partial ACL tears. The implant allows for natural ACL healing and is the first major change from graft-based reconstruction in over 30 years. It was only available to people 14 and older with complete tears before.
Asia Pacific is expected to exhibit the fastest growth. ACL and PCL reconstruction surgeries are growing quickly in countries like China, India, and Japan. driven by more people are learning about sports medicine, more people are getting ligament injuries, and healthcare infrastructure is getting bigger.
Government programs and private investments in orthopedic care are speeding up the use of advanced reconstruction devices. The Asia-Pacific region is expected to grow faster than other regions, making it the most dynamic market segment in 2026.
The United States will have a strong demand for Anterior Cruciate Ligament and Posterior Cruciate Ligament Reconstruction Devices in 2026 because so many people play sports, the healthcare system is advanced, and insurance is widely available. The U.S. is the biggest market for bioabsorbable fixation systems and synthetic grafts due to the are always being improved.
For instance, in June 2025, At ValleyOrtho in Glenwood Springs, Colorado, Dr. Tomas Pevny addressed about new ACL repair technology, such as the BEAR implant. The new method is a biological alternative to traditional ACL reconstruction. It aims to speed up healing and improve outcomes for active patients in the region's ski-heavy community.
In 2026, China's need for Anterior Cruciate Ligament and Posterior Cruciate Ligament Reconstruction Devices is growing owing to more people are playing sports, more people are getting ligament injuries, and the country's healthcare system is growing quickly. China is the leader in orthopedic care in the Asia-Pacific region because of strong government programs and private investments in the field.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 14,450 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.2% | 2033 Value Projection: | USD 23,660 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Miach Orthopaedics, Arthrex, Smith & Nephew, Zimmer Biomet, Conmed Linvatec, MTF, RTI Biologics, Stryker, LifeNet Health, JRF, Parcus Medical, Medtronic, Tissue Regenix, Synthasome, Artelon, MedShape, Rotation Medical, CITIEFFE S.R.L, DJO Global Inc., Johnson & Johnson Services, Inc., and Meira Corporation. |
||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
As people get older, they are more likely to get musculoskeletal disorders like ligament degeneration and knee instability. Older people are more likely to fall and hurt their joints, which often need surgery to fix. This change in demographics is having a direct effect on the Anterior Cruciate Ligament and Posterior Cruciate Ligament Reconstruction Devices Market growth, because healthcare systems around the world are witnessing more demand for advanced orthopedic solutions that are specifically designed for older patients.
A greater number of individuals are playing sports and engaging in physical activities, which has led to a big rise in anterior cruciate ligament injuries, especially in younger and middle-aged people. ACL reconstruction surgeries are becoming more common because surgical techniques are getting better and rehabilitation protocols are getting better. The Anterior Cruciate Ligament and Posterior Cruciate Ligament Reconstruction Devices market demand is growing because hospitals and clinics need new implants and fixation systems to meet the growing number of procedures.
The creation of bioabsorbable fixation devices, hybrid graft technologies, and biocompatible materials is opening up new ways to improve surgical results. These new ideas lower the risk of complications like donor site morbidity and speed up recovery times, which makes them appealing to both surgeons and patients. As minimally invasive arthroscopic techniques become more common, the need for more advanced devices is expected to grow. This will make the overall Anterior Cruciate Ligament and Posterior Cruciate Ligament Reconstruction Devices Market forecast even stronger.
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients