Global anti venom market is estimated to be valued at USD 1,506.4 Mn in 2026 and is expected to reach USD 2,753.7 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 9.0% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
Rapid growth in biopharmaceutical industry and increasing cases of snake bites across various regions can boost demand for anti venoms. Rising investment by key players for development of monoclonal antibodies and recombinant anti venoms to reduce side effects can also drive the market growth. Growing availability of anti venoms, owing to capacity expansion by major companies along with ongoing clinical trials for advanced anti venom therapeutics can offer growth opportunities in the near future. However, short shelf life of anti-venoms and lack of awareness regarding snake bites in certain regions can hamper the market growth.
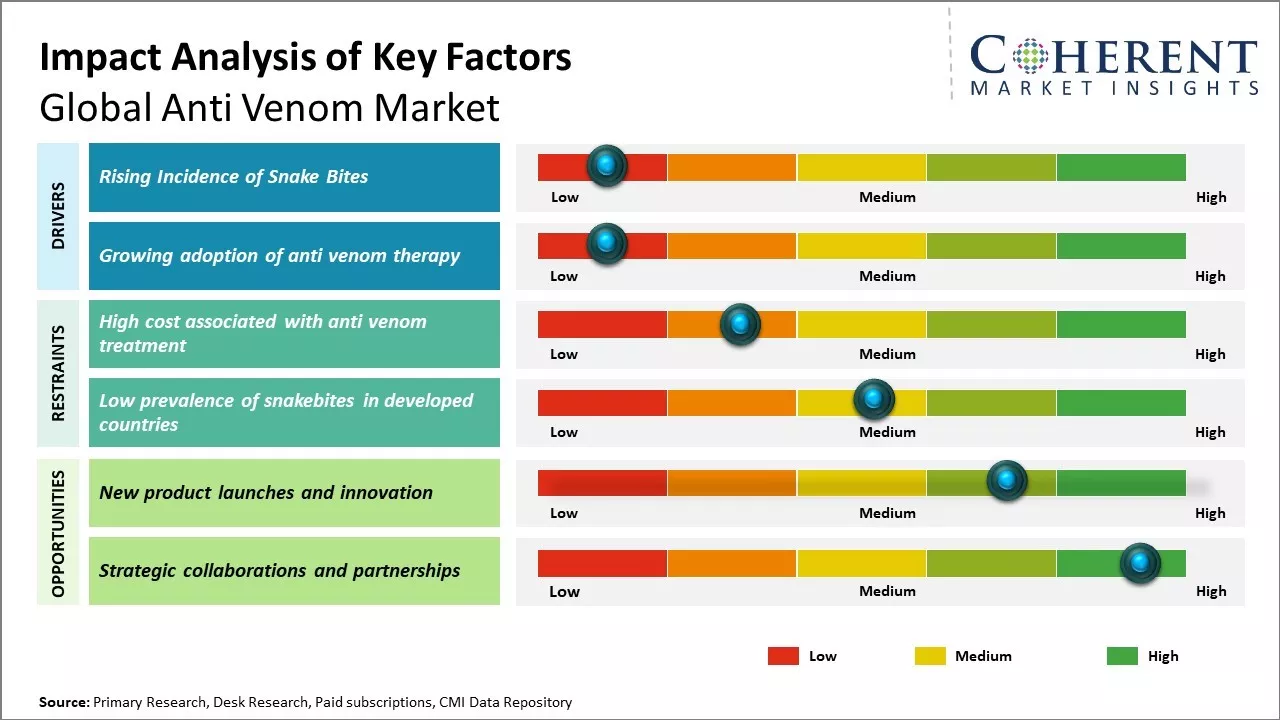
Rising Incidence of Snake Bites
With increasing human encroachment into natural habitats, there has been increase in interaction between snakes and humans. Several regions across the globe that are home to various species of venomous snakes have witnessed increase in reported cases of snake bites. Tropical and sub-tropical regions in Asia, Africa and Latin America have a high risk of snake bites due to the presence of various venomous species. Unfortunately, many of these regions have poor access to basic healthcare facilities and anti-venom treatments. According to some studies, it is estimated that around 5 million people suffer from snake bites annually around the world, out of which, around 1.8 to 2.7 million develop clinical manifestations of envenoming while 81,000 to 138,000 die due to snake bite. The true burden of snake bites is probably higher than reported owing to under-reporting from remote rural areas in developing nations. Some of the countries with highest incidence of snake bites include India, Sudan, Sri Lanka, Burma, Thailand and parts of Latin America. With growing population in these at-risk regions and continuous habitat destruction pushing snakes and humans into closer contact, there will be high number of snake encounters and resultant in the near future. This will boost demand for anti-venom treatments and immunoglobulins to treat clinical envenoming and prevent loss of lives.

Get actionable strategies to beat competition: Request Free Sample
Growing adoption of anti venom therapy
Increasing use of anti-venom therapy can drive the global anti venom market growth. More communities are recognizing the importance and effectiveness of anti-venom in treating snake bites and other venomous animal bites, which were earlier considered fatal. According to the World Health Organization (WHO), around 5.4 million snakebites occur annually, resulting in approximately 2.7 million envenomings with around 138,000 deaths worldwide. Increasing incidence of snakebites and scorpion stings boosts demand for anti-venom globally. Several medical and governmental initiatives are being taken to spread awareness about anti-venom therapy and make it more accessible in rural areas where majority of such incidences occur. For instance, a project undertaken in 2020 by the Ministry of Health and Family Welfare, Government of India aimed to set up five new sub-regional anti-snake venom units in different states to ensure adequate availability of anti-venom vials across the country. Improving accessibility to anti-venom in developing regions boosts higher consumption. Many biopharmaceutical companies are also focusing on developing anti-venoms for more exotic species and regions to expand treatment options.
Key Takeaways from Analyst:
North America and Europe dominate the market, owing to high healthcare spending and presence of major pharmaceutical companies. However, Asia Pacific especially India and Southeast Asia will witness fastest growth due to rising incidence of snake bites. Increased awareness about snake bite prevention and availability of anti venoms will boost demand for anti-venom. Developing effective anti-venom through modern purification techniques against various species can expand available treatment options.
Key players are investing in R&D to develop novel anti-venoms with less side-effects and higher efficacies. High costs associated with anti-venom production can hamper the market growth. Venom extraction and anti-venom manufacturing processes involve maintaining live snakes. This adds to production costs. Furthermore, stringent regulations pertaining to safety and efficacy halt introduction of new products. Social and environmental factors like poor healthcare infrastructure in developing nations can hamper the market growth in those regions. However, collaborative efforts between drug makers, healthcare organizations and local administrations can help expand access in emerging territories. Growing need for snake bite treatment can drive the global anti venom market growth over the next five years.
Market Challenges: High cost associated with anti venom treatment
The high cost of anti venom treatment can hamper the global anti-venom market growth. Anti-venom production is an expensive process involving milking venomous snakes, injecting horses with small doses of venom to stimulate antibody production, collecting antibody-rich plasma from the horses, and purifying and freeze-drying the immunoglobulins. This complex production pipeline makes anti-venom among the most expensive biological drugs available. Since snake bites are relatively rare in developed countries, anti-venom manufacturers have to rely on smaller markets in the developing world, where cost sensitivity is very high. The high prices of anti-venoms have pushed the life-saving drugs out of reach for many people who need them the most. According to a study by the World Health Organization in 2020, nearly one third of snake bite victims in South Asia are unable to afford anti-venom treatment due to the high costs involved. In Nepal, a single vial of anti-venom costs around US$ 150 which is prohibitive for many people, and people often have to buy multiple vials depending on the type and severity of the bite. The Nepalese government provides free anti-venom treatment at public health centres but limited availability and long travel distances prevent many people from accessing this subsidized treatment.
Market Opportunities: New product launches and innovation
New product launches and innovation can offer opportunity for global antivenom market growth. With increasing incidence of snake bites and scorpion stings across geographic regions such as sub-Saharan Africa, South Asia, and Latin America, there has been huge need for effective anti-venom treatments. According to the World Health Organization, 2.7 million envenomings and 100,000 deaths globally occur each year from snake bites alone. Many regions where the burden is highest still rely on older generation anti-venom products with questionable efficacy and availability issues. Developing novel formulations that have multi-specificity against various clinically relevant venoms and improved pharmacokinetic profiles could help address unmet needs in these underserved areas. Leading pharmaceutical companies have begun investing in recombinant anti-venom technologies that can neutralize a broader spectrum of venom toxins as compared to traditional products made from horse serum. For example, clinical studies are ongoing for an innovative anti-spider venom created using monoclonal antibodies isolated from people who were previously bitten. This "humanized" approach promises reliable supply, reduced allergenicity risk, and superior neutralizing abilities as compared to available therapies. As the recombinant space develops further, it may help decrease dependence on live animal harvesting methods, while improving access and health outcomes in venom-prone locales.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
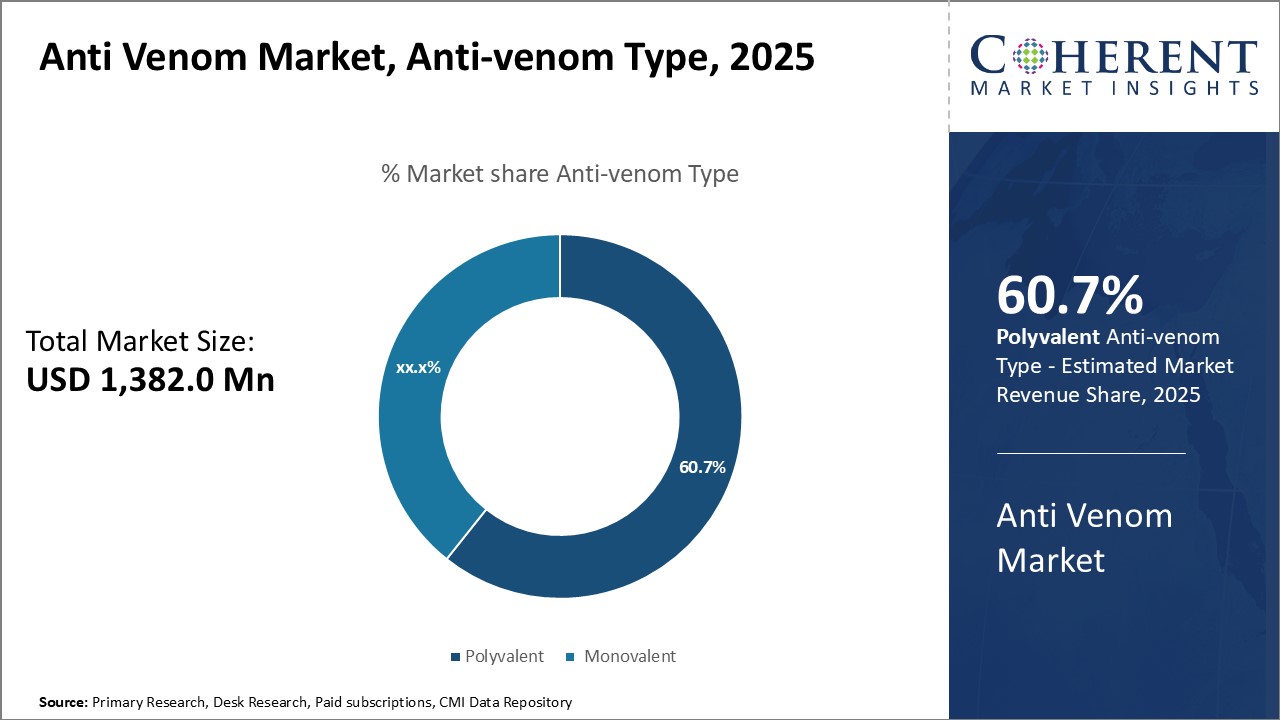
By Anti-venom Type - Rising Prevalence of Snake and Scorpion Bites
In terms of anti-venom type, polyvalent segment is estimated to contribute the highest market share of 61.7% in 2026, owing to increasing prevalence of multi-species venomous bites across various geographies. Polyvalent anti-venoms provide protection against bites from multiple species of snakes or scorpions, and thus, witness huge demand mostly in rural areas where people are more exposed to risk of bites from different types of venomous species. Given that the identification of offending species in such areas can be challenging, polyvalent anti-venoms offer higher clinical efficacy since these neutralize venoms from more than one species. Moreover, polyvalent anti-venoms are known to reduce adverse reactions associated with incorrect species identification as compared to monovalent anti-venoms. Considering the rising population living in remote and challenging terrains globally, there has been huge demand for broad-spectrum polyvalent anti-venoms during the forecast period.
To learn more about this report, Request Free Sample
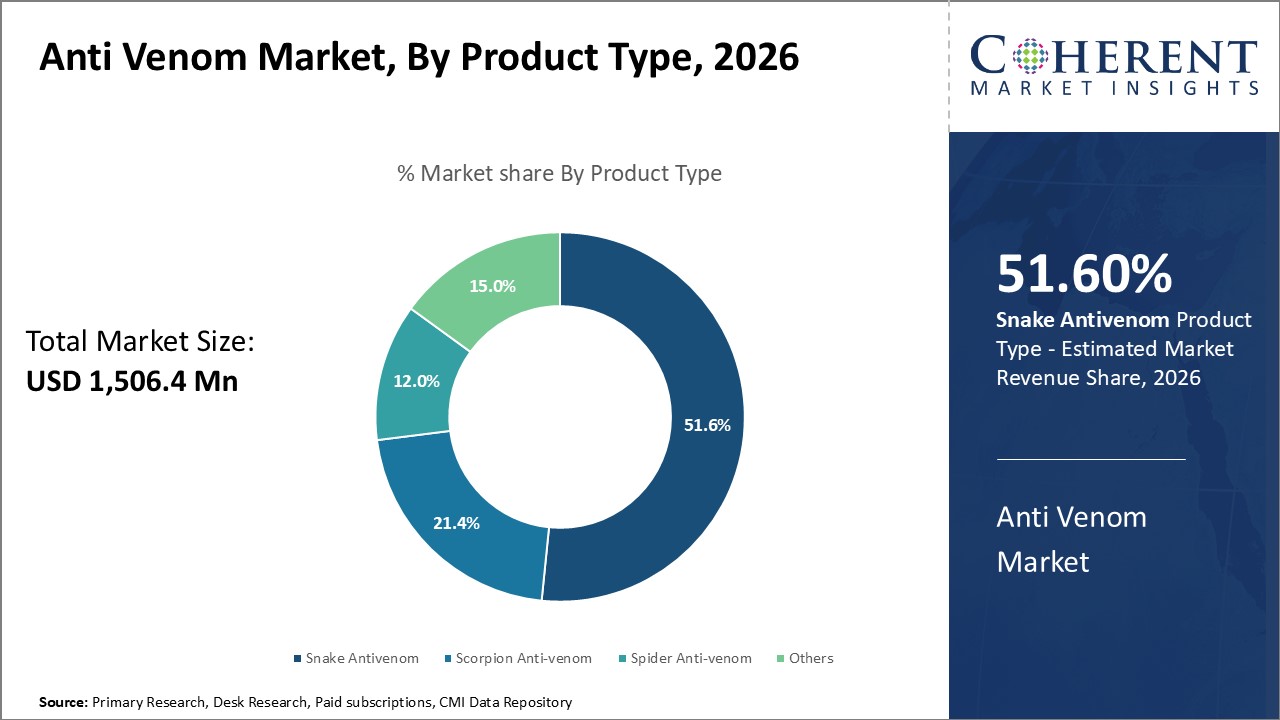
By Product Type - High Incidence of Snake Bites Boosts Snake Anti-venom Adoption
In terms of product type, snake anti-venom segment is estimated to contribute the highest market share of 51.6% in 2026, owing to the alarming incidence rate of snake bites around the world, especially across emerging economies in Asia Pacific, Latin America, Middle East and Africa. According to the data from WHO, snake bites kill about 138,000 people each year globally with highest mortality reported in South East Asia and Western Pacific regions. This can be attributed to factors such as warm tropical climate, farming activities in snake-infested areas, unawareness about first-aid, and limited access to affordable anti-venom treatment in rural communities. Thus, there is high dependence on anti-venoms for snake bite management in these developing markets. Moreover, rising agrarian activities and proliferation of snake species in new geographical areas have further increased human-snake encounters, thus, boosting demand for effective snake anti-venoms.
By End User – Hospitals & Clinics Emerging as Major End-users of Anti-venoms
In terms of end user, hospitals & clinics segment is estimated to contribute the highest market share of 41.6% in 2026, owing to the fact that majority of venomous bites require prompt management and administration of anti-venom under medical supervision, which is best provided in hospital settings. Although anti-venoms are also used in pre-hospital emergency treatments, hospitals offer better infrastructure, trained medical professionals, intensive care facilities and expertise needed for effective anti-venom therapy. Increasing number of tertiary care hospitals along with expanding healthcare infrastructure in emerging nations has enhanced access to anti-venom treatment over the years. This has reduced case-fatalities associated with pre-hospital delays and mismanagement of bites. Moreover, with rising healthcare awareness and affordability, more patients prefer hospitals over other healthcare facilities for anti-venom administration, thus, driving the segment growth.
Need a Different Region or Segment? Request Free Sample
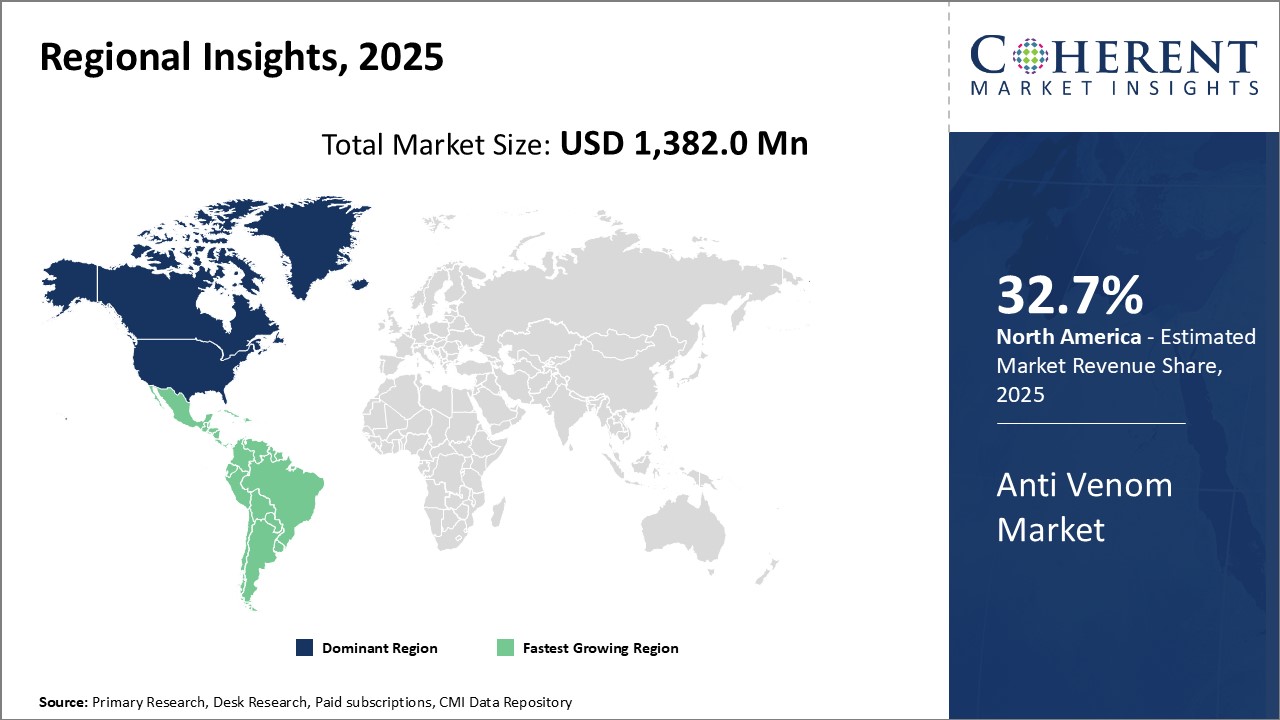
North America dominates the global anti venom market with an estimated market share of 32.7% in 2026.The large pharmaceutical industry presence along with concentration of many leading anti venom manufacturers drives the regional market growth. Strong research capabilities of companies based in the U.S. and Canada have resulted in development of advanced anti venom products. Moreover, high healthcare spending and availability of latest treatment options also drives the market growth. The governments in the region also provide favorable regulations and reimbursement policies for anti-venom therapies.
Latin America has emerged as the fastest growing market for anti venom. Several tropical and subtropical countries in Central and South America face risk of snake bites due to presence of venomous species. This has boosted demand for anti venoms from Brazil, Colombia and Mexico. The governments of these nations have also acknowledged snake bite envenoming as a neglected public health issue and are taking initiatives to improve access to treatment. Various international organizations are collaborating with local manufacturers to boost production and supply of affordable anti venoms. Growing healthcare infrastructure and increasing spending on serotherapies can drive the anti venom market in Latin America.
Anti Venom Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,506.4 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.0% | 2033 Value Projection: | USD 2,753.7 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pfizer, Inc., Merck & Co., Inc., Sanofi Pasteur, 4CSL Limited, Haffkine Bio-Pharmaceutical Corporation Ltd., Instituto Bioclon S.A. de C.V., Rare Disease Therapeutics, Inc., Vins Bioproducts Limited, Bharat Serums and Vaccines Limited, Serum Institute of India Pvt. Ltd., Incepta Pharmaceuticals Ltd., MicroPharm Limited, Laboratorios Silanes S.A., BTG plc, Vacsera, Indian Immunologicals Limited, AfrIPI (African Institute of Biomedical Science and Technology) |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: Global anti venom market consists of pharmaceutical products developed for treating venomous animal bites and stings. These products include both monovalent as well as polyvalent anti venoms targeted towards treating bites and envenomations from snakes, spiders, scorpions and other venomous fauna. The anti venoms work by supplying pre-formed antibodies that act against toxins present in the venom, thereby stopping its harmful effects on the body. The global anti venom market caters to the worldwide need for affordable and effective treatments against venomous animal encounters.
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients