Asthma Spacers Market is estimated to be valued at USD 2.26 Bn in 2026 and is expected to reach USD 3.60 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 6.9% from 2026 to 2033.
Analysts’ Views on Global Asthma Spacer Market:
The global use of asthma spacers is currently expanding due to increased awareness and product effectiveness. Asthma spacers, also known as inhaler chambers, are devices that help improve the delivery of medication to the lungs by reducing the amount of medication that is lost in the mouth and throat. They are particularly beneficial for children and elderly individuals who may have difficulty coordinating their breathing with inhaler use. The growing adoption of asthma spacers not only enhances medication efficacy but also promotes better asthma management and control worldwide. In addition, asthma spacers have been found to reduce the risk of side effects associated with inhaler use, such as oral thrush and hoarseness. Furthermore, their compact and portable design makes them convenient for use in various settings, allowing individuals to effectively manage their asthma symptoms on the go.
Figure 1. Global Asthma Spacer Market Share (%), By Spacer Type, 2026
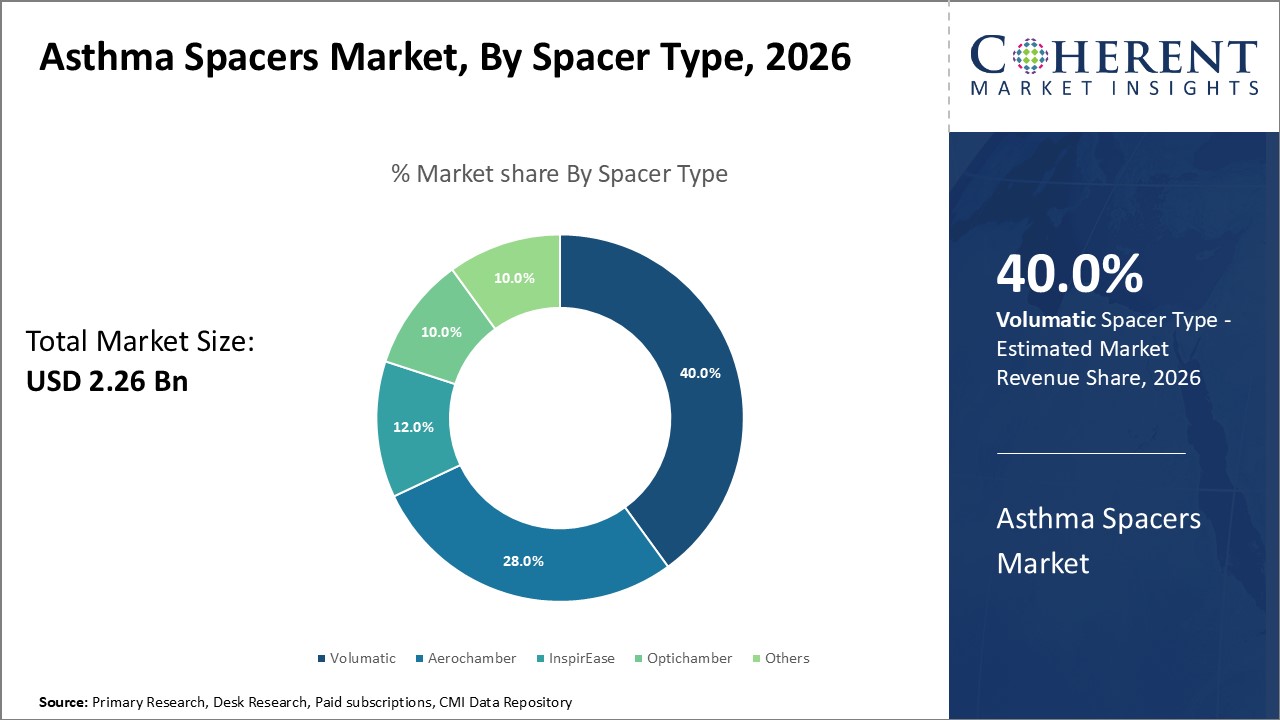
To learn more about this report, Request Free Sample
Global Asthma Spacer Market– Drivers
Increasing incidence of asthma patients globally: The prevalence of asthma in children was found to have decreased in low-income countries but increased in lower-middle-income countries. The prevalence in upper-middle and high-income countries was stable, found the Global Asthma Network (GAN) study, which shows how the worldwide burden of children’s asthma symptoms is changing. The University of Auckland, with partners including the London School of Hygiene & Tropical Medicine (LSHTM), conducted a research that included nearly 120,000 children and adolescents from 27 centers in 14 countries in phase 1. And, the findings also imply that asthma is a disease of Westernisation, with rates rising as countries with low and moderate incomes grow more Westernised. There is an urgent need for improved asthma management globally and better access to inexpensive asthma medicines.
Figure 2. Global Asthma Spacer Market Share (%), By Region, 2026
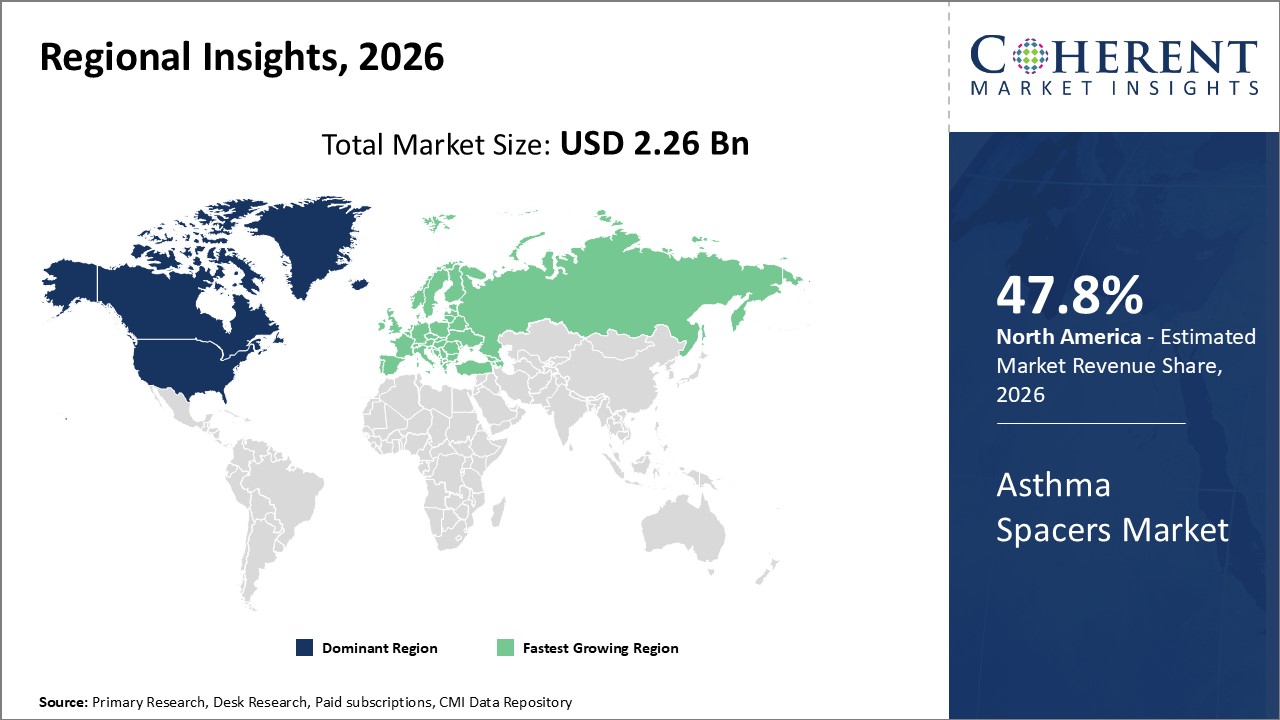
To learn more about this report, Request Free Sample
Global Asthma Spacer Market- Regional Analysis
Among regions, North America is estimated to hold a dominant position in the global asthma spacer market over the forecast period. North America is estimated to hold 47.8% of the market share in 2026. The global asthma spacer market is expected to witness significant growth in the near future, driven by the increase in the patient pool in the asthma. In 2019, around 7.8 percent of the population of the U.S. currently had asthma. Asthma is more common among females and children aged 5 to 14 years.
Asthma Spacer Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2.26 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.9% | 2033 Value Projection: | USD 3.60 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Trudell Medical International, PARI Respiratory Equipment, Inc., Koninklijke Philips N.V, GlaxoSmithKline Plc., Lupin, Medical Developments International, Visiomed Group Ltd., Cipla Inc., Clement Clarke International Ltd., Rossmax International Ltd., Luckys Pharma, Mediacare Equipments (India) Pvt. Ltd., Asia Connection Co., Ltd., and Laboratoire Protecsom SAS. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Asthma Spacer Market Segmentation:
The global asthma spacer market is segmented into spacer type, distribution channel, and region.
Among all the segmentation, the spacer type segment is expected to dominate the market over the forecast period, and this is attributed to the increasing preference of asthma spacers by patient rather than the use of asthma inhaler.
Global Asthma Spacer Market- Cross Sectional Analysis:
The market's key players are focusing on the development of asthma spacers, considering the current challenges associated with the inhaler and asthma spacer. These challenges include issues such as improper inhaler technique leading to inadequate medication delivery and difficulties in cleaning and maintaining the asthma spacer. By addressing these challenges, the key players aim to enhance the effectiveness and convenience of asthma treatment for patients. In addition, the development of asthma spacers also aims to improve patient adherence to medication regimens. Studies have shown that using a spacer can significantly increase the amount of medication delivered to the lungs, reducing the risk of asthma exacerbations and improving overall asthma control. Additionally, technological developments are being made to provide more portable and user-friendly asthma spacers, enabling patients to take them with them wherever they go for on-the-go relief.
Global Asthma Spacer Market: Key Developments
In September 2022, CanadaPharmacyOnline.com offers the over-the-counter aerosol chamber (inhaler device mask) for pets. Owners now don't need a prescription for their pet to purchase the product from Canada. However, it is recommended that owners only purchase an aerosol chamber for pets to treat and control pet asthma if the owner’s veterinarian prescribes it. It is used with metered dose aerosol inhalers such as Flovent and Albuterol for pets, which are prescribed by the owner’s vet.
Global Asthma Spacer Market: Key Trends
Global Asthma Spacer Market: Restraint
Global Asthma Spacer Market - Key Players
Major players operating in the global asthma spacer market include Trudell Medical International, PARI Respiratory Equipment, Inc., Koninklijke Philips N.V, GlaxoSmithKline Plc., Lupin, Medical Developments International, Visiomed Group Ltd., Cipla Inc., Clement Clarke International Ltd., Rossmax International Ltd., Luckys Pharma, Mediacare Equipments (India) Pvt. Ltd., Asia Connection Co., Ltd., and Laboratoire Protecsom SAS.
*Definition: People with asthma often use inhalers (also called puffers) to take their medications. A spacer (also sometimes called a holding chamber) is a device that makes using an inhaler easier and more effective. It attaches to the inhaler on one end and to a mouthpiece or mask on the other end. When the medication from the inhaler is released into the spacer, it's held inside until it's slowly inhaled through the mouthpiece or mask. It doesn't require the special coordination that the use of an inhaler alone requires. A spacer also helps deliver the medication to the airways instead of the mouth and throat, where it can work better with fewer side effects.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients