The Global Atrial Fibrillation Market is estimated to be valued at USD 28.44 Bn in 2026 and is expected to reach USD 47.49 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 7.6% from 2026 to 2033.
To learn more about this report, Request Free Sample
Atrial fibrillation is among the most common cardiac conditions and significantly raises the risk of strokes, heart failure, and other cardiovascular complications. The rising geriatric population globally is a major factor fueling the atrial fibrillation market demand, as older adults are more susceptible to developing this arrhythmia. The market is expected to experience steady growth, driven by increasing awareness, improved diagnostic tools, and therapeutic advancements. Innovations such as cryoablation are gaining traction as effective alternatives to traditional drug therapies. Additionally, progress in catheter ablation technologies is enhancing treatment precision and outcomes, further encouraging a shift toward minimally invasive procedures.
|
Current Event |
Description and Its Impact |
|
Regulatory Expansions for Pulsed Field Ablation (PFA) Systems |
|
|
AI Integration in Diagnostics and Monitoring |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The atrial fibrillation market has a strong pipeline driven by innovations in drugs, devices, and digital health. New anticoagulants like milvexian aim to offer safer stroke prevention, while advanced antiarrhythmics target better rhythm control. On the device front, companies like Medtronic and Boston Scientific are developing pulsed field ablation (PFA) and improved left atrial appendage (LAA) closure systems for safer, more effective treatment. Digital tools such as AI-powered ECG wearables and implantable monitors like Reveal LINQ II are enhancing early detection and remote care. These advancements, supported by clinical trials and regulatory backing, are shaping a dynamic future for the AF market.
In terms of type, the paroxysmal atrial fibrillation segment is expected to contribute the highest share of the market with 44.6% in 2026 owing to advancements in diagnosis and treatment options. Paroxysmal AFib is characterized by recurrent episodes that self-terminate within seven days. Though the symptoms can vary between individuals, common signs include palpitations, shortness of breath, dizziness, and fatigue. Traditionally, diagnosis depended on electrocardiograms (ECGs) conducted during symptomatic episodes. However, new diagnostic tools like implanted cardiac monitors can continuously monitor heart rhythm over long durations, increasing the likelihood of detecting asymptomatic episodes.
For instance, Mayo Clinic researchers report that timing of catheter ablation significantly affects atrial fibrillation (AFib) recurrence. A meta‑analysis of 43,711 patients across 23 studies revealed that those undergoing ablation within one year of diagnosis experienced notably reduced recurrence rates of 59% in paroxysmal AFib, compared to later intervention. This is further accelerating the atrial fibrillation market growth.
To learn more about this report, Request Free Sample
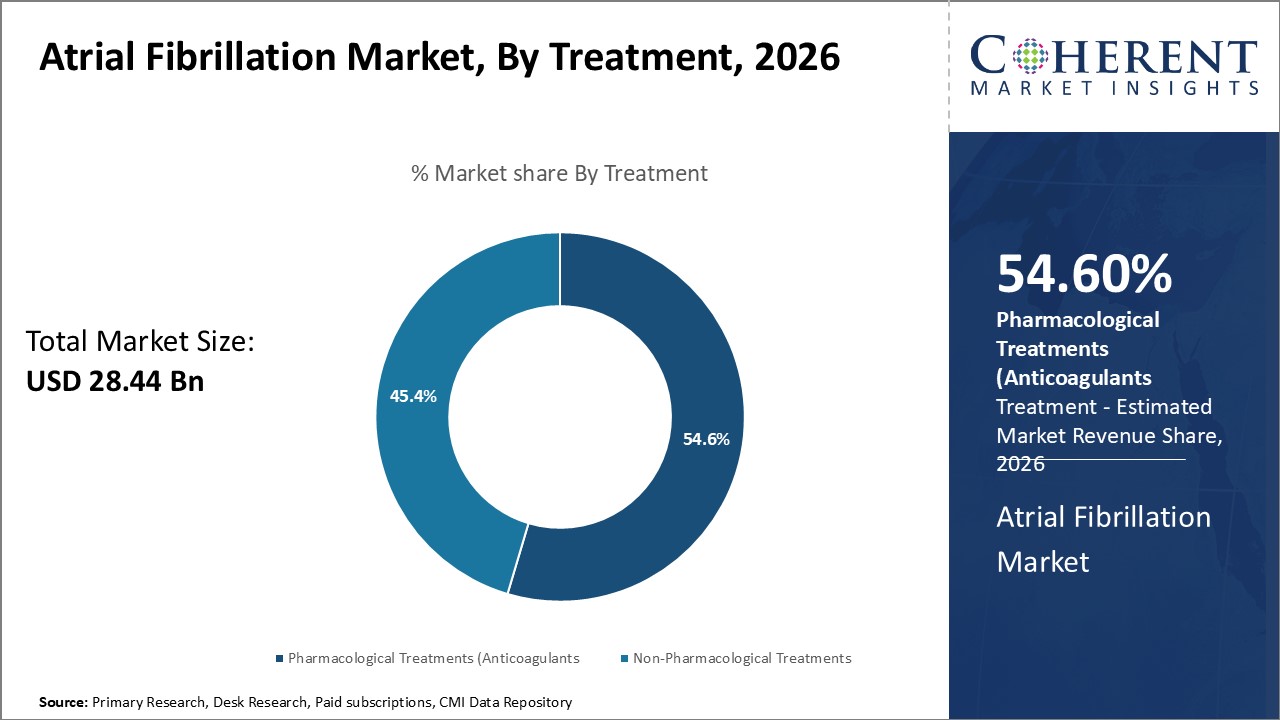
In terms of treatment, the pharmacological treatments segment is expected to contribute the highest share with 54.6% in 2026 owing to their widespread usage. Pharmacological options primarily rely on anticoagulants and antiarrhythmic drugs to address the risks as well as symptoms associated with, atrial fibrillation, Anticoagulants like warfarin and novel oral anticoagulants (NOACs) help prevent clot formation and reduce chances of stroke in atrial fibrillation patients. Antiarrhythmic drugs work to normalize heart rhythm or reduce arrhythmia burden. Their oral administration, convenience of self-administration, and reversible mode of action have made them a preferred first-line therapy. Additionally, advancements in drug delivery systems like nanoparticle encapsulation aim to overcome existing limitations like side effects or non-specific targeting. Combined with enhanced patient awareness and compliance, pharmacological treatments continue to dominate the treatment landscape for atrial fibrillation management.
In July 2025, The FDA has broadened the indication for Boston Scientific’s FARAPULSE™ Pulsed Field Ablation (PFA) System to include drug-refractory, symptomatic persistent atrial fibrillation, defined as AF lasting longer than seven days, providing a non‑thermal ablation option for patients’ intolerant to Class I/III antiarrhythmic drugs.
In terms of technology, the radiofrequency segment is expected to contribute the highest share of the global atrial fibrillation market with 31.6% in 2026. Radiofrequency ablation devices uses high-frequency electric currents delivered via catheter tips to generate heat, which eliminates arrhythmia-causing triggers in heart tissue. Compared to alternative techniques, radiofrequency ablation provides reliable energy delivery and precise lesion formation. It has proven safety and efficacy in treating various types and sources of atrial fibrillation. Technological advancements now allow the integration of 3D mapping systems, irrigated-tip catheters, and contact-force sensing capabilities for optimized, high-resolution ablation procedures. Hospitals and physicians also prefer radiofrequency for its flexibility in addressing complicated arrhythmia cases. With ongoing automation and reductions in procedure time, radiofrequency ablation for atrial fibrillation continues to gain popularity over laser, cryoablation, and other technologies. Its superior clinical outcomes continue propelling its leading share of the global atrial fibrillation market.
For instance, radiofrequency-based ablation delivers outstanding results in real-world atrial fibrillation (AF) care. Analysis of 2,470 patients from the REAL‑AF registry revealed an 85.7% success rate in remaining AF-free one-year post-procedure, with nearly 90% able to discontinue antiarrhythmic medications. Freedom from all atrial arrhythmias reached 81.6%, surpassing outcomes seen in randomized trials. The registry’s emphasis on best‑practice protocols for pulmonary vein isolation, reduced fluoroscopy, and optimized energy delivery and also yielded faster, safer procedures. Researchers now aim to replicate these advances in treating persistent AF, promising broader improvements in electrophysiology care.
In terms of end user, the hospital segment is expected to dominate the global atrial fibrillation market with the largest share in 2026, due to their ability to offer comprehensive and specialized treatment under one roof. Atrial fibrillation often requires advanced diagnostic tools such as ECG, Holter monitoring, and electrophysiological studies, which are readily available in hospital settings. Hospitals also have the infrastructure and skilled personnel to perform complex procedures like catheter ablation, cryoablation, and surgical interventions, making them the preferred choice for both initial diagnosis and ongoing treatment.
Additionally, AF is often associated with other cardiovascular conditions that require multi-disciplinary care, which hospitals are better equipped to handle. Emergency departments in hospitals are also crucial in managing acute AF episodes, particularly in elderly patients who form a major part of the AF patient base. These capabilities collectively make hospitals the primary hub for atrial fibrillation management.
In September 2024, Kauvery Hospital’s Alwarpet facility in Chennai launched fluoroless ablation, a pioneering radiation-free procedure for treating atrial fibrillation (AF). The minimally invasive technique replaces traditional X‑ray–based guidance with intracardiac echocardiography and 3D electro-anatomical mapping, significantly reducing patient and staff radiation exposure. Such developments are propelling the atrial fibrillation market demand.
To learn more about this report, Request Free Sample
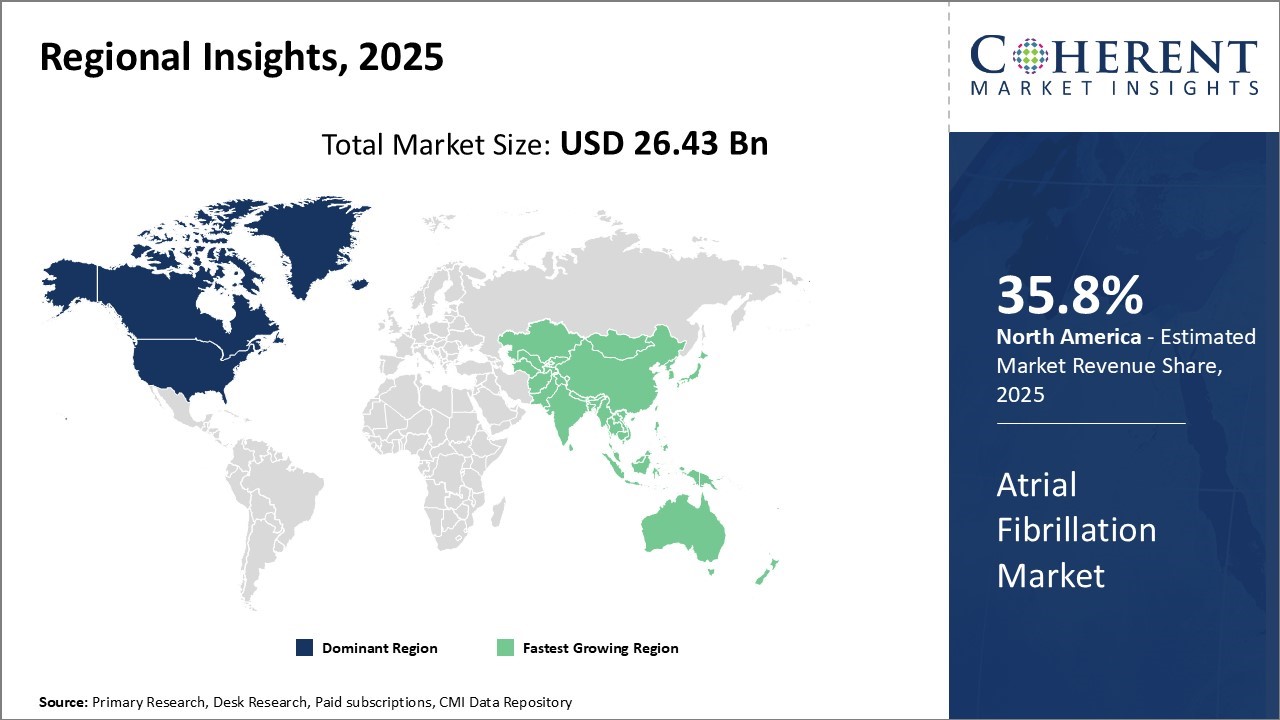
North America's dominance in the atrial fibrillation market—accounting for approximately 35.8% of the global share in 2026 is driven by a combination of technological innovation, early clinical adoption, and strong regulatory and reimbursement frameworks. Leading medical device companies in the region, such as Medtronic and Boston Scientific, have introduced groundbreaking technologies that are transforming atrial fibrillation treatment. For instance, Medtronic received FDA approval in late 2023 for its PulseSelect™ Pulsed Field Ablation (PFA) System, the first of its kind in the U.S., which offers faster and safer pulmonary vein isolation using non-thermal electric fields.
Additionally, in 2024, Medtronic secured FDA clearance for its Affera™ Mapping and Ablation System featuring the Sphere-9™ catheter, which combines pulsed field and radiofrequency ablation with real-time mapping. Similarly, Boston Scientific’s Farapulse PFA platform was approved in early 2024, offering shortened procedure times and improved precision. Major hospitals across the U.S., including Mount Sinai and St. David’s Medical Center, have already begun integrating these technologies into clinical workflows, favoring their minimally invasive, fluoroscopy-free capabilities. This is further proliferating the atrial fibrillation market demand.
The Asia Pacific region is emerging as a rapidly growing region in the atrial fibrillation market, commanding approximately 28.2% of global share in 2026, driven by its expansive patient base, rising healthcare investment, and growing awareness of advanced treatment modalities. A recent development underlining this momentum is Johnson & Johnson MedTech's launch of the VARIPULSE™ Pulsed Field Ablation (PFA) system across Asia-Pacific in July 2026. This platform, fully integrated with the CARTO™ 3 cardiac mapping system, is designed to streamline ablation procedures for AFib, offering precise, efficient, and low-fluoroscopy treatment—a major advancement for the region’s electrophysiology landscape.
In addition, clinical trials like China’s PROMPT-AF, conducted across 12 tertiary centers, are exploring refined ablation approaches (like vein of Marshall ethanol infusion) to enhance the effectiveness of pulmonary vein isolation (PVI), achieving significantly improved arrhythmia-free rates. Such trials are propelling the atrial fibrillation market share.
The U.S. leads in atrial fibrillation (AF) cases, with over 6 million affected as of 2023, driven by an aging population and lifestyle-related health issues. In response, healthcare providers are rapidly adopting advanced treatments like Pulsed Field Ablation (PFA). For example, Penn State Health and MedStar Washington Hospital Center implemented PFA systems in 2024, offering faster, safer procedures and same-day discharge. The FDA’s recent approvals have accelerated national rollout. These advancements highlight growing atrial fibrillation market in the U.S., as hospitals invest in technologies to improve outcomes and reduce AF-related complications.
Germany’s atrial fibrillation market is advancing rapidly due to strong government support, cutting-edge research, and technological innovation. In 2026, the German Centre for Cardiovascular Research received $8 million in funding to explore the role of immune cells in AF, aiming to develop more targeted therapies. Simultaneously, AI-powered ECG chips developed under the ARTEMIS project now enable real-time AF detection, enhancing patient monitoring. Additionally, a 2024 study from the University Medical Center Göttingen confirmed the safety and effectiveness of pulsed field ablation over traditional methods. Germany’s leadership in setting clinical guidelines further reflects its commitment to improving AF outcomes and boosting atrial fibrillation market demand.
Japan is seeing a rise in atrial fibrillation (AF) cases, especially among the elderly, driving demand for advanced treatments. In March 2024, Bristol Myers Squibb introduced a new anticoagulant targeting AF, reflecting the country’s focus on safer and more effective therapies. Additionally, Japan’s long-standing use of drugs like landiolol and apixaban, along with recent approval of reversal agents like andexanet alfa, highlights a strong commitment to improving AF management. These developments point to growing atrial fibrillation market driven by an aging population and supportive healthcare policies.

To learn more about this report, Request Free Sample
The Atrial Fibrillation market value is undergoing a significant paradigm shift, primarily fueled by a convergence of demographic trends, technology-led treatment precision, and persistent underdiagnosis in emerging economies. From a clinical and commercial standpoint, the electrophysiology segment—particularly catheter ablation and advanced mapping systems—is not just growing; it is redefining the treatment standard.
Despite the long-standing dominance of pharmacologic therapies like antiarrhythmic drugs (AADs), clinical inertia is increasingly giving way to interventional strategies. A notable example is the EAST-AFNET 4 trial, which demonstrated a 21% relative risk reduction in cardiovascular outcomes with early rhythm control, favoring ablation-based approaches. This study alone is reshaping guideline-based decision-making across Europe and the United States. Clinicians are now initiating rhythm control much earlier, reversing the traditional "rate-control-first" mindset that prevailed for decades.
Moreover, innovation in contact force-sensing ablation catheters, pulsed field ablation (PFA), and AI-enhanced mapping systems is creating a significant competitive advantage for technology-focused medtech firms. Boston Scientific’s FARAPULSE™ PFA platform and Medtronic’s Affera system represent not just product innovation but strategic pivots toward non-thermal ablation, which reduces esophageal injury risks—a key clinical concern that limited adoption of RF ablation in paroxysmal AF.
In terms of patient stratification, the market is witnessing a tilt toward persistent and long-standing persistent AF, where treatment gaps remain vast. Approximately 50% of patients with AF in the U.S. and Europe fall into this category, yet only 10–15% are treated with ablation—highlighting a stark opportunity for procedural expansion. Furthermore, with over 12 million Americans projected to be living with AF by 2030, the procedural volume ceiling is far from being reached.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 28.44 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.6% | 2033 Value Projection: | USD 47.49 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pfizer Inc., Boehringer Ingelheim GmbH, Bristol-Myers Squibb Company, Johnson & Johnson, Abbott Laboratories, Boston Scientific Corporation, Medtronic plc, AtriCure, Inc., Sanofi, Bayer AG, Gilead Sciences, Inc., Amgen Inc., Merck & Co., Inc., St. Jude Medical, Inc., and Cardiovascular Systems, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
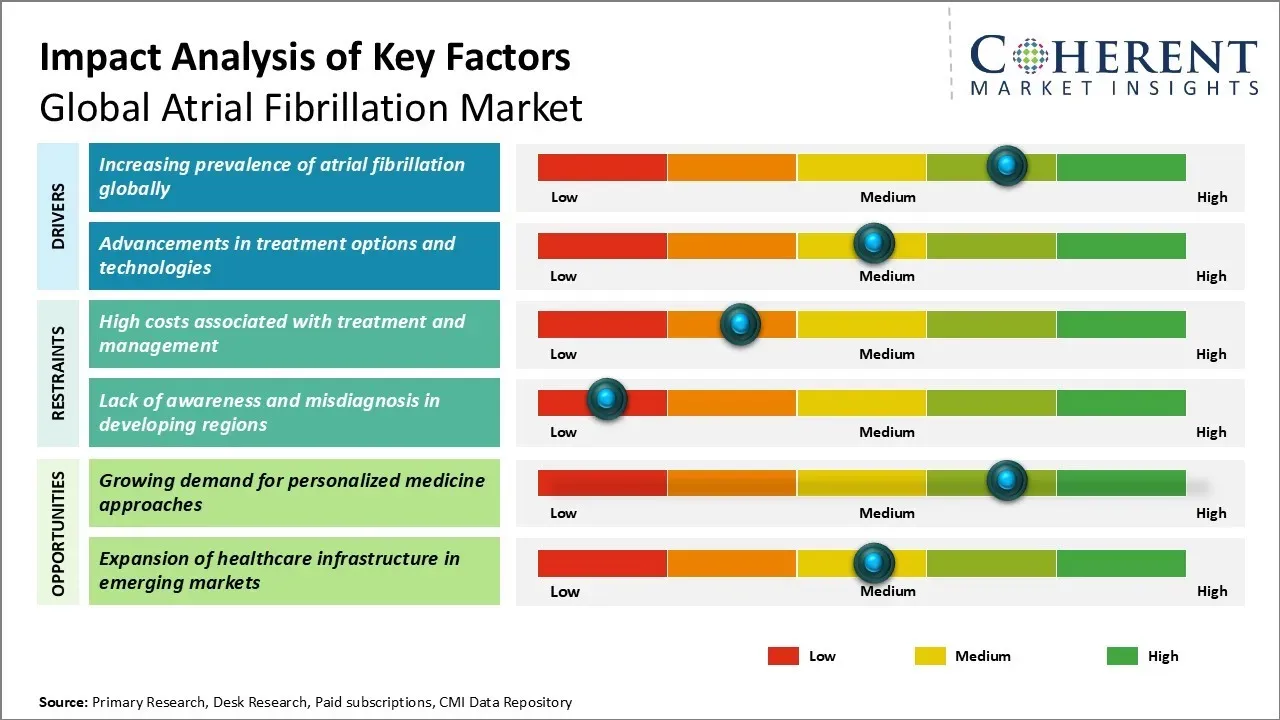
The global prevalence of atrial fibrillation (AF) is steadily increasing, significantly driving the growth of the atrial fibrillation market share. For instance, according to an article published by U.S. Centers For Disease Control and Prevention in 2024, Atrial fibrillation is the most common type of treated heart arrhythmia. As populations age, the incidence of AF rises sharply, particularly among the elderly who are more vulnerable to age-related cardiac changes. The growing elderly population globally is highly susceptible to developing atrial fibrillation due to age-related degenerative changes in the heart. Lifestyle changes such as rising levels of stress, unhealthy diet, lack of exercise, and increasing incidence of obesity are also contributing to higher cases of atrial fibrillation. The rising prevalence of atrial fibrillation and its link to associated strokes are anticipated to fuel market growth during the forecast period. According to National Library of Medicine, in Asia Pacific, atrial fibrillation cases are forecasted to reach 72 million by 2050, while in Europe, the number of individuals aged 55 and older with atrial fibrillation is projected to grow from 9 million in 2010 to 14 million by 2060, which is further accelerating the atrial fibrillation market growth.
The global atrial fibrillation industry sees notable opportunities arise from the growing demand for personalized medicine approaches. With increasing understanding of genetic and molecular basis of atrial fibrillation etiology, there is significant scope for tailored therapies and predictive diagnostics. Personalized approaches enable identification of high-risk patients, selection of best-suited drugs and treatment plans based on individual patient characteristics. This paradigm shift towards precision medicine is expected to deliver superior treatment outcomes.
Leading market players such as Pfizer Inc. are investing heavily in the development of companion diagnostics, biomarkers, and gene expression profiling tools. The trend is gaining policy support in many countries. As personalized care becomes standard practice, it can transform atrial fibrillation management by making it more efficient and cost-effective over the long-run. These advancements are expected to significantly contribute to the atrial fibrillation market forecast, opening new opportunities for innovation and growth.
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients