The Automated Endoscope Reprocessors Market is anticipated to grow at a CAGR of 8.1%, with an estimated market size of USD 780 Mn in 2026 and expected to reach approximately USD 1,350 Mn by 2033. This growth in the market is driven by stricter infection-prevention requirements for endoscope reprocessing, continued FDA focus on AER safety and effectiveness, and rising use of endoscopy in minimally invasive diagnosis and treatment.
In 2026, the Dual Basin AER segment dominates overall market share with 59% of total market share, with providers focusing more attentively on reprocessing systems that offer greater throughput, i.e., the ability to handle larger workloads to meet heavy endoscopy workloads. This preference has been further motivated by disease burden and demand for screening. The American Cancer Society predicts that there will be 158,850 new colorectal cancer cases and 55,230 deaths from colorectal cancer in the U.S. by 2026, and the CDC continues to recommend that adults aged 45–75 receive regular screening for colorectal cancer.
The use of dual-basin AER to streamline the sequencing of reprocessing tasks and reduce the pressure on instrument turnaround time at heavily utilized facilities and the emphasis on infection control that is increasing demand for systems that can deliver cleaner workflow separation, a more consistent application of high-level disinfection, and improved reprocessing reliability for complex flexible endoscopes.
As of 2026, Standalone AER systems will continue to dominate the market with a projected 62% market share due to the healthcare providers’ preference for standalone reprocessing systems, which they can deploy within many types of endoscopy facilities and enjoy operational flexibility. In addition, ongoing upper-GI disease burden supports continued increases in demand; The American Cancer Society has projected that, in 2026, there will be 22,530 new cases of esophageal cancer and 31,510 new cases of stomach cancer in the U.S., thereby continuing to support ongoing volumes of endoscopic diagnosis and intervention. (Source: Cancer.org)
The significant growth in the Standalone AER segment can be attributed to fewer challenges in integrating these systems into existing endoscopy departments, particularly in facilities that have not developed highly centralized designs. This situation has made it more practical for administrators to decide on replacing and expanding these systems. Additionally, the ongoing use of automated reprocessing systems drives growth, as there remains considerable potential for enhancing manual reprocessing methods. Consequently, these systems provide facilities with more reliable and validated disinfection processes, enabling them to meet and maintain compliance with standards related to staff consistency and infection prevention.
To learn more about this report, Request Free Sample
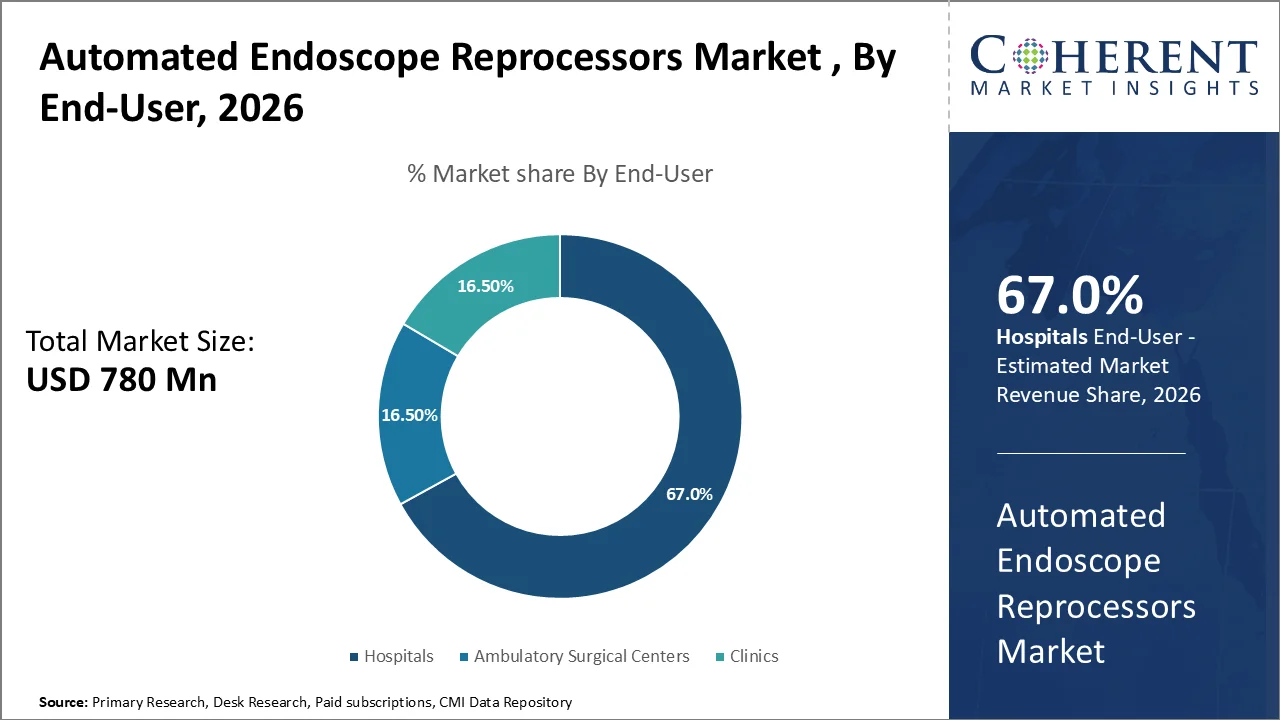
In 2026, hospitals hold the majority share of endoscope reprocessing equipment, accounting for 67%. This is due to their involvement in the highest number of complicated diagnostic and therapeutic endoscopic procedures, which necessitates a large volume of consistent reprocessing capacity. This leading position also reflects the economies of scale highlighted by the American Hospital Association's 2026 Fast Facts, indicating that U.S. hospitals had 907,216 staffed beds and a total of 35,658,583 admissions, thus creating the largest demand base for automated endoscope reprocessors.
There is an increasing reliance on the timely and efficient turnaround of flexible endoscopes, driven by the higher acuity of inpatients and the greater complexity of procedures performed in hospitals. Infection prevention is even more critical in hospitals compared to other healthcare facilities. Consequently, hospitals are investing in automated reprocessing systems that ensure a higher standard of disinfection, enhanced workflow control, thorough documentation, and improved safety for the reuse of flexible endoscopes.
On April 11, 2025, Portsmouth Hospitals University NHS Trust opened its new Elective Endoscopy Centre at Queen Alexandra Hospital. The trust announced that its endoscopy team performs over 15,000 procedures each year. The new facility was financed with £13 million in national funding and features dedicated decontamination space.
|
Current Events |
Description and Its Impact |
|
Stricter quality-system alignment for medical device manufacturers |
|
|
Wider acceptance of digital instructions for professional-use medical devices |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
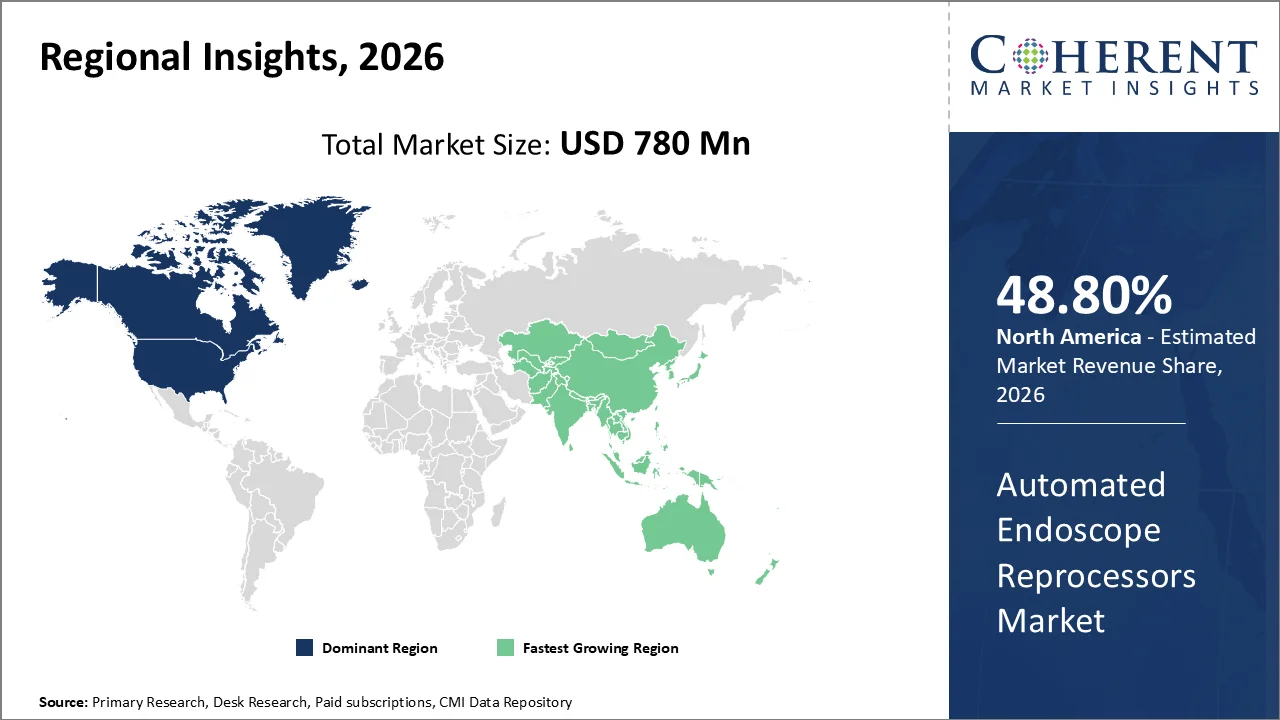
North America is projected to continue as the leading region in the Automated Endoscope Reprocessors Market, holding a share of 48.80% by 2026. The region's well-established endoscopy infrastructure, rising infection-control standards, and increasing adoption of automated reprocessing workflows support this. Additionally, North America upholds higher standards for procedure quality compared to other regions. The American Gastroenterological Association states that the multi-society guidelines utilized by U.S. endoscopists set a minimum standard target level of 90% adequacy for bowel preparation among endoscopists and facilities. Moreover, Ontario’s College of Physicians and Surgeons mandates that facilities performing endoscopic procedures install Automated Endoscope Reprocessors (AERs) and maintain sufficient AER capacity to meet operational requirements. These compliance-driven conditions in North America foster a strong demand for the structural replacement and upgrades of AERs. (Source: GASTRO)
The Asia Pacific region is projected to experience the highest growth rate in the Automated Endoscope Reprocessors Market, with an expected market share of 14.3% by 2026. This rapid growth is driven by several factors, including the expansion of the endoscopy ecosystem, increased availability of clinical training networks, and greater standardization of procedures throughout the region. For instance, the Japan Gastroenterological Endoscopy Society has reported nearly 35,000 members in Japan. Additionally, the Japan Endoscopy Database recorded 597,707 upper gastrointestinal (GI) procedures, 279,486 colon procedures, and 30,206 endoscopic retrograde cholangiopancreatography (ERCP) procedures in just one year. This significant procedural volume in the region is expected to sustain the demand for automated, high-volume throughput reprocessing systems in healthcare facilities across the Asia Pacific.
The United States has the largest and most established market for screening and guiding clinical practices related to reprocessing equipment in North America. This is primarily due to the increasing number of colonoscopies being performed. The American College of Gastroenterology recommends that average-risk adults begin colorectal cancer screening at age 45 and highlights that colonoscopy is the most frequently performed gastrointestinal procedure in the country. Together, these factors create a consistent demand for reliable automated reprocessing capabilities in both outpatient and hospital environments.
Japan is a significant growth market in the Asia Pacific region, characterized by a comprehensive and systematic endoscopy ecosystem that prioritizes quality. According to the Japan Gastroenterological Endoscopy Society (JGES), there are 20,988 board-certified fellows and 6,754 board-certified trainers, indicating a substantial number of specialists skilled in both routine and advanced endoscopic techniques. With a high concentration of professionals, an active national endoscopy database, and a robust culture of adherence to guidelines, Japan continues to invest in standardized automated reprocessing infrastructure.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 780 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.1% | 2033 Value Projection: | USD 1,350 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
STERIS Plc., Olympus Corporation, Laboratory Anios, Hoya Corporation, Getinge AB, Metull Zug AG, Steelco S.p.A., Cantel Medical Corp., Advanced Sterilization Products, Custom Ultrasonics, and Johnson & Johnson. |
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Major companies STERIS Plc., Olympus Corporation, Laboratory Anios, Hoya Corporation, Getinge AB, Metull Zug AG, Steelco S.p.A., Cantel Medical Corp., Advanced Sterilization Products, Custom Ultrasonics, and Johnson & Johnson.
On March 25, 2025, the FDA granted a first‑ever clearance for Nanosonics’ automated CORIS endoscope cleaning system, designed to clean the complex internal channels of flexible endoscopes and tackle biofilm linked to hospital‑acquired infections. ( Source: Fierce Biotech)
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients